* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Print - Circulation
Cardiovascular disease wikipedia , lookup
Remote ischemic conditioning wikipedia , lookup
Electrocardiography wikipedia , lookup
Heart failure wikipedia , lookup
Lutembacher's syndrome wikipedia , lookup
Cardiothoracic surgery wikipedia , lookup
Drug-eluting stent wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Mitral insufficiency wikipedia , lookup
Aortic stenosis wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Myocardial infarction wikipedia , lookup
History of invasive and interventional cardiology wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Coronary artery disease wikipedia , lookup
Dextro-Transposition of the great arteries wikipedia , lookup
A New Technic for Complete Correction of Transposition
of the Great Vessels
An Experimental Study with
By F. S. IDRISS, M.D.,
a
Preliminary Clinical Report
R. GOLDSTEIN, M.D., L. GRANA, M.D.,
D. FRENCH, M.D., AND W. J. POTTS, M.D.
I.
Downloaded from http://circ.ahajournals.org/ by guest on June 15, 2017
COMPLETE TRANSPOSITION of the
great vessels is a common congenital
cardiovascular anomaly. In this disease the
aorta with its coronary arteries originates
from the right ventricle, and the pulmonary
artery arises from the left ventricle. Consequently, the aorta carries venous blood; the
pulmonary artery, oxygenated blood.
Surgical correction of this malformation
has been beset with many difficulties and remains a challenge to the cardiac surgeon.
Many technics have been devised for its complete or partial correction.12 A logical
method for correcting this anomaly is retransposing the aorta and pulmonary artery. A
major technical difficulty in this approach
has been transfer of the coronary arteries.
Obviously, if they are left in place after transplanting the major vessels, the myocardium
will continue to be supplied with venous
blood.
Experimental Work
for switching the pulmonary artery and the aorta
with its associated coronary arteries. This technic
depends on the basic maneuver of creating an isolated aortic segment containing the ostia of both
coronary arteries. This segment is then turned
over and sutured proximally to the left ventricular
outlet (fig. 2, nos. 6 to 8) and distally to the
remaining aorta (fig. 2, no. 9).
The procedure was performed on 19 dogs with
use of extracorporeal circulation. Obviously, no
survivals would be expected in normal dogs when
the switch-over is performed. Nevertheless, when
the dogs were sustained by extracorporeal circulation at the termination of the operation their
heart beats continued strong and rhythmic. The
myocardium invariably remained oxygenated and
pink, and the coronary circulation appeared functionally intact. Figure 3 is a roentgenogram of a
dog's heart upon which the procedure was performed. The contrast material (Micropaque in
gelatin solution) was injected into the aorta, demonstrating the complete filling of the coronary
arteries.
The arterial switch-over was performed post
mortem and in situ on several patients that died
of transposition of the great vessels. Then two
clinical trials were carried out.
From a study of autopsy heart specimens with
transposition of the great vessels, the following
observations were made: The right and left coronary arteries arise from the posterior sinuses of
the aorta (fig. 1, nos. 3 and 4), approximately
5 to 7 mm. above the bottom of the aortic sinuses.
The proximal portion of the coronary artery segments is not embedded in the myocardium as is
usual in the normal heart. At their origin the
coronary arteries are separated from the mnyocardium by a small triangular space filled with
fat and areolar tissue, which facilitates their dissection.
These observations led us to develop a technic
From the Department of Surgery, The Children 's
Technic
To decrease the chanees of confusion, we are
giving technical details of this procedure as applied to the human heart with transposition of the
great vessels. Figures 1 and 2 depict the operation as it was performed in our two clinical trials.
The procedure as originally developed on dogs
is slightly different from the one used in patients
that is described below.
1. The chest is entered via a longitudinal sternum-splitting incision.
2. The thymus is excised and the pericardium
is opened widely.
3. Umbilical tapes are placed around the superior and inferior venae cavae and the femoral
artery in preparation for circulatory bypass.
4. The ascending aorta is dissected distally to
the level of the ligamentum or ductus arteriosus.
Proximally, the root of the aorta is freed from
the fat and thin epicardium covering it.
Memorial Hospital, Chicago, Illinois.
Aided by a grant from the Chicago Heart Association.
Dr. French was supported in this work by grant
HF-A, 766 from the National Heart Institute, U. S.
Public Health Service.
Circulation, Volume XXIV, July 1961
5
IDRISS ET AL.
6
Downloaded from http://circ.ahajournals.org/ by guest on June 15, 2017
Outflow-volve Of
t
-rig vehicle
Site a?
Aortic segment
l
Ofmonw a.
Out flaw vilve of left -veit.
~ ~ ~ ~ ~-. tcoronary crr't.
R. coronR~~~coror
art.
-etcopr
Outdlow valve of right ventricle
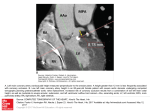
Figure 1
No. 1. External anatomy of a heart with transposition of the great vessels. No. 2. The
dissection of the aorta, pulmonary artery, and left coronary artery is completed. The
scissors are clearing the triangular space between the proximal portion of the right
coronary artery, aorta, and myocardium. No. 3. The aortic clamp is in place and the
first incision is being completed. The interrupted line demonstrates the site of the second
incision. No. 4. The second incision is being completed. No. 5. The aortic segment containing both coronary ostia is shown. The segment is pulled to the right to demonstrate
the outflow stumps and valves of the left and right ventricles.
Circulation, Volume XXIV, July 1961
7
CORRECTION OF TRANSPOSITION
OtloCsw valve o{right ventticle
Downloaded from http://circ.ahajournals.org/ by guest on June 15, 2017
cortn. art
Outflow valve of
rPght ventricle
Lef{ coronary arteiy
Figure 2
betwee n the isoiatel (tortic segment and th(
first
the
anastonlosis
of
No. 6. Beginning
outflow stump of the left rentricle. No. 7. Late)val v iev of the hear(1t (lmd great Vessels
demonstrating how the aortic segm}ienit is turne(d Ol'(C') to fit the ouitflow stttnlp of the left
Opi(ted. No. 9. The first ainastolmnosis
ventricle. No. 8. The first aniastom}iosis bei)ng cn
completed acl the seconid anastomnosis start( 4. Notice that the (lortic segmeniewt is conipletel!, tn med upside dowun. No. 10. The (con tin11nit!, of th e systemciccircuit is co ilnpete (1.
The aortic clamp is removed. The pnlnlonicia1!atery is pflfled ov.er and sututrieed to the
outflow stump of the right ventricle. No. 11. Thie swcitch-over is cniopletedi.
Circulation, Volume XXIV,
Jutly
1961
IDRISS ET AL.
8
Downloaded from http://circ.ahajournals.org/ by guest on June 15, 2017
This second incision completes the creation of an
isolated a1tor1tic segminent, containing the ostia of
both corona.ry arteries. The average length of this
segolleiit is allout 8 1miii.
11. The pulmonary artery is transeeted about 1
mm. distal to the valve commissures (fig. 1, no. 5).
(C'are should be taken niot to injure the valves
wenl the prulmonay arter'y is tranli.stiected. These
valv-es ultiima-1tely become svstedic. The lenlgth of
arteryl'N stump (outflow
the proxilalapl
st1111(p of the left velltricle) is of conlsiderable
imupoiltanelxe. Too long a 4-tumllp faRvor's the plrOduction of reg1ritation; too <lort a stunllp allya illnrse the v 1lve l('flets.
12. With the (0oul'\ ,arteiies us('d a1s thealXiS,
the ao(r1.tic( Segmn111t1lt is tinneld over to lit thie left
venltriculal outflov st14umnp (ti(g. 2, 11(0. 7) . An
aiastomilosi' is then performed betwseen the .oarti c
seg(.iiieiit aidl the left ventricular. stlmuil) N itlh (conItimmous 5-() silk suture (fig. 2, nos.6(, 7, and S).
Figure 3
omle0])( (( flling of tile ('0(('lolte'lies is sholen
('0 (1 tycl0(gl'l
iJl t[Il
f ((o/g's 71(c it (PO w('ll elh
/1(1
Th71e ('oiOp)(iqelf
))ocedl(e
per 'ffo rflm
,ss)l ittio~i wa's inl jeX tt'd inltO thle' aor}taz.
oh
is
(l.
The
ai'teries are dissected fre>e with
of iliolilie(d Potts' teill stllisv scissors (11-g-. 1,
2) The arilteries should not ble c(omipletely denuded (If their ad veentitma, hich sulpp)orts theill
and. also carries nutrient vessels to the aortic seg.
use
a
n.
6. The
artery is dissected and freed
left divisions as1 far dlista.llv'
pulmiionar'v
with its ri'Lt
and
as
It is ilml)ortant to perholria1
thoolrloug
dissectiolln to lpmevent undlue tension on the suture
line when the pulmuonamy artery is 1)ulle(d over the
ao'ta (fig. 2, no. 10).
7. After adequate lhel)p.ainiizaitioin, the superior
.;m1d inferior venae cavaise, fenmral .artery, and left
.atrium are cannulated, .abir extrac(orporeal circula-
p((ss*<;h(l
a
tion is initiated.
8. The aorta
atretation
is
clamip,
cross-elamped with
producing, iseheimic
a
Potts'
co-
miyoardial
afrrest.
distal
9. The aorta is tranilsecte('d al)out 5)
to the coronary ostia (fig'. 1, no. 3).
10. A second transecting incision is made in the
aorta proximal to the coronary ostia (fig. 1, no. 4).
This incision is placed so as to leave a 3- to 4-mm.
lip proximal to the coronary ostia and at the samce
time to preserve as much as possible of the outmm.
tlow valve of the right ventricle.
i
nalftomnosn is tlenic performed
1:3. Tlie e(eoiid
)t'tccit'i tine .()1rtic seinlenit anid the (listal aom'ta
(fig'. 2, no. 9), so as to restore continuity of the
s!vste4 c( irlcuit with the left ventricle.
14. The aotie clamp) is thlen removed, alloing
tie c()0r1ia.iv arte lies to l3e l)erflused cand the heart
to 'et'uio onItractions. The duration of anioxic
a rrlsest is lit aited to .ani average of 30 to 3,5 minutes.
Becaunlse )of llel)arinization, bleeding(, from the
aalstoilotic site is a, mllajOr problem. Meticulous
iihiceiiieiit Iof the sutur'es helps to prevent excessive l)dillg. Gelfoaiii imloisteied with topical
l)oviiie, tlriombil placed over the suture line aids
in stoppinlg bleeding from needle holes. When
bleeding- is excessive, thromibin is w,,iaslhed out anird
b)ecome(s ineffective. We found it advaiitageous to
(ro''Ss-( clamip the aior'ta for 2 to 3 miniutes so that
(lottillng m1la1y oeccur.
15. The pulmiioni,-airy artery is analstomiiosed to tlme
outflo\w stumllp of the riglht ventricle (fig. 2, no. 10),
tol colJp)lete the switch-over (fig. 2, no. 11).
16. Circulatory bypass is stopped.
1)ue to the aim,.ltomic differences betweeni the
dog's h('amt with norimial outflow tracts and the
humanl heart +witl transposition of the gr eat yessets, the procedure described above is slighltly differenit flomr the one originally developed on dogs.
The coronary arteries of the normal dog's heart
.are embedded in the myiA~ocardiumii. They are more
(lifficult to dissect than in the huinan heart with
transposition of the great vessels. Furthermore,
the left (or0oniaryN. artery is covered by the puh11uenarv artely. We found it advantageous in dogs
to perform as much as possible of the dissection
lnId then to start bypass and transect the pulmonairy artery. This imaneuver facilitates the exposure and the completion of the dissection of the
left coronary artery.
Circulation, Volume XXIV, July 1961
CORRECTION OF TRANSPOSITION
9
Regurgitation was a frequent occurrence after
completion of the switch-over in dogs. A single
suture placed as shown in figure 4 helps to control
any valvular insufficiency. Regurgitation was not
encountered in the clinical trials.
Downloaded from http://circ.ahajournals.org/ by guest on June 15, 2017
Case Reports
Two patients with transposition of the great
vessels were operated upon at The Children's Meinorial Hospital in Chicago with use of this procedure.
Case 1
J. S. was a 7-year-old white boy with severe
cyanosis since birth and severe clubbing of the
digits. Clinical diagnosis of transposition of the
great vessels was made soon after birth. Six months
prior to surgery there was progressive clinical deterioration with decreasing exercise tolerance. Cardiac catheterization then confirmed the diagnosis
of transposition. A bidirectional shunt at the atrial
level and an intact ventricular septum were found.
The pressure in the right ventricle was 108/8 mm.
Hg, in the aorta 108/80 mm. Hg, in the left ventricle 37/8 mm. Hg, and in the pulmonary artery
37/15 mm. Hg. The arterial saturation was 69
per cent. The estimated pulmonary blood flow was
7.4 L./M.2/minute and the systemic blood flow
5.9 L./M.2/minute. The electrocardiogram demonstrated right ventricular and right atrial hypertrophy. The chest roentgenogram showed cardiac
enlargement and increased pulmonary vascularity.
A cineangiocardiogram corroborated the diagnosis
of transposition of the great vessels.
After adequate digitalization, the operative procedure described was performed on June 29, 1960.
After the continuity of the aorta with the left
ventricle was established, the aortic clamp was removed and the heart resumed its beats spontaneously. The heart contractions were strong and
rhythmic, and the myocardium was uniformly pink.
The electrocardiogram showed no evidence of coronary insufficiency at that time. The pulmonary
artery anastomosis was then performed, and the
interatrial septal defect was closed with a continuous suture through a right atriotomy.
The extracorporeal circulation was then stopped.
The heart continued normal contractions for a few
minutes, then started to slow down and the left
ventricle became markedly distended. Cardiopulmonary bypass was resumed, and the heart rate
increased and the cardiac contractions again became vigorous. There was no evidence of aortic
valve regurgitation. Stopping and resuming bypass several times reproduced the same sequence
of events; good cardiac contractions resulted on
bypass, and weakening of the contractions with
left ventricular dilatation occurred when off bypass.
Circulation, Volume XXIV, July 1961
Figure 4
Suture placed in the myo(ardiump of the right ventricle in dog ait the completion of the operation to
prevent herniation of the valves with the production of regurgitation.
The sutured interatrial defect was reopened with
the hope that it would decompress the left side
of the heart, but to no avail. The patient died
of acute left ventricular dilatation and failure.
Postmortem examination showed a thin-walled and
dilated left ventricle. The right ventricular wall
was markedly hypertrophied, three times the thickness of the left ventricular wall. The coronary
arteries and ostia were patent to probing.
Comment
In retrospect, this patient may not have been a
suitable candidate for this procedure. The age
and size of this patient and his general satisfactory
condition coupled with the fact that he had an
intact ventricular septum were strong factors in
our choice. Although the left ventricular pressure
was low, it was anticipated that the left ventricular
myocardium could support the pressure postoperatively, since it had been carrying a considerably
increased flow load. We now think that for this
type of procedure, the left ventricular pressure
should approximate or equal the right ventricular
pressure.
Case 2
M. B. was a 31/2-month-old white girl, cyanotic
since birth. The diagnosis of transposition of the
great vessels was considered clinically, and it was
then confirmed by cineangiocardiogram and cardiac
catheterization. A left-to-right shunt at the atrial
level, a patent ductus arteriosus, and an intact
interventricular septum were found. The left ventricular pressure was 52/0 to 5 mm. Hg, the right
ventricular pressure 62/5 mm. Hg, the aortic pressure 62/38 mm. Hg. The arterial saturation was
10
IDRISS ET AL.
aorta became flabby, and the patient died from
acute blood loss. Postmortem examination showed
the right and left ventricles to be of almost equal
size. The coronary ostia and arteries were patent
to probing. Figure 5 shows a view of the left
ventricle with the outflow tract in the postmortem
specimen. Two probes indicate the coronary ostia.
Comment
In this ease, bleeding appeared to be the canse
of failure.
Discussion
This work demonstrates that switching the
pulnmonary artery and aorta with both
Downloaded from http://circ.ahajournals.org/ by guest on June 15, 2017
Figure 5
Postmortem specimen of case 2 showing a view of
the left ventricle in continuity weith the aortic segment and aorta. The two aortic suture lines ore
seen. Two probes point to the coronary ostia.
67 per cent. Chest roentgelnogm'aiii showed 2-+
cardiac enlargement and increased pulmonary vascularity.
The operation was performed on August 17,
1960, and the procedure as described above was
done. Since this patient was an infant, we thought
that we should interrupt the suture line at several
points in the anastomosis to prevent any interference with the circumferential growth of the major
vessels. At the completion of the second anastomosis, the aortic clamp was removed and the heart
resumed its beats spontaneously. Excessive bleeding from the anastonmosis compelled us to reclamp
the aorta and try to control the bleeding. We also
elected to perform the pulmonary artery anastomosis at this time while we had a dry field. The
aortic clamp was removed and again the heart resumed its beats. Bleeding from the anastomotic
sites continued to be excessive and could not be
controlled. Although Gelfoam soaked in topical
bovine thrombin had previously been very effective in controlling bleeding, it did not have any
clotting effect in this case.
A vicious circle resulted from the bleeding. The
pump had to be transfused rapidly with large
amounts of cold blood. This dropped the temperature of the infant to 29 C. Since we were using
a radiant heating unit against the oxygenator,
warming of the blood was not sufficiently effective.
Circulatory bypass was stopped, and protamine
sulfate was given intravenously but it did not
control the bleeding. The heart rate was slow fromn
the effect of cooling. The left ventricle showed
no sign of dilatation. The bleeding continued, the
cor-
onary arteries is technically feasible and appears to be practical in correcting transposition of the great vessels. The basic features
that make it possible to transfer both coronary arteries with the aorta are the intact
circular aortie segment and the "turning
over" maneuver. The intact aortic segment
acts in reality as a graft lengthening the coronary arteries. This lengthening obviates the
difficulty associated with the individual transfer of the small coronary arteries. Since the
coronary ostia in transposition of the great
vessels are usually on the side of the aorta
adjacent to the pulmonary artery, the only
way to transfer this segment of the aorta containing both coronary arteries is by turning
the segment over to fit the outflow stump of
the left ventricle. This maneuver does not
interfere with coronary circulation. Adequacy
of the coronary circulation was demonstrated
by the uniform presence of good heart action
in the dog experiments and the roentgenogram
of the postoperative injection study of a dog's
heart (fig. 3). The experience with the clinical trials further demonstrates sufficiency of
the coronary circulation as evidenced by good
filling of the coronary arteries, the uniformly
oxygenated myocardium, the strong myoeardial contractions, and the absence of coronarv
insufficiency pattern in the electrocardiogram
during the bypass.
Our experience with the first case demonstrates the importance of patient selection.
This first patient died because the left ventricle was unable to handle the sudden load
imposed on it. A left ventricle adequate to
cope with a work load comparable to that preCirculation. Volume XXIV, July 1961
CORRECTION OF TRANSPOSITION
Downloaded from http://circ.ahajournals.org/ by guest on June 15, 2017
sented by the systemic circuit seems to be a
prerequisite to the success of this operation.
Young infants with equally developed right
and left ventricles and older children with
assoeiated ventricular septal defects and high
left ventricular pressure would probably be
the best candidates for the arterial switching
procedures.
A high percentage of children with transposition of the great vessels succumb in the
first few months of life. Many of them are
without associated ventricular septal defects.
We think that this operation may provide the
best opportunity for salvage of such infants.
The question of repairing associated defects
at the time of arterial correction remains to
be answered. Patent ductus arteriosus must
be closed and preferably divided and sutured
to permit complete mobilization of the pulmonary artery. We do not plan now to close
the ventricular septal defects in subsequent
cases.
Summary
A procedure for complete anatomic correction of transposition of the great vessels is
described. The technic is based on the concept
of isolating an iortic segment containing both
coronary ostia and turning the segment over
to fit the outflow stump of the left ventricle.
Experimental work and two clinical trials are
presented and discussed.
We believe that this operation provides an
anatomic and physiologic correction of the
great vessels and coronary arteries in transposition. In spite of two clinical failures, we
think this operation is worthy of further
11
2.
3.
4.
5.
6.
7.
8.
9.
10.
11.
trials.
Acknowledgment
We wish to thank Dr. W. Riker, Attending
Surgeon; Dr. R. Miller, Chief of Cardiology; and
Dr. M. H. Paul, Head of the Cardiovascular Laboratories, at The Children's Memorial Hospital of
Chicago for their assistance and advice in this work.
References
1. BAFFES, T. G.: A new method for surgical
correction of transposition of the aorta and
Circulation, Volume XXIV, July 1961
12.
13.
14.
pulmonary artery, Suirg., Gynec. & Obst. 102:
227, 1956.
BAILEY, C. P., COOKSON, B. A., DOWNING, D. F.,
AND NEPTU7NE, W. E.: Cardiac surgery under
Ilypotllerl-ial. J. Thoracic Surg. 27: 73, 1954.
BJORK, V. 0., AND BOUCKAERT, L.: Complete
tirailsl)ositioii of the aorta and the pulmonary
artery. All experimental study of the surgical
possibilities for its treatment. J. Thoracic
Surg. 28: 632, 1954.
BLALOCK, A., AND HANLON, C. R.: The surgical
treatment of complete transposition of the
aorta and the pulmonary artery. Surg., Gynec.
& Obst. 90: 1, 1950.
CREECH, 0., JR., MAHAFFEY, D. E., SAYEGH,
S. F., AND SAILORS, E. L.: Complete transposition of the great vessels. Surgery 43: 349,
1958.
GLENN, W. W. L.: Circulatory bypass of the
right side of the heart. IV. Shunt between
superior vena cava and distal pulmonary
artery. Report of clinical application. New
England J. Med. 259: 117, 1958.
GLOTZER, P., BLOOMBERG, A. E., AND HuRwITT,
E. S.: An experimental procedure for correction of transposition of the great vessels.
A.M.A. Arch. Surg. 80: 12, 1960.
KAY, E. B., AND CROSS, F. S.: Surgical treatmienit of transposition of the great vessels.
Surgery 38: 712, 1955.
MERENDINO, K. A., JESSEPH, J. E., HERnON,
P. W., THOMAS, G. I., AND VETTO, R R.:
Interatrial venous transposition. Surgery 42:
898, 1957.
MURPHY, T. 0., GOTT, V., LILLEHEI, C. W., AND
VARCo, R. L.: The results of surgical palliation in 32 patients with transposition of the
great vessels. Surg., Gynec. & Obst. 101: 541,
1955.
MUSTARD, W. T., CHUTE, A. L., KEITH, J. D.,
SIREK, A., ROWE, R. D., AND VLAD, P.: A
surgical approach to transposition of the
great vessels with extracorporeal circuit. Surgery 36: 39, 1954.
SENNING, A.: Surgical correction of transposition
of the great vessels, Surgery 45: 966, 1959.
SCHLICHTER, J. G.: Vascularization of the aorta
in different species in health and disease. Am.
Heart J. 35: 850, 1948.
SCHLICHTER, J., AND HARRIS, R.: The vascularization of the aorta. II. A comparative study
of the aortic vascularization of several species
in health and disease. Am. J. Med. Sc. 218:
610, 1949.
A New Technic for Complete Correction of Transposition of the Great Vessels:
An Experimental Study with a Preliminary Clinical Report
F. S. IDRISS, I. R. GOLDSTEIN, L. GRANA, D. FRENCH and W. J. POTTS
Downloaded from http://circ.ahajournals.org/ by guest on June 15, 2017
Circulation. 1961;24:5-11
doi: 10.1161/01.CIR.24.1.5
Circulation is published by the American Heart Association, 7272 Greenville Avenue, Dallas, TX
75231
Copyright © 1961 American Heart Association, Inc. All rights reserved.
Print ISSN: 0009-7322. Online ISSN: 1524-4539
The online version of this article, along with updated information and services, is
located on the World Wide Web at:
http://circ.ahajournals.org/content/24/1/5
Permissions: Requests for permissions to reproduce figures, tables, or portions of articles
originally published in Circulation can be obtained via RightsLink, a service of the Copyright
Clearance Center, not the Editorial Office. Once the online version of the published article for
which permission is being requested is located, click Request Permissions in the middle column
of the Web page under Services. Further information about this process is available in the
Permissions and Rights Question and Answer document.
Reprints: Information about reprints can be found online at:
http://www.lww.com/reprints
Subscriptions: Information about subscribing to Circulation is online at:
http://circ.ahajournals.org//subscriptions/