* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download INTODUCTION TO THE TRANSITION ELEMENTS
Survey
Document related concepts
Transcript
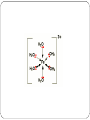
INTRODUCTION TO THE TRANSITION ELEMENTS General physical properties Formation of complexes Colour of complexes What are transition elements? Transition elements are elements in the d-block of the Periodic Table. ** Transition elements (d-block elements) can form one or more stable ions with incompletely filled d-orbitals. Scandium and zinc don't count as transition metals. Sc3+ : Zn2+ : Sc3+ ion has no d electrons Zn has full 3d subshell They have very similar physical and chemical properties. Metals with high melting and boiling point. High densities. Have variable oxidation numbers. Form coloured ions in water. Form a large number of complexes and complex ions. Can use orbitals from 2 different shells for bonding (e.g. 4s & 3d). Have catalytic activity. Electronic Configuration Electronic configuration of first row transition elements. #Briggs 308 Half –filled 3d subshell, more stable. Completely filled 3d subshell, more stable. Zn not considered as transition element. Transition element forms at least one ion with an incomplete subshell. They can use orbitals from 2 different shells for bonding. E.g Fe3+. Physical Properties Transition elements have very similar physical properties due to relatively small difference in effective nuclear charge. The atomic (metallic) radius, ionic radius & first ionisation energy are relatively constant. Melting Points Transition elements have higher melting and boiling points compared to non-transition metals. Due to strong metallic bonds. Transition elements have a lot of electrons (from 3d and 4s subshells) which can be used for metallic bonding. Atomic Radii The atomic and ionic radii of elements V to Cu remains almost constant compared to the elements in the third Period (Na to Cl). From Na to Cl, electrons are added to the outer shell (4s subshell) nuclear charge increase electrons are pulled closer towards the nucleus atomic radii decrease across the period. But from Sc to Zn, electrons are added to inner shell (3d subshell). The inner electrons are able to shield the outer electrons form the nucleus. Hence cancelling the increasing nuclear charge across the period. The atomic radii remains almost constant from V to Cu. First ionisation energy There is little change in the first ionisation energy of transition elements from Ti to Cu. This is because : a) the atomic radii remains almost constant. b) the attractive forces on the outer electrons remains almost constant. Comparison with s-block elements. Variable oxidation states Transition elements have variable oxidation states in their compounds. E.g Fe(II) and Fe(III) compounds. The variable oxidation states is due to the small energy diffence between the 4s and 3d subshells. Fe has oxidation states +1, +2, +3, +4, +5 and +6. Only +2 and +3 are common. Compare the ionisation energies of Fe and Mg. Element 1st 2nd 3rd 4th Mg 736 1450 7740 10500 Fe 762 1560 2960 5400 When compounds are formed, energy is needed to remove electrons from the atoms. The energy for this ionisation is supplied by energy given out when the new bonds are formed. Mg : Mg2+ ions are easily formed when Mg forms compounds. E.g : when Mg2+ ions and O2- ions form electrovalent bonds, energy liberated is enough to supply the ionisation energy to form Mg2+. But the energy liberated is not high enough (7740 kJ) to remove the 3rd electron from the inner shell. Hence Mg can only form Mg2+. Fe : Fe2+ ions are easily formed when Fe forms compounds. But there is no big increase in the 3rd ionisation energy (from 3d subshell). Due to small energy difference between 4s and 3d orbitals. Energy released in formation of Fe(III) compound must be enough to remove the 3rd electron to form Fe3+ ion. The same applies to the loss of more electrons to form higher oxidation state compounds of iron though it is rare in the case of iron. Oxidation States in Compounds # Briggs 311 Variable oxidations states arises from similar energies required for removal of 4s and 3d electrons. The maximum oxidation number of an element corresponds to the maximum number of electrons available for bonding. E.g : Max. oxidation state for Cr is VI – all 3d5 and 4s1 electrons used in bonding. all (except scandium) have an M2+ ion. stability of the +2 oxidation state increases across the row marked increase in 3rd I.E. maximum oxidation state increases from Sc to Mn and decreases from Mn to Zn due to decrease in num. of single electrons in the 3d subshell and the 4s electrons. From Mn to Zn, some electrons in the 3d subshell are paired – unreactive and not used in bonding. Ni atom : Cu atom : Fe atom : Fe2+ ion : Fe3+ ion : Use of Transition Element Ions in Redox Reactions. Iron Commonly forms compounds with oxidation states of II and III. Solutions of Fe2+(aq) are readily oxidised to Fe3+ by many oxidising agents (atmospheric oxygen, K2Cr2O7) E.g : Solution of Fe3+(aq) readily reduced by many reducing agents including KI(aq). E.g : Chromium Solutions of chromium(VI), e.g Cr2O72-, are oxidising agents. E.g : Cr2+(aq) ions are readily oxidised to Cr3+(aq) (green) by air. Manganese MnO4- are oxidising agents. E.g : This reaction is used in redox titrations. Formation of Complexes Transition element complex consist of a transition metal atom surrounded by ligand particles (molecules or anions that have lone pairs of electons). The ligands form coordinate bonds with the empty orbitals of the transition metal atom. The number of coordinate bonds formed by ligand with transition metal in a complex coordination number (usually 4 or 6). Transition metal atom is often a positive ion complex ions. 6-co-ordinate complex ion. Central metal ion is forming six bonds (attached to 6 ligands). Ions have an octahedral shape. E.g : [Fe(H2O)6 ]3+ The single electrons in the 3d level are NOT involved in the bonding in any way. Instead, the ion uses 6 orbitals from the 4s, 4p and 4d levels to accept lone pairs from the water molecules. In some complex ions, the single electrons in the 3d subshell pair up to help produce empty orbitals . E .g, [Fe(CN)6]4- ion. Fe2+ ion : Fe2+ ion in [Fe(CN)6]4- : Common ligands in transition metal complex ions : a) Neutral molecules : H2O, NH3 b) Anions : Cl-, F-, CN-, SCN All common transition metal forms complex ions in water [M(H2O)6]2+ Some important transition metal complexes: (refer to next slide) Questions : 1. A complex ion consists of six CN- ions joined by dative bonds to a Mn3+ ion. What is the formula of the complex ion? 2. A complex ion has the formula [Cr(NH3)4(Cl)2]+. What is the oxidation state of chromium ion? Ligand exchange The ligands in a complex can be exchanged with other ligands. E.g.: When excess NH3 is added to [Cu(H2O)6]2+ ions, four of the H2O ligands are replaced with NH3 to make [Cu(NH3)4(H2O)2]2+. The colour of the complex changes from blue to dark blue. Ligand exchange in haemoglobin. The haemoglobin molecule absorbs oxygen from the lungs by forming oxyhaemoglobin. The oxygen molecule displaces the water molecule and forms a dative bond with Fe atom. O2 + Hb HbO2. Ligands such as CO forms dative bond with Fe atom in haemoglobin more readily than O2 molecule (Fe-CO bond is stronger). Hence CO displaces the O2 in HbO2 to form carboxyhaemoglobin (HbCO). HbO2 + CO HbCO + O2 This cuts down the O2 supply to the body. A patient suffering from carbon monoxide poisoning can be treated by being given pure oxygen to breath. Pure oxygen displaces carbon monoxide by a mass action effect. Shapes of d-orbitals Colour of Complexes Ions with partially filled d-orbitals tend to be coloured. Colour of complexes are caused by the splitting of the energy of the 3d orbitals. Isolated transition metal or ion all d orbitals are degenerate (have same energy). When ligand forms co-ordinate bond with transition metal atom repulsion between electrons of ligands and electrons in the 3d orbitals of transition metal ion 3d orbital split into 2 sets of 3d orbitals having slightly different energy. The amount of splitting depends on the : • central ion • ligand The energy difference between the two 3d energy levels is equal to energy of light in the visible region of the spectrum. E = hf =c/f E = hc / Each wavelength of light has a particular energy associated with it. Red light has the lowest energy in the visible region. Violet light has the greatest energy. When light pass through transition metal ions, electrons in the low energy 3d level absorb photons and are promoted into the high energy 3d level. E.g : Cu2+ complex ion. Complementary colours Colours directly opposite each other on the colour wheel are said to be complementary colours. Copper(II) sulphate solution is pale blue (cyan) because it absorbs light in the red region of the spectrum. Cyan is the complementary colour of red. The colour of transition metal complex depends on E, which depends on : a) Nature of ligand : Cl-, F-, OH-, H2O, NH3, CNE increase E.g : When ligand exchange occur, the colour of the complex change. [Cu(H2O)6]2+ + 4NH3 [Cu(NH3)4(H2O)2]2+ Yellow light has a higher energy than red light.You need that higher energy because ammonia causes more splitting of the d orbitals than water does. b) Nature and oxidation state of transition metal ion : oxidation state increase, splitting increase, E increase. c) Coordination number : Octahedral ion splits more than tetrahedral due to more repulsion.