* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chlamydia trachomatis
Anaerobic infection wikipedia , lookup
Orthohantavirus wikipedia , lookup
Neglected tropical diseases wikipedia , lookup
Gastroenteritis wikipedia , lookup
West Nile fever wikipedia , lookup
Yellow fever wikipedia , lookup
Herpes simplex virus wikipedia , lookup
Chagas disease wikipedia , lookup
Hepatitis C wikipedia , lookup
Brucellosis wikipedia , lookup
Dirofilaria immitis wikipedia , lookup
Typhoid fever wikipedia , lookup
Onchocerciasis wikipedia , lookup
Human cytomegalovirus wikipedia , lookup
Yellow fever in Buenos Aires wikipedia , lookup
Trichinosis wikipedia , lookup
Hepatitis B wikipedia , lookup
Neisseria meningitidis wikipedia , lookup
Middle East respiratory syndrome wikipedia , lookup
Sarcocystis wikipedia , lookup
Oesophagostomum wikipedia , lookup
Marburg virus disease wikipedia , lookup
African trypanosomiasis wikipedia , lookup
Neonatal infection wikipedia , lookup
Sexually transmitted infection wikipedia , lookup
Hospital-acquired infection wikipedia , lookup
Schistosomiasis wikipedia , lookup
Lymphocytic choriomeningitis wikipedia , lookup
Leptospirosis wikipedia , lookup
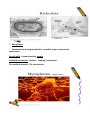
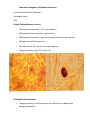
Obligate Intracellular Pathogen Rickettsia Chlamydia D. Rickettsia - Virus-like bacteria Infect cells lining the capillaries (intracellular). Transmitted by insects (arthropods). Defective bacteria leaky plasma membranes, very small 0.3-1.0 micrometers. Ex. Rocky Mountain Spotted Fever, typhus, Q fever. E. Chlamydia - “virus-like” (intracellular) Spread from one human to the next. Very small: 0.2-0.7 micrometers. Defective bacteria - can’t make their own ATP. Complicated reproductive cycle. Diseases: blindness, urethritis, and pneumonia. F. Mycoplasma Sometimes form long strands that resemble fungi in microscopic appearance. No cell walls - instead contains sterols. Ameboid locomotion - disease: “walking” pneumonia The smallest bacteria - 0.2 micrometers Chlamydia & Rickettsia General Characteristics • Obligate intracellular organisms • Can not be cultured on agar plates • Dependant on a host for survival Rickettsia • Intracellular, gram negative organism • Requires host to replicate and survive • Cause febrile illness through the bite of an arthropod • Patient often presents with a rash • Posses a cell wall Family Rickettsiaceae: Genera • Zoonotic infection – Human microbial pathogens ~61% zoonotic – Rickettsia are arthropod-borne infections • Spotted Fever Group – Rickettsia rickettsii – Rocky Mountain spotted fever; rodent, tick • Typhus Group – Rickettsia typhi – Endemic typhus; rodent, flea – Rickettsia prowazekii – Epidemic typhus; mammal, louse Rickettsia: Gram Stain and Culture • Gram (-) small, pleomorphic coccobacilli • Gram stain poorly, observed by Giemsa stain of infected cell • Grow in phagocytic, nonphagocytic cells • Lab culture in embryonated eggs or cell tissue culture (similar for virus) • Cultivation costly and hazardous; aerosol transmission occurs easily Rickettsia: Lab Immuno-diagnosis • Giemsa, or Immunofluorescence assay (IFA) - direct detection MO in tissue • Weil-Felix reaction – Nonspecific test – Rickettsial antibody agglutinate Proteus vulgaris – Presumptive evidence of typhus group infection – Not very sensitive or specific, many false positives • Agglutination or Complement Fixation (CF) assay - use specific Rickettsial antigen, test for infection and antibody Rickettsia: Virulence Factors • Induced phagocytosis, intracellular growth – protected from host immune clearance • Replicates in endothelial cells – cell damage, vasculitis • Recruitment of actin - intracellular spread R. rickettsii: Rocky Mountain Spotted Fever (RMSF) • USA ~500-1000 cases/year • Ticks must remain attach for hours • Incubation 7 days - headache, chills, fever, aching, nausea • Followed by maculopapular rash on extremities (including palms and soles), spread chest, abdomen • If untreated – Petechial rash, hemorrhages skin and mucous membranes – Vascular damage, MO invades blood vessels – Death up to 20%, due to kidney or heart failure Rickettsia: Typhus Group • Incubation 5-18 days • Symptoms - severe headache, chills, fever, maculopapular rash (subcutaneous hemorrhaging as MOs invade blood vessel) • Rash begins on upper trunk; spread to whole body except face, palms of hands, soles of feet • Lasts ~2 weeks • Patient may have prolonged convalescence R. typhi : Endemic Typhus Fever • typhus” “fever” • Disease worldwide in warm, humid areas (Gulf states, So Cal.; S. America, Africa, Asia, Australia, Europe) • Murine typhus - rat primary reservoir, transmitted to human by rat flea • Disease occurs sporadically • Clinically same, but less severe than epidemic typhus • Restricted to chest, abdomen; generally uncomplicated, lasts <3 weeks • Low fatality R. prowazekii : Epidemic Typhus Fever • Disease C & S Americas, Africa; less common USA • Human, squirrel primary reservoir • Transmitted by louse; bites, defecates in wound • At risk - people living in crowded, unsanitary conditions; often war, famine, natural disaster • Complications - myocarditis, CNS dysfunction • Mortality high untreated cases, up to 20% • Brill-Zinsser disease - individual may harbor MO, latent infection with occasional relapses Rickettsia: Treatment and Prevention • RMSF – Doxycycline drug of choice – Avoid ticks, wear protective clothing, use insect repellents, insecticides – In infested areas, check and remove ticks immediately • Typhus Fever – Doxycycline effective – Improve personal hygiene and living conditions, reduce lice by insecticides, control rodent population – Inactivated vaccine for epidemic typhus Laboratory Diagnosis of Rickettsial Disease Immunohistochemical detection Serological tests PCR Family Chlamydiaceae: Genera • Chlamydia trachomatis – STD, eye infection • Chlamydophila pneumoniae – pneumonia • Chlamydophilia psittaci – pneunomia (psittacosis); birds, humans • Obligate intracellular parasite • Cell wall similar G(-) bacilli, lack peptidoglycan • Energy parasites, use ATP of host cell Chlamydia Characteristics • Unique growth cycle because they are deficient in independent energy metabolism • Replication involves elementary body (EB) and reticulate body (RB) – EB’s are infectious and non-metabolically acitve – RB’s are noninfectious and metabolically active Chlamydia: Life Cycle – Elementary Body (EB) • Circular, infectious form; 300-400 nm • Metabolically inactive • Resistant to harsh environments • 0 hour - EB binds to host cell, induced phagocytosis • Outer membrane of EB prevents lysosome fusion, survives in phagosome • 8 hours - EB reorganizes into Reticulate Body (RB) Chlamydia: Life Cycle – Reticulate Body (RB) • Noninfectious form, larger, less dense, 800-1000 nm • Metabolically active • 8-30 hours – Synthesize new materials – Multiply by binary division – Form inclusion body – Reorganize, condense into EB • 35-40 hours - cell lyses, releases EB, begins cycle again Chlamydia: Lab Immuno diagnosis • Stain tissue – Giemsa stain – Direct fluorescent antibody (DFA) – ELISA – Less sensitive • Cell culture – More sensitive method – Grow MO in tissue culture, stain infected cells • DNA amplification test – Recently developed – Specific, sensitive Now routine test of choice Chlamydia: Virulence Factors • Intracellular replication – protected from host immune defense • Prevent fusion of phagolysome – evades phagocytic killing • Repeated infections by C. trachoma result in cell pathology • Serotypes A-K and L1, L2, L3 - serotype identifies strain’s clinical manifestation Chlamydia pneumoniae • Important respiratory pathogen (acute respiratory disease, pneumonia, and pharyngitis) • Common (50% of adults have antibodies) • College age students most susceptible • Implicated in asthma • Risk factor for Guillain-Barre’ syndrome • Important respiratory pathogen (acute respiratory disease, pneumonia, and pharyngitis. • Common (50% of adults have antibodies • College age students most susceptible • Implicated in asthmaRisk factor for Guillain-Barre’ syndrome • Reinfection common • Biphasic clinical picture - Prolonged sore throat and hoarseness, followed by flu-like lower respiratory symptoms - Pneumonia and bronchitis Chlamydia trachomatis • Most commonly sexually transmitted bacterial pathogen in U.S. – Only HPV is a more commonly sexually transmitted disease – Major cause of sterility in U.S. – May be transmitted to newborns during delivery • Results in conjunctivitis • Other sites of infection - Trachoma – infection of the conjunctiva, resulting in scarring and blindness (Mostly in India and Egypt) - Lymph granuloma venereum • Infects lymph nodes • STD found in immigrants from the tropics Chlamydia psittaci • Causes psittacosis (parrot fever) • Identification based on history of close contact with birds and serologic evaluation • parrot” “parrot fever” • Naturally infects avian species • Mild to severe respiratory infections • Human infection by contact infected bird • Infection - subclinical to fatal pneumonia • Commonly causes atypical pneumonia with fever, chills, dry cough, headache, sore throat, nausea, and vomiting Chlamydia trachomatis: Trachoma • “rough” “trachoma” granulations on conjunctiva • Serotypes A-C • Single, greatest cause blindness developing countries • Infections mainly children (reservoir), infected first three months life • Transmission eye-to-eye, direct contact (droplet, hand, clothing, fly) • Chronic infection, reinfection common • Conjunctival scarring, corneal vascularization • Scars contract, upper lid turn in so eyelashes cause corneal abrasions • Leads to secondary bacterial infections, blindness Trachomatis: Lymphogranuloma Venereum • Venereal disease, occurs developing, tropical areas • Primary stage - painless lesion (vesicle or an ulcer) occurs site of entry in few days, heals with no scarring; but widespread dissemination • Secondary stage - occurs 2-6 weeks later, symptoms of regional suppurative lymphadenopathy (buboes), may drain for long time, accompanied by fever and chills. Arthritis, conjunctival, CNS symptoms • Tertiary stage - urethrogenital perineal syndrome; structural changes, such as non-destructive elephantiasis of the genitals, rectal stenosis C. trachomatis: STD • Urogenital tract infection - serotypes D-K • Major cause of nongonococcal urethritis; frequently found concomitantly with N. gonorrhoeae • In males - urethritis, dysuria, sometimes progresses to epididymitis • In females - mucopurulent cervical inflammation, can progress to salpingitis and PID • USA - #1 STD C. trachomatis: Inclusion Conjunctivitis • Newborns and adults • Genital tract infection source of eye infection (serotypes D-K) • Benign, self-limited conjunctivitis, heals with no scarring • Newborns infected during birth process: – 1-2 weeks, mucopurulent discharge – Lasts 2 weeks, subsides – Some develop afebrile, chronic pneumonia • In adults – causes an acute follicular conjunctivitis with little discharge Chlamydia: Treatment and Prevention • Genital tract infection and conjunctivitis: – Adult - azithromycin or doxycycline, prompt treatment of patients and partners – Newborn – erythromycin – Public Health education • Trachoma: – Need prompt treatment, prevent reinfection – Systemic tetracycline, erythromycin; long term therapy necessary – Improve living, sanitary conditions – Difficult to prevent endemic disease in developing countries due to lack of resources, medical care Laboratory Diagnosis If cultured, must be in cells Direct microscopic examination to find EB’s visualized with fluorescein-conjugated antibodies Enzyme immunoassay Nucleic acid probes with and without amplification (PCR) Serologic tests are method of choic