* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Unit 3
Chemical biology wikipedia , lookup
Biochemical cascade wikipedia , lookup
Cell culture wikipedia , lookup
Biochemistry wikipedia , lookup
Symbiogenesis wikipedia , lookup
Western blot wikipedia , lookup
Artificial cell wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Developmental biology wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Signal transduction wikipedia , lookup
Cell-penetrating peptide wikipedia , lookup
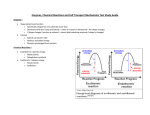
YOUR NOTES UNIT 3 NOTES Cell Structure CELLS: BUILDING BLOCK OF LIFE ORGANELLES Organelle Plasma or Cell Membrane • Structure Composed of a bi-layer of phospholipids with proteins embedded in it • • Function Holds cell together and gives shape Regulates the movement of substances in and out of the cell Nucleus • • Dark granule in centre of cell Surrounded by a double membrane called the nuclear envelope/membrane • • • • Controls cell activities through protein synthesis Contains genetic info Directs cell division Site of DNA replication and transcription Nucleolus • • • • Small, dark spot in nucleus Made up of RNA No membrane A double membrane made of phospholipids which has nuclear pores This is the cytoplasm of the nucleus • Makes rRNA, which then makes ribosomes • • Separates nuclear material from cytoplasm Pores allow RNA and proteins in & out of nucleus • • Double membrane of phospholipids Inner membrane is very folded – CRISTAE (increased surface area) • “powerhouse” – makes energy (ATP) for the cell in a process called CELLULAR RESPIRATION • • Fluid in between MATRIX Has its own DNA Endoplasmic Reticulum (ER) 2 types: • Membrane channels running from the nuclear envelope throughout cytoplasm. It is a transport system. Rough ER rER • • Nuclear membrane/ envelope Nucleoplasm Mitochondria Smooth ER sER • • C6H12O6 + 6O2 → 6CO2 + 6H2O + ATP • Cristae (inner folds) provides more surface area for enzyme reactions to make ATP Has ribosomes attached • Usually connected with the nuclear membrane No ribosomes attached • Ribosomes make proteins and then place them in the rER The rER packages proteins in a vesicle and sends them to the Golgi Body Makes lipids and steroids Also, detoxifies harmful material or waste products You’ll find a lot of sER in liver cells and glands that • • • make hormones Ribosome Polysome aka polyribosome Golgi apparatus / body / complex • • • • Small, dense granules made of rRNA & protein No membrane 2 subunits (large & small) • Free floating group of ribosomes • Makes large proteins (faster) with a single mRNA molecule • • Stacks of flattened sacs Surrounded by vesicles • • Collects, sorts, packages and distributes materials Modifies proteins and lipids from ER • • Site of protein synthesis (translation) Usually attached to rER so proteins can be easily exported Can be free in cytoplasm – these proteins not exported Organelle inter-relationship • • • • • DNA copies a gene as RNA RNA moves through pore and attaches to ribosome to make protein Protein put into rER, then sent to Golgi in a vesicle Golgi modifies protein, stores it until needed, and sends it to plasma membrane in a vesicle. Protein released at the Plasma Membrane via exocytosis Vesicles • Small, membranous (bilayer) bound sac usually made by Golgi body Vacuole • • A large vesicle typically one large one in plants (but small in animals) membranous sacs double membraned vacuoles with hydrolytic (digestive) enzymes Lysosome • • Cytoskeleton • Made up of: Microfilaments, Microtubules, intermediate filaments 1.Microfilaments • Storage sac (temporary storage) ex. H20, food, digestive enzymes, hormones 2 types: • Transport vesicle: moves substances from ER to Golgi • Secretory vesicle: moves substances from Golgi to cell membrane • long term storage of H20 with dissolved sugars and salts • • • • • hydrolysis! Destroys harmful substances Can kill the cell if it breaks open- “suicide sacs” Many in white blood cells Series of protein fibres in the cytoplasm • • • Maintains cell shape Monorail to transport organelles around the cell Assemble and disassemble as needed Long & thin protein fibres • Gives structure and support to cell membrane aka actin filaments 2. Microtubules 3. Intermediate filaments • Moves organelles around cell Anchor for organelles and monorail for organelle movement Used to make cilia, flagellum & centrioles Intermediate size between actin filaments and microtubules Made of keratin • Keeps the nucleus in place “watery gel” between cell membrane and nuclear envelope Contains water with dissolved salts, proteins & other organic compounds • • • Support & suspend organelles Provide water Allows diffusion to occur in cells • • Double membrane of phospholipids Inside stacks of discs called GRANUM • photosynthesis (light energy converted into chemical energy) • ATP + 6CO2 + 6H2O → C6H12O6 + 6O2 • Contain: their own DNA, ribosomes, & enzymes • Granum= contains chlorophyll which traps solar energy • Maintains cell shape and skeletal support • • • • Cytoplasm • • In addition plants have: Chloroplasts Cell wall • • Largest fibre Cylinder shaped & made of tubulin (protein) Tough, rigid outermost wall Made up cellulose (very strong) • • Cell Membrane and Transport The cell membrane is the outer living boundary of the cell. Fxns of the cell membrane: • Separates cell from outside environment • Provides structure (strength & shape) & protection • Regulates substances entering and leaving cell (gatekeeper) Fluid Mosaic Model =a mosaic of protein molecules bobbing in a fluid bilayer of phospholipids Membrane Structure – phospholipid bilayer in water Cell (Plasma) Membrane PROTEINS: determine membrane function (glycoprotein); transmit chemical signals into the cell (channel or receptor proteins); act as carriers for specified substances CARBOHYDRATES: Attached to outside & allows cell to recognize as self (e.g. ABO blood typing & possible immune system rejection GLYCOLIPIDS : phospholipids with covalently bonded carbohydrate chains GLYCOPROTEINS: proteins with covalently bonded protein chains CHOLESTEROL: Reduces membrane fluidity by reducing phospholipid movement. Also stops the membrane from becoming solid at room temperatures. The Cell (plasma) Membrane is SELECTIVELY PERMEABLE: Controls what comes in and out of the cell. Does not let large, charged or polar things through without help. The phospholipids and proteins move, thus allowing small non-polar molecules to slip through. Transport Across the Membrane: Everything that is transported across the cell membrane takes place by one of two fundamental processes: 1. Passive transport moves molecules from a [high] to [low] in order to establish equilibrium. Does not require ATP (energy) 2. Active transport moves molecules from [low] to [high] AGAINST the concentration gradient and this process requires ATP. 3 TYPES OF PASSIVE TRANSPORT 1. Diffusion 2. Osmosis 3. Facilitated Transport / facilitated diffusion 1. Diffusion - passive process (no energy required). - The molecules move from an area of [high] to [low] until they are evenly distributed (equilibrium) - EXAMPLES of molecules: oxygen, carbon dioxide, alcohols, fatty acids, glycerol, and urea. The rate of diffusion will be affected by: 1. Concentration: The greater the difference in concentration, the faster the diffusion. 2. Molecular size: smaller substances diffuse more quickly. 3. Temperature of the Medium: the lower the temperature, the more slowly molecules can move through it. 4. Solubility: lipid - soluble molecules will dissolve through the phospholipid bilayer easily, as will gases like CO2 and O 2. 5. charge: charged molecules do not pass through the membrane easily 2. Osmosis - Osmosis is the diffusion of water across a selectively permeable membrane driven by a difference in the concentration of solutes across the membrane. - So it is the WATER THAT MOVES to create equilibrium!!! - Just remember that Solute Sucks! Tonicity of a Solution The tonicity is the solute concentration of a solution & it will affect the size & shape of cells: ISOTONIC SOLUTION: • the solution concentration is equal on both sides of the membrane . • Water moves back and forth, but there is no net gain or loss of water. HYPERTONIC SOLUTION: • solution outside the cell is more concentrated than inside. • water will move out of the cell. • causes the cell to shrink • *Memory Trick... Hyper people get skinny! HYPOTONIC SOLUTION: • concentration inside the cell is more concentrated than outside. • water will move into the cell. • cause the cell to swell. • *Memory Trick... Hippos (hypos) are FAT! In Biology we usually talk about the SOLUTION’S tonicity, NOT the cells! Water flows from a hypotonic solution to a hypertonic solution • ie: tap water hypertonic to distilled water but hypotonic to sea water In the diagram, Which side is hyper/hypo? Which way will water move? Terminology associated with result of tonicity • Crenation – RBC shrinks and shrivels up • Hemolysis – RBC swells and may burst • Plasmolysis – plant cell membrane shrinks and pulls away from the cell wall • Turgor pressure – plant cell membrane swells and organelles are crushed against the cell wall – When we speak of plants, we speak of TURGOR PRESSURE. • • plasmolysis and crenation of cells happens if a cell is put in a hypertonic solution hemolysis and turgor pressure happen if a cell is put in a hypotonic solution 3. Facilitated Transport (aka Facilitated Diffusion) • • • • Some molecules are not normally able to pass through the lipid membrane, they need channel or carrier proteins to help them move across. does not require energy If molecules are POLAR, CHARGED, or TOO LARGE they need a protein the help them across the membrane EXAMPLES: sugars, amino acids, ions, nucleotides ACTIVE TRANSPORT • • • Active Transport: the movement of polar, large, and charged molecules moving against the [ ] gradient (uphill). Requires ATP (energy) EXAMPLES: ions (like Na+ and K+ in cells, and iodine) and sugars, amino acids, nucleotides... 3 TYPES OF ACTIVE TRANSPORT: ALL REQUIRE ENERGY – FUEL!!!! 1. sodium-potassium (Na/K) pump 2. endocytosis i. Phagocytosis ii. Pinocytosis 3. exocytosis 1. Sodium potassium (Na/ K) pump (mostly in nerve membranes). + • Na attaches to protein carrier, moves across membranes and released outside + • “pump” (protein) changes shapes so Na can’t re-enter + • K attaches, moves across & release inside + • Pump changes back to Na accepting shape 2. Endocytosis: (“Endo” means “in”). • the taking in of molecules or particles by invagination of the cell membrane forming a vesicle. • requires energy. • There are two types of endocytosis: i. Pinocytosis (cell drinking): small molecules are ingested and vesicle is formed ii. Phagocytosis (cell eating): ): large particles are invaginated into the cell (ie: white blood cells ‘eat’ bacteria). 3. Exocytosis: (“Exo” means “out”.) • reverse of endocytosis. • a cell releases the contents of a vesicle out of the cell. • requires energy. • Example: vesicles from the Golgi fuse with the plasma membrane & the proteins are released outside of the cell • A balance needs to exist between endocytosis & exocytosis. Why? CELL SIZE • • Cells size is a very important factor for cell survival. Cells are the size they are because that is the size they have to be in order to function effectively. 4 factors regulate cell size: 1. Surface Area (SA) to Volume Ratio: • As a cell grows, it suffers the possibility of the volume increasing faster than the surface area. • If the cell gets too big, there will not be enough room on the plasma membrane for things to get in and out quickly. • Why? Its surface area has not kept up to its volume size. • When the volume of a cell increases, the amount of surface area does not increase in the same proportion. • To accommodate, cells can get thinner, become folded or elongated, become irregular or convoluted ie. Intestinal villi, mitochondria, surface of brain • This increases SA without changing the volume – brings cell contents closer to membrane for more efficient exchange • As a cell grows, its’ SA/Volume ratio decreases because SA increases but so does the volume • You want a large surface area to small volume to maximize exchange of nutrients and wastes. 2. Metabolic Rate: Cells have high metabolic rate • produces heat energy so must remain small enough to release heat 3. Nuclear Control: if cell gets too big, nucleus loses control over its activities and reproduction • e.g. large, one celled organisms (Paramecium & Amoeba) have 2 nuclei, each with different jobs 4. Support: a large cell would collapse under its own weight Rule: smaller is better or having a “funny shape” is better ENZYMES Enzymes are catalysts (a substance that speeds up a reaction without being consumed). Enzymes are proteins and are reusable. They work in low concentrations and speed up the reaction rate. amylase Starch → Glucose lipase Lipids → Fatty Acids and Glycerol protease Proteins → Amino Acids • • • • • • Enzymes allow reactions to proceed at lower temperatures than they would normally occur. The reactant(s) acted on is known as the substrate(s). Enzymes work by forming a very temporary complex called the ENZYME SUBSTRATE COMPLEX. Enzyme Structures • Enzymes have grooves (or pockets) • These areas are called active sites and this is where the substrate attaches E + S → ES → E + P Lock and Key Model • Specific groove shapes in an active site means that enzymes can only bond with one specific substrate (reactant) Induced Fit Model • When substrate and active site shapes don’t precisely match, the shape of the enzyme changes, which makes it more reactive Coenzymes • Many enzymes are made up of 2 pieces: a protein portion (inactive) & CO-ENZYME, a non-protein portion • When co-enzyme binds to active site, the enzyme becomes active and the substrate will now ‘fit’ into the active site. • co-enzymes tend to be vitamins (vit. B, riboflavin, niacin, etc). Metabolism • Definition: Metabolism is the sum of all the chemical reactions that occur in a cell. • These chemical reactions occur in organized sequences from reactants to end products with the help of enzymes. • This organized sequence of reactions is known as a metabolic pathway. Enzyme Action Enzymes operate by lowering the energy of activation (EA) needed for a reaction to occur. EA = the initial investment of energy to cause a reaction Factors Affecting Reaction Rates: Lowering EA 1. Concentration: more enzyme and/or substrate means more collisions. a. The reaction speeds up as the [substrates] increases, and it levels out when the enzymes are working at the maximum speed (saturation). What can you do to cause an increase in reaction rate? = Add enzymes! b. The reaction speeds up as you increase the [enzyme], and slows down as the substrate has all been turned into product. What can you do to cause an increase in reaction rate? -‐ Add substrate! 2. Temperature: the reaction rate will increase as the molecules move faster, increasing collisions • At a certain point, the rate of these collisions will be at the fastest rate. This is the OPTIMUM TEMPERATURE. • above the optimum temperature, the enzyme becomes denatured (changes shape) and no longer functions properly. o • Most of our enzymes have an optimal temperature of 37 C (body temperature). • temperature changes affect enzyme shape & hence, its activity. › Below 40°C change can be reversed › Above 40°C change is permanent • in cold, molecules move slowly so few collisions › In cold temp., it gets hard to tie shoes because enzymes operating slowly – fingers can’t work 3. pH:. All enzymes have an optimal pH. Extremes of pH can alter enzyme shape & therefore, affect enzyme • • • • activity. Saliva pH 7 Stomach pH 2.5 Intestines pH 8.5 Vagina pH 2.5 4. Inhibitors: Chemicals that interfere with the enzyme action. • There are two types of INHIBITORS: a) Competitive Inhibitors resemble normal substrate molecule & competes for active site • Reduces productivity of enzyme by blocking substrate from entering the active site • In humans, hydrogen cyanide (poison contained within marijuana and tobacco smoke) inhibits cytochrome oxidase – an important enzyme for oxygen use by cells – lethal affect in potent doses b) Non-Competitive Inhibitors bind to enzyme (not at active site) and this denatures the enzyme. An example is heavy metals, such as lead and DDT in the nervous system. • • • Another example of non-competitive inhibition is when a metabolic product can feedback on a metabolic pathway to control how much product is made. The final product can temporarily attaches to the first enzyme (not in the active site) The enzyme will be denatured and the reaction will stop. A Metabolic Pathway • • o Product B is substrate for 2nd reaction whose product is substrate for 3rd reaction……. Until final product is reached (G) B to F are known as intermediate product This is an example of NEGATIVE FEEDBACK or FEEDBACK INHIBITION. When the concentration of the final product gets low again, there will be less inhibition on the enzymes and the metabolic pathway is reactivated. • feedback inhibition prevents cell from wasting chemical resources to synthesize more than is necessary Feedback Inhibition & Control of Metabolic Rate • • Thyroxin, the hormone that controls the metabolic rate of all cells in your body, is produced by the thyroid gland in the neck. If the [thyroxin] in your body is high, your metabolic rate will be raised, and if thyroxin levels are low, your metabolic rate will be low.