* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Carbohydrate PPT Notes
Survey
Document related concepts
Transcript
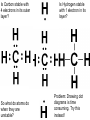
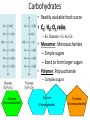
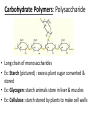
Organic Molecules • Carbon = building block of organic molecules • Carbon is unique – 2nd electron level not full – Only has 4 electrons in 2nd level – Will bond up to four times • Monomer: Small carbon molecules – Ex: Amino acid • Polymer: chain of linked monomers – Ex: Protein Amino acid Amino acid Amino acid Amino acid Amino acid Amino acid Amino acid Amino acid Amino acid Amino acid Polymer (protein) Is Carbon stable with 4 electrons in its outer layer? So what do atoms do when they are unstable? Is Hydrogen stable with 1 electron in its layer? Problem: Drawing dot diagrams is time consuming. Try this instead! Carbon creates 4 bonds to be stable Carbohydrates • Readily available food source • C1: H2: O1 ratio – Ex: Glucose = C6 H12 O6 • Monomer: Monosaccharides – Simple sugars – Bond to form larger sugars • Polymer: Polysaccharide – Complex sugars Glucose (monosaccharide) Sucrose (Polysaccharide) Fructose (monosaccharide) Carbohydrate Polymers: Polysaccharide • Long chain of monosaccharides • Ex: Starch (pictured) : excess plant sugar converted & stored • Ex: Glycogen: starch animals store in liver & muscles • Ex: Cellulose: starch stored by plants to make cell walls REview 1) How many electrons does Carbon have in the 1st energy level? 2nd energy level? 2) Which type of organic molecule is most commonly used as energy for cells? 3) If a carbohydrate has 8 carbon atoms, how many oxygen and hydrogen atoms will it most likely contain? 4) What are the monomers of carbohydrates called? 5) What are the polymers of carbohydrates called? 6) Which polymer of carbohydrates is stored by animals? 7) Which polymer of carbohydrates is stored by plants?