* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Inflamation Historical Review
Cell culture wikipedia , lookup
Cellular differentiation wikipedia , lookup
Cell encapsulation wikipedia , lookup
Signal transduction wikipedia , lookup
Extracellular matrix wikipedia , lookup
List of types of proteins wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Celsus,
• listed the four cardinal signs of
inflammation: rubor, tumor, calor, and
dolor (redness, swelling, heat, and pain).
• A fifth clinical sign, loss of function (functio
laesa), was later added by Virchow.
Scottish surgeon John Hunter
• inflammation is not a disease but a nonspecific
response
• Julius Cohnheim (1839-1884) first used the
microscope to observe inflamed blood vessels in
thin, transparent membranes, such as in the
mesentery and tongue of the frog.
• Noted the initial changes in blood flow, the
subsequent edema caused by increased
vascular permeability, and the characteristic
leukocyte emigration,
the Russian biologist Elie
Metchnikoff 1880s,
• discovered the process of phagocytosis by
observing eating bacteria by mammalian
leukocytes.
• concluded the purpose of inflammation
was to bring phagocytic cells to the injured
area to engulf invading bacteria.
• both cells (phagocytes) and serum factors
(antibodies) were critical for defense
against microorganisms,
Sir Thomas Lewis,
• that chemical substances, such as
histamine locally induced by injury,
mediate the vascular changes of
inflammation.
Definition
• inflammatory process is the reaction of blood
vessels, leading to the accumulation of fluid and
leukocytes in extravascular tissues.
• Inflammation is a complex reaction to injurious
agents such as microbes and damaged, usually
necrotic, cells that consists of vascular
responses, migration and activation of
leukocytes, and systemic reactions.
Definition
• a host response of vascularized tissues to
exogenous and endogenous stimuli is
called inflammation.
The inflammatory response is
closely intertwined with the process
of repair.
• Inflammation serves to destroy, dilute, or
wall off the injurious agent, and it sets into
motion a series of events that try to heal
and reconstitute the damaged tissue.
Repair
• Repair begins during the early phases of
inflammation but reaches completion usually
after the injurious influence has been
neutralized.
• During repair, the injured tissue is replaced
• regeneration of native parenchymal cells,
• by filling of the defect with fibrous tissue
(scarring) or,
• by a combination of these two processes.
Inflammation
• a protective response, the ultimate goal of which is to rid
the organism of both the initial cause of cell injury (e.g.,
microbes, toxins) and the consequences of such injury
(e.g., necrotic cells and tissues).
• Inflammation and repair may be potentially harmful,
• Inflammatory reactions, for example, underlie common
chronic diseases, such as rheumatoid arthritis,
atherosclerosis, and lung fibrosis,
• . Repair by fibrosis may lead to disfiguring scars or
fibrous bands that cause intestinal obstruction or limit the
mobility of joints.
•
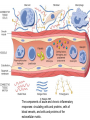
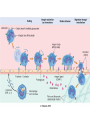
The components of acute and chronic inflammatory
responses: circulating cells and proteins, cells of
blood vessels, and cells and proteins of the
extracellular matrix.
Compnents
• The inflammatory response consists of two
main components,
• a vascular reaction and a cellular
reaction.
Components
• The circulating cells include neutrophils,
monocytes, eosinophils, lymphocytes,
basophils, and platelets.
Components
• The connective tissue cells
• mast cells, which intimately surround blood vessels;
•
•
•
•
•
•
fibroblasts;
resident macrophages;
lymphocytes.
The extracellular matrix,
structural fibrous proteins (collagen, elastin),
adhesive glycoproteins (fibronectin, laminin, nonfibrillar collagen,
tenascin, and others),
• proteoglycans.
• The basement membrane is a specialized component of the
extracellular matrix consisting of adhesive glycoproteins and
proteoglycans.
Patterns
• Acute inflammation is rapid in onset
(seconds or minutes) and is of relatively
short duration, lasting for minutes, several
hours, or a few days;
• characteristics are the
– exudation of fluid and plasma proteins
(edema)
– emigration of leukocytes, predominantly
neutrophils.
Patterns
• Chronic inflammation is of longer duration
Chronic inflammation is associated with
• the presence of lymphocytes and
macrophages,
• the proliferation of blood vessels,
• fibrosis,
• tissue necrosis.
PLEASE NOTE
• The vascular and cellular reactions of both
acute and chronic inflammation are
mediated by chemical factors
Chemical mediators
• chemical mediators are derived from
– plasma proteins or cells
• are produced in response to or activated by the
inflammatory stimulus.
• mediators, acting singly, in combinations, or in
sequence,
• amplify the inflammatory response and influence its
evolution.
• Necrotic cells or tissues themselves-whatever the cause
of cell death-can also trigger the elaboration of
inflammatory mediators.
– acute inflammation after myocardial infarction
Inflammation is terminated
•
•
•
•
offending agent is eliminated
the secreted mediators are broken down
dissipated.
there are active anti-inflammatory
mechanisms
• control the response
– prevent it from causing excessive damage to
the host.
Acute inflammation has three major
components:
• . (1) alterations in vascular caliber that lead to an
increase in blood flow;
• (2) structural changes in the microvasculature
that permit plasma proteins and leukocytes to
leave the circulation
• (3) eukocytes
– emigration of the leukocytes from the microcirculation
– Accumulation of the leukocytes in the focus of injury,
– activation of the leukocytes to eliminate the offending
agent
exudation
• The escape of fluid, proteins, and blood
cells from the vascular system into the
interstitial tissue or body cavities is known
as exudation.
exudate
• An exudate is an inflammatory
extravascular fluid that has a high protein
concentration, cellular debris, and a
specific gravity above 1.020.
• It implies significant alteration in the
normal permeability of small blood vessels
in the area of injury.
transudate
• a transudate is a fluid with low protein
content (most of which is albumin) and a
specific gravity of less than 1.012.
• It is essentially an ultrafiltrate of blood
plasma that results from osmotic or
hydrostatic imbalance across the vessel
wall without an increase in vascular
permeability.
• Edema denotes an excess of fluid in the
interstitial or serous cavities; it can be
either an exudate or a transudate.
Pus
• Pus, a purulent exudate, is an
inflammatory exudate rich in leukocytes
(mostly neutrophils), the debris of dead
cells and, in many cases, microbes.
STIMULI FOR ACUTE
INFLAMMATION
• Infections (bacterial, viral, parasitic) and
microbial toxins
• Trauma (blunt and penetrating)
• Physical and chemical agents (thermal injury,
e.g., burns or frostbite; irradiation; some
environmental chemicals)
• Tissue necrosis (from any cause)
• Foreign bodies (splinters, dirt, sutures)
• Immune reactions (also called hypersensitivity
reactions)
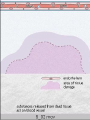
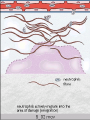
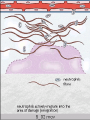
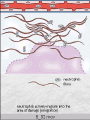
The major local
manifestations of
acute inflammation,
compared to normal.
(1) Vascular dilation
and increased blood
flow (causing
erythema and
warmth), (2)
extravasation and
deposition of plasma
fluid and proteins
(edema), and (3)
leukocyte emigration
and accumulation in
the site of injury.
VASCULAR CHANGES
• Changes in Vascular Flow and Caliber
– vasodilatation
• increased permeability of the
microvasculature
• Effects on the endothelial cells
Changes in Vascular Flow and
Caliber
• Vasodilation
– Vasodilation first involves the arterioles and
then results in opening of new capillary beds
in the area.
– Thus comes about increased blood flow,
which is the cause of the heat and the
redness
increased permeability of the
microvasculature,
• outpouring of protein-rich fluid into the extravascular
tissues;
• The loss of fluid results in concentration of red cells in
small vessels and increased viscosity of the blood,
• dilated small vessels packed with red cells and slower
blood flow, a condition termed stasis.
• stasis, leukocytes, principally neutrophils, accumulate
along the vascular endothelium.
• Leukocytes then stick to the endothelium, and soon
afterward they migrate through the vascular wall into the
interstitial tissue,
increased permeability of the
microvasculature,
• The loss of protein from the plasma reduces the
intravascular osmotic pressure
• The loss of protein from the plasma increases
the osmotic pressure of the interstitial fluid.
• increased blood flow through the dilated vessels
increases hydrostatic pressure
• a marked outflow of fluid and its accumulation in
the interstitial tissue
• The net increase of extravascular fluid results in
edema.
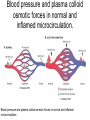
Blood pressure and plasma colloid
osmotic forces in normal and
inflamed microcirculation.
Blood pressure and plasma colloid osmotic forces in normal and inflamed
microcirculation.
• A, Normal hydrostatic pressure (red arrows) is about 32
mm Hg at the arterial end of a capillary bed and 12 mm
Hg at the venous end; the mean colloid osmotic pressure
of tissues is approximately 25 mm Hg (green arrows),
which is equal to the mean capillary pressure. Although
fluid tends to leave the precapillary arteriole, it is
returned in equal amounts via the postcapillary venule,
so that the net flow (black arrows) in or out is zero. B,
Acute inflammation. Arteriole pressure is increased to 50
mm Hg, the mean capillary pressure is increased
because of arteriolar dilation, and the venous pressure
increases to approximately 30 mm Hg. At the same time,
osmotic pressure is reduced (averaging 20 mm Hg)
because of protein leakage across the venule. The net
result is an excess of extravasated fluid.
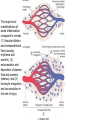
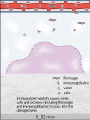
Diagrammatic representation of
five mechanisms of increased
vascular permeability in
inflammation
Diagrammatic representation of five
mechanisms of increased vascular
permeability in inflammation
Effects on the endothelial cells
• Formation of endothelial gaps in venules
due to contraction of the endothelial cells
and separation of intercellular junctions
• Direct endothelial injury, resulting in
endothelial cell necrosis and detachment.
• Leukocyte-mediated endothelial injury
• Leakage from new blood vessels.
in acute inflammation, fluid loss
from vessels with increased
permeability occurs in distinct
phases:
• (1) an immediate transient response lasting for
30 minutes or less, mediated mainly by the
actions of histamine and leukotrienes on
endothelium;
• (2) a delayed response starting at about 2 hours
and lasting for about 8 hours, mediated by
kinins, complement products, and other factors;
and
• (3) a prolonged response that is most noticeable
after direct endothelial injury, for example, after
burns.
• CELLULAR EVENTS: LEUKOCYTE
EXTRAVASATION AND
PHAGOCYTOSIS
A critical function of inflammation is
to
• deliver leukocytes to the site of injury and to
activate the leukocytes to perform their normal
functions in host defense.
• Leukocytes ingest offending agents, kill bacteria
and other microbes, and get rid of necrotic tissue
and foreign substances.
• A price that is paid for the defensive potency of
leukocytes is that they may induce tissue
damage and prolong inflammation, since the
leukocyte products that destroy microbes and
necrotic tissues can also injure normal host
tissues.
The multistep process of leukocyte
migration through blood vessels
• The leukocytes first roll, then become activated
and adhere to endothelium,
• then transmigrate across the endothelium,
pierce the basement membrane,
• and migrate toward chemoattractants emanating
from the source of injury.
• Different molecules play predominant roles in
different steps of this process-selectins in rolling;
chemokines in activating the neutrophils to
increase avidity of integrins (in green);
• integrins in firm adhesion
• The sequence of events in the journey of leukocytes
from the vessel lumen to the interstitial tissue, called
extravasation, can be divided into the following In the
lumen: margination, rolling, and adhesion to
endothelium.
• Vascular endothelium normally does not bind circulating
cells or impede their passage.
• In inflammation, the endothelium has to be activated to
permit it to bind leukocytes, as a prelude to their exit
from the blood vessels.
• Transmigration across the endothelium (also called
diapedesis)
• Migration in interstitial tissues toward a chemotactic
stimulus
• Because blood flow slows early in inflammation
(stasis), hemodynamic conditions change (wall
shear stress decreases), and more white cells
assume a peripheral position along the
endothelial surface. This process of leukocyte
accumulation is called margination.
• Subsequently, individual and then rows of
leukocytes tumble slowly along the endothelium
and adhere transiently (a process called rolling),
finally coming to rest at some point where they
adhere firmly (resembling pebbles over which a
stream runs without disturbing them).
• In time, the endothelium can be virtually lined by white
cells, an appearance called pavementing. After firm
adhesion, leukocytes insert pseudopods into the
junctions between the endothelial cells, squeeze through
interendothelial junctions, and assume a position
between the endothelial cell and the basement
membrane.
• Eventually, they traverse the basement membrane and
escape into the extravascular space. Neutrophils,
monocytes, lymphocytes, eosinophils, and basophils all
use the same pathway to migrate from the blood into
tissues.
• The next step in the process is migration of the
leukocytes through the endothelium, called
transmigration or diapedesis. Chemokines act on the
adherent leukocytes and stimulate the cells to migrate
through interendothelial spaces toward the chemical
After traversing the endothelium, leukocytes are
transiently retarded in their journey by the continuous
basement membrane of the venules, but eventually the
cells pierce the basement membrane, probably by
secreting collagenases. The net result of this process is
that leukocytes rapidly accumulate where they are
needed.
• Once leukocytes enter the extravascular
connective tissue, they are able to adhere
to the extracellular matrix by virtue of β1
integrins and CD44 binding to matrix
proteins. Thus, the leukocytes are retained
at the site where they are needed.
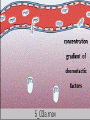
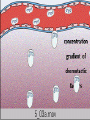
Chemotaxis
• After extravasation, leukocytes emigrate in
tissues toward the site of injury by a process
called chemotaxis, defined most simply as
locomotion oriented along a chemical gradient.
• All granulocytes, monocytes and, to a lesser
extent, lymphocytes respond to chemotactic
stimuli with varying rates of speed. Both
exogenous and endogenous substances can act
as chemoattractants. The most common
exogenous agents are bacterial products.
• Endogenous chemoattractants, which are
detailed later, include several chemical
mediators: (1) components of the
complement system, particularly C5a;
• (2) products of the lipoxygenase pathway,
mainly leukotriene B4 (LTB4); and
• (3) cytokines, particularly those of the
chemokine family (e.g., IL-8).
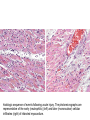
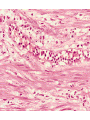
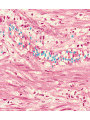
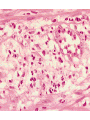
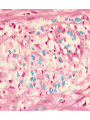
histologic sequence of events following acute injury. The photomicrographs are
representative of the early (neutrophilic) (left) and later (mononuclear) cellular
infiltrates (right) of infarcted myocardium.
Leukocyte Activation
• Microbes, products of necrotic cells,
antigen-antibody complexes, and
cytokines, including chemotactic factors,
induce a number of responses in
leukocytes
Phagocytosis
• Phagocytosis and the release of enzymes by neutrophils
and macrophages are responsible for eliminating the
injurious agents and thus constitute two of the major
benefits derived from the accumulation of leukocytes at
the inflammatory focus.
• Phagocytosis involves three distinct but interrelated
steps (
• (1) recognition and attachment of the particle to be
ingested by the leukocyte;
• (2) its engulfment, with subsequent formation of a
phagocytic vacuole; and
• (3) killing or degradation of the ingested material
CONTROLS OF ACUTE
INFLAMMATION
• inherent capacity to cause tissue damage,
• needs tight controls to minimize the damage.
• inflammation declines simply because the
mediators of inflammation have short half-lives
• , are degraded after their release,
• and are produced in quick bursts, only as long
as the stimulus persists.
• In addition as inflammation develops, the
process also triggers a variety of stop signals
that serve to actively terminate the reaction.42
CONTROLS OF ACUTE
INFLAMMATION
• switch in the production of proinflammatory leukotrienes to antiinflammatory lipoxins
• the liberation of an anti-inflammatory
cytokine,
• transforming growth factor-β (TGF-β),
from macrophages and other cells;
• {\B Neutrophils are the main effector cells in acute
inflammation}
• The neutrophil is the main cell to mediate the effects of
acute inflammation. If tissue damage is slight, an
adequate supply is derived from normal numbers
circulating in blood. If tissue damage is extensive, stores
of neutrophils, including some immature forms, are
released from bone marrow to increase the absolute
count of neutrophils in the blood. To maintain the supply
of neutrophils, growth factors derived from the
inflammatory process stimulate division of myeloid
precursors in the bone marrow, thereby increasing the
number of developing neutrophils.
• {\B Neutrophils adhere to endothelial cells prior to emigration}
• The adhesion of neutrophils to endothelium causes them to
aggregate along the vessel walls in a process termed margination.
Following margination, neutrophils migrate through the wall of the
vessel into the surrounding tissue in a process termed emigration,
as shown in {\I Fig. 5.3.}
• ¶{\B Activation of endothelium is a key process in acute
inflammation}
• Endothelium in local vessels is activated both by products of tissue
damage and by cytokines. This induces the expression of surface
cell adhesion molecules, which interact with complementary
molecules in the neutrophil cell membrane.
• Some of the factors involved in the activation of endothelial cells,
together with its role in neutrophil margination,
•
•
•
•
•
•
•
•
are summarized in the pink box opposite. The endothelium is modified to
become sticky for neutrophils, to secrete factors mediating vasodilation and
to promote platelet adhesion and aggregation
{\B Neutrophils are attracted to the area of tissue damage by chemical
mediators}
The movement of neutrophils from the vessel lumen into a damaged area is
mediated by substances known as chemotactic factors, which diffuse from
the area of tissue damage. The main neutrophil {\B chemotactic factors} are
listed in the Key Facts on mediators (see {\L page 67}). These factors bind
to receptors on the surface of neutrophils and activate secondary
messenger systems, stimulating increased cytosolic calcium, with resulting
assembly of cytoskeletal specializations involved in motility.
+}
FIGLEGEND {+
{\B Fig. 5.3 Neutrophil emigration in acute}
{\B
inflammation.}
+}
The acute inflammatory exudate is
derived from local vessels}
• The acute inflammatory exudate is composed of:
•
• {\B Fluid} containing salts and a high concentration of
•
proteins including immunoglobulins.
•
•
• {\B Fibrin,} a high molecular weight filamentous insoluble
•
protein.
•
•
• Many {\B neutrophil polymorphs,} from the blood white
•
cell population.
•
•
• A few {\B macrophages,} phagocytic cells derived from
•
blood monocytes.
•
•
• A few {\B lymphocytes.}
• All of these components are derived from
the blood as a result of changes that occur
in blood vessels in the surviving tissue
around the area of damage. These
changes, the vascular and cellular
responses of acute inflammation, are
illustrated diagrammatically in {\I Fig. 5.2.}
Briefly, the steps are :
•
•
•
•
•
•
•
•
•
•
•
{\B 1} Small blood vessels adjacent to the area of tissue
damage initially become dilated with increased blood
flow, then flow along them slows down.
{\B 2} Endothelial cells swell and partially retract so that they
no longer form a completely intact internal lining.
{\B 3} The vessels become leaky, permitting the passage of
water, salts, and some small, molecular-sized protein
from the plasma into the damaged area (exudation).
One of the main proteins to leak out is the small
soluble molecule, fibrinogen.
• {\B 4} Circulating neutrophil polymorphs initially adhere
to
•
the swollen endothelial cells (margination), then
•
actively migrate through the vessel basement
•
membrane (emigration), passing into the area of
tissue
•
damage.
• {\B 5} Later, small numbers of blood monocytes
•
(macrophages) migrate in a similar way, as do
•
lymphocytes.
• +}
• FIGLEGEND {+
• Death of tissue leads to release of substances (chemical mediators)
which act on nearby blood vessels.
• Mediators produce:
•
• Persistent vasodilatation and loss of axial flow
•
• Endothelial cell swelling and separation
•
• Increased permeability, with exudation of water, salts
•
and small proteins, including fibrinogen. Fibrinogen is
•
converted to fibrin.
• Mediators cause neutrophils to adhere to endothelium (margination),
and move through vessel walls into damaged tissue (emigration).
Blood monocytes/macrophages also emigrate by a similar
mechanism slightly later. The damaged area becomes progressively
replaced by the components
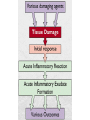
Acute inflammation is the most
common early tissue response
to tissue damage and
destruction
The acute inflammatory response
has three main functions:
• The affected area is occupied by a transient
material called the acute inflammatory exudate.
The exudate carries proteins, fluid, and cells
from local blood vessels into the damaged area
to mediate local defences.
• If an infective causative agent (e.g. bacteria) is
present in the damaged area, it can be
destroyed and eliminated by components of the
exudate.
• The damaged tissue can be broken down and
partly liquefied, and the debris removed from the
site of damage.
• The acute inflammatory response is
controlled by the production and diffusion
of chemical messengers derived both from
damaged tissues and from the acute
inflammatory exudate.
Overview of outcome
When tissue is damaged and
cells die, more than one
outcome is possible
restitution
• The damaged area may be replaced by
tissue identical in structure and function to
that originally present.
• this is the ideal outcome
It can take place
only if
• The damaging agent is removed
• The cell debris is cleared from the site
• The specialized cells which have been
destroyed have the capacity to re-grow or
regenerate.
most frequent outcome of
substantial tissue damage.
• If damaged cells cannot re-grow, or local
damage is so severe that tissue
architecture is entirely destroyed,
complete restitution of a damaged area is
not always possible.
• In this case, the tissue response is to heal
the damaged area by replacing it by nonspecialized scar tissue a process
termed fibrous repair.
If the damaging agent persists
• If the damaging agent persists (particularly if it involves
infection), continuing tissue destruction, attempts to heal
by fibrous repair and immune responses occur
concurrently, a process known as {\B chronic
inflammation.}
• ¶Irrespective of the ultimate outcome of tissue damage,
the initial response of the tissue is termed acute
inflammation or the acute inflammatory reaction. This
response is relatively non-specific, its main roles being to
clear away dead tissues, protect against local infection,
and allow the immune system access to the damaged
area.
• +}
Vascular and
exudative
changes in}
acute
inflammation.
• {\B Local vascular flow and permeability
alter in acute inflammation}
• The main vascular changes that arise in
acute inflammation are slowing of flow and
dilatation of vessels, and increase in the
permeability of their walls, allowing
diffusion of large proteins and fluid.
• {\B Fibrin in the acute inflammatory exudate may
have roles in allowing cell movement}
• Fibrin is a long, insoluble, filamentous protein,
formed by the polymerization of numerous
molecules of the smaller, soluble, precursor
plasma protein fibrinogen. The fibrinogen passes
out from vessels with the fluid and salts,
polymerizing into insoluble fibrin threads once
outside the vessel lumen by activation of the
blood coagulation cascade.
• ¶It is widely speculated that the network of
fibrin threads prevents migration of microorganisms and produces a scaffold which
might assist the migration of neutrophils
and macro-phages through the damaged
area. However, there is no real proof that
these are the precise functions of the
network.
• {\B Fluid in the acute inflammatory exudate carries
nutrients, mediators and immunoglobulins}
• It is logical to assume that the presence of fluids and
salts may dilute or buffer any locally produced toxins in
an area of tissue damage, but little more is known about
their precise functions in the acute inflammatory
reaction. Glucose and oxygen can diffuse into the area
of inflammation to support macrophages. Fluid also
allows diffusion of mediators of the inflammatory
process, particularly plasma-derived precursors (see {\L
page 66}).
• If immunity to an invading organism already
exists, immuno-globulins in the exudate act as
opsonins for neutrophil phagocytosis.
• The fluid in the exudate is not static but is
constantly circulating from local vessels, through
the extracellular space of the damaged tissue, to
be re-absorbed by lymphatics. This increased
flow of lymph takes antigens to the local nodes
and assists in later development of a specific
immune response.
• ¶{\B Cellular reactions are also needed in acute inflammation}
• The main cellular events in acute inflammation, all of which
• are caused by chemical mediators, are as follows:
•
• The normally inactive endothelium has to be activated to
•
allow adhesion of neutrophils.
•
• Normally inactive neutrophils have to be activated to
•
enhance their capacity for phagocytosis, bacterial killing,
•
and generation of inflammatory mediators.
•
• Neutrophils have to develop the ability to move actively,
•
in a directional fashion, from vessels towards the area of
•
tissue damage.
LFA
ELA2
• Endothelial activation in acute inflammation}
• The endothelium plays a vital role as a physical
barrier against diffusion of plasma outside
vessels, as well as being the source of many
regulatory molecules. Because of its extent and
its constant secretion of messenger substances,
the endothelium has been called the largest
endocrine organ in the body.
• The main factors secreted by the endothelium
are:
• • Nitric oxide and prostacyclin, which induce
vascular
•
relaxation and inhibit platelet aggregation.
• • Endothelin, thromboxane A2, and angiotensin
II, which
•
cause vascular constriction.
• • Growth factor PDGF promotes inhibitors, e.g.
heparin-like
•
substances.
• In the normal state the endothelium provides a
surface that prevents platelet aggregation and
degranulation. The balance of secreted factors is
a major determinant in control of regional blood
flow. In acute inflammation this balance is
changed and there is increased synthesis of a
lipid-derived molecule known as platelet
activating factor (PAF), which increases vascular
permeability; increased synthesis of nitric oxide,
which promotes vascular dilatation; and more
cell adhesion molecules are expressed, which
allows neutrophil adhesion.
• ¶In addition to modulation of secreted factors, the surface properties
of the endothelium are altered in acute inflammation (Fig. 5.4).
•
• IL-1 and TNF increase the expression of adhesion
•
molecules on the endothelium, especially P-selectin.
•
• Endothelial leukocyte adhesion molecule 1 (ELAM-1 or
•
E-selectin ) promotes adhesion of neutrophils.
•
• Intercellular adhesion molecule 1 (ICAM-1) promotes
•
adhesion of neutrophils and lymphoid cells.
•
• Vascular cell adhesion molecule 1 (VCAM-1)
•
promotes adhesion of lymphoid and monocyte cells.
• At the same time, other mediators of inflammation, particularly the
C5a fragment of complement, induce increased
• expression of complementary cell adhesion molecules on
neutrophils (CD11/CD18 complex).
• The endothelium in acute inflammation is therefore meta-bolically
altered to produce vasoactive factors (particularly PAF and nitric
oxide) as well as to be sticky for neutrophils.
• +}
• FIGLEGEND {+
• {\B Fig. 5.4 Cell adhesion molecules involved in }
• {\B
neutrophil adhesion.}
• {\B
IL-I interleukin-I, TNF tumour necrosis factor, }
• {\B
LTB4 leukotriene B4}
• +}