* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download File
Survey
Document related concepts
Transcript
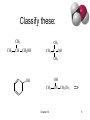
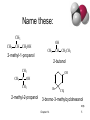
Chapter 14 Structure and Synthesis of Alcohols Classification • Primary: carbon with –OH is bonded to one other carbon. • Secondary: carbon with –OH is bonded to two other carbons. • Tertiary: carbon with –OH is bonded to three other carbons. • Aromatic (phenol): -OH is bonded to a benzene ring. Chapter 14 2 Classify these: CH3 CH3 CH3 CH CH2OH CH3 C OH CH3 OH OH CH3 Chapter 14 CH CH2CH3 => 3 IUPAC Nomenclature • Find the longest carbon chain containing the carbon with the -OH group. • Drop the -e from the alkane name, add ol. • Number the chain, starting from the end closest to the -OH group. • Number and name all substituents. => Chapter 14 4 Name these: CH3 CH3 CH CH2OH 2-methyl-1-propanol OH CH3 CH CH2CH3 2-butanol CH3 CH3 OH C OH CH3 2-methyl-2-propanol Br CH3 3-bromo-3-methylcyclohexanol => Chapter 14 5 Unsaturated Alcohols • Hydroxyl group takes precedence. Assign that carbon the lowest number. • Use alkene or alkyne name. OH CH2 CHCH2CHCH3 4-penten-2-ol (old) pent-4-ene-2-ol (1997 revision of IUPAC rules) => Chapter 14 6 Naming Diols • Two numbers are needed to locate the two -OH groups. • Use -diol as suffix instead of -ol. OH HO 1,6-hexanediol => Chapter 14 7 Practice • TIME FOR PRACTICE. • Try the handout review of alcohol naming Chapter 14 8 Physical Properties • Unusually high boiling points due to hydrogen bonding between molecules. • Small alcohols are miscible in water, but solubility decreases as the size of the alkyl group increases. => Chapter 14 9 Boiling Points FUNCTIONAL GROUP RANKING BY BOILING POINTS Name Amide B.P Rank Name Brief Explanation 222o 1 ethanamide hydrogen bonds on both the oxygen and the nitrogen hydrogen bonding from the of 2 oxygen atoms. hydrogen bonding from the presence one oxygen Acid 118o 2 ethanoic acid or acetic acid Alcohol 117o 3 propanol Chapter 14 10 Solubility in Water Solubility decreases as the size of the alkyl group increases. Chapter 14 => 11 USES FOR ALCOHOLS 1)Fuels 2)Solvents Chapter 14 12 Methanol • “Wood alcohol” • Common industrial solvent • Fuel at Indianapolis 500 Chapter 14 13 Ethanol • Fermentation of sugar and starches in grains • 12-15% alcohol, then yeast cells die. • MUST BE ANEROBIC or vinegar results • • • • Distillation produces “hard” liquors for drinking alcohol used as solvent Gasahol: 10% ethanol in gasoline Toxic dose: 200 mL ethanol, 30 mL methanol Chapter 14 14 Chapter 14 15 2-Propanol • “Rubbing alcohol” • Catalytic hydration of propene • Much more efficient makes 100% alcohol CH2 CH CH2 + H2O 100-300 atm, 300°C catalyst CH3 CH CH3 OH => Chapter 14 16 Biofuels Pros: -Some argue it’s Carbon neutral, but not quite, why? -Protects nonrenewable fuels needed for drugs and plastics • Cons • Food is scarce, should we burn it when people are hungry? • Uses a lot of land, water, energy to make Chapter 14 17 https://www.youtube.com/watch?v=C-qJ-6XJKBk Chapter 14 18 Reactions of Alcohols 1) Combustion: R-OH CO2 2) Dehydration: R-OH C=C (Zaitsev) Major and Minor Product 3) Oxidation: a) R-OH (Primary) R-C-HO R-COOH b) R-OH (secondary) R1R2-C=O c) Tertiary alcohols do not oxidize as the alpha carbon (containing OH) has no H’s Chapter 14 19 Combustion Complete C3H7OH(g) + 4.5O2 3CO2 + 4H2O _________________________________ Incomplete Combustion C3H7OH(g) + 3O2 3 CO + 4 H2O ALCOHOL OXIDATION 10 • gentle heating, a primary alcohol can be oxidised to produce an aldehyde. With H2SO4 or acidified [O] BUT • With strong heating and excess [O] (acidified) a carboxylic acid is formed. To heat strongly we need a Reflux apparatus is generally used to produce carboxylic acids. Note: Aldehydes must be distilled as they are formed to prevent further oxidation to carboxylic acids. Simply take away the reflux equipment and replace with distillation, see next slide. . OXIDIZING REAGENTS OXIDAIZING AGENT OBSERVATIONS OXIDIZING AGENTS REACTION ACIDIFIED DICROMATE ORANGE TO GREEN Cr2O72-(aq) to Cr3+(aq) ORANGE GREEN BENEDICT’S REAGENT BLUE TO RED Cu2+ + e- Cu+ BLUE Tollen's Reagent RED COLOURLESS TO Ag+(aq) + e- Ag(s) SILVER MIRROR COLOURLESS SILVER MIRROR Chapter 14 22 Ag+ Ag (s) Tollen's Reagent Chapter 14 23 No hard, nothing special • This reaction goes easily and quickly, • Nothing special is needed, other than the oxidizing agent is required, a hot water bath will do. The problem is when the aldehyde is made it boils off immediately. To form the –COOH we need to trap the aldehyde and force it back with reflux, or a condenser. Chapter 14 24 OXIDATION OF PRIMARY ALCOHOLS Controlling the products e.g. CH3CH2OH(l) + [O] ——> CH3CHO(l) + H2O(l) then CH3CHO(l) + [O] ——> CH3COOH(l) OXIDATION TO ALDEHYDES DISTILLATION OXIDATION TO CARBOXYLIC ACIDS REFLUX Aldehyde has a lower boiling point so distils off before being oxidised further Forces Aldehyde to condenses back into the mixture and gets oxidised to the acid OXIDATION OF PRIMARY ALCOHOLS Primary alcohols are easily oxidised to aldehydes e.g. CH3CH2OH(l) + [O] ethanol ——> CH3CHO(l) + H2O(l) ethanal it is essential to distil off the aldehyde before it gets oxidised to the acid CH3CHO(l) + [O] ethanal ——> CH3COOH(l) ethanoic acid Practical details • • • • the alcohol is dripped into a warm solution of acidified K2Cr2O7 aldehydes have low boiling points - no hydrogen bonding - they distil off immediately if it didn’t distil off it would be oxidised to the equivalent carboxylic acid to oxidise an alcohol straight to the acid, reflux the mixture compound formula intermolecular bonding ETHANOL C2H5OH HYDROGEN BONDING ETHANAL CH3CHO DIPOLE-DIPOLE ETHANOIC ACID CH3COOH HYDROGEN BONDING boiling point 78°C 23°C (volatile) 118°C Primary OH to HO green orange Simplified Molecular Equation Chapter 14 27 Stop OR Go • We can stop at OH to HO (aldehydes), • But the oxidizing agent is powerful and will continue unless we stop it. • 1) Use excess Alcohol reagent, so all the KMNO4 / K2Cr2O7 is used up • 2) Remove the Aldehyde as it forms via distillation (bp) Chapter 14 28 OH all the way to COOH Alcohol 10 Alcohol 10 Aldehyde C.A. A more simplified ONE step Version 1o Alcohol Carboxylic Acid 29 OH to HO, then COOH Chapter 14 30 Secondary Alcohols • Secondary alcohols are oxidised to produce ketones, then stop. Gentle heating as with aldehydes 2o OH to Ketones e.g. CH3CHOHCH3(l) + [O] propan-2-ol ——> Chapter 14 CH3COCH3(l) + H2O(l) propanone 32 ALCOHOL TO KETONE e.g CH3CHOHCH3(l) + [O] propan-2-ol ——> CH3COCH3(l) + H2O(l) propanone Chapter 14 33 Tertiary Alcohols • These are resistant to oxidation, there will be no colour change. • This is because the carbon (of the alcohol group) is not bonded to any other hydrogen atoms. • Thus, NO COLOUR CHANGE WITH [O] 3o lack a hydrogen Remember , hydrogen removal is the Definition of Oxidation, as is the addition of oxygen Chapter 14 35 Test Results Chapter 14 36 Dehydration of Alcohols Alcohol Alkene + water • when heated with an acid catalyst:such as a concentrated acid (a dehydrating agent) or pumice, or Al2O3 • with the loss of H and OH to form water alcohol alken e 37 Zaitsev’s Rule the dehydration of a secondary alcohol favors the product in which hydrogen is removed from the carbon atom in the smaller number of H atoms, the poor get poorer, or most substituted carbon chain with the 38 Zaitsev’s Rule… A shorter way of thinking of it is that the more substituted product is the one that is preferred, since it is more stable… so which is preferred here? 39 Disubstituted vs. monosubstituted… The pink structure (or 2-pentene) is preferred over 1pentene since it is disubstituted (vs. monosubstituted) 40 Try to draw the product of the reaction shown in your composition book (there is a hint on the next slide) Hint… Answer… Remember that the more substituted product is prefe (1-butene also forms, but it is the minor product). This one, then one more… 44 The more substituted product is preferred… 45 Pick out the product, last one Recall that Zaitsev’s rule indicates that the more substituted double bond will form… Under the OH, CH has 3 H’s, the green arrow the carbon in the hex chain has only 2 arrows Minor No reaction product Missing the double b