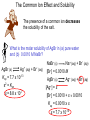* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 1412_lecture_ch16 Fall_2014
Rate equation wikipedia , lookup
Double layer forces wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
Stoichiometry wikipedia , lookup
Metastable inner-shell molecular state wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Electrolysis of water wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Elastic recoil detection wikipedia , lookup
Biological aspects of fluorine wikipedia , lookup
Electrochemistry wikipedia , lookup
Liquid–liquid extraction wikipedia , lookup
Acid–base reaction wikipedia , lookup
Calcium looping wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
Transition state theory wikipedia , lookup
Acid dissociation constant wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
Crystallization wikipedia , lookup
Coordination complex wikipedia , lookup
Determination of equilibrium constants wikipedia , lookup
Metalloprotein wikipedia , lookup
Chemical equilibrium wikipedia , lookup
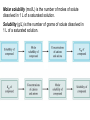
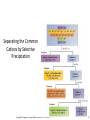
Chapter 16 Solubility and Complex Ion Equilibria Solubility Equilibria • Many natural processes depend on the precipitation or dissolving of a slightly soluble salt. In the next section, we look at the equilibria of slightly soluble, or nearly insoluble, ionic compounds. Their equilibrium constants can be used to answer questions regarding solubility and precipitation. The Solubility Product Constant • When an excess of a slightly soluble ionic compound is mixed with water, an equilibrium is established between the solid and the ions in the saturated solution. For the salt calcium oxalate, CaC2O4, you have the following equilibrium. CaC2O 4 (s ) H2O 2 2 Ca (aq) C 2O 4 (aq) The Solubility Product Constant • When an excess of a slightly soluble ionic compound is mixed with water, an equilibrium is established between the solid and the ions in the saturated solution. The equilibrium constant for this process is called the solubility product constant. 2 2 K sp [Ca ][C 2O 4 ] The Solubility Product Constant • In general, the solubility product constant is the equilibrium constant for the solubility equilibrium of a slightly soluble (or nearly insoluble) ionic compound. It equals the product of the equilibrium concentrations of the ions in the compound. Each concentration is raised to a power equal to the number of such ions in the formula of the compound. The Solubility Product Constant • In general, the solubility product constant is the equilibrium constant for the solubility equilibrium of a slightly soluble (or nearly insoluble) ionic compound. For example, lead iodide, PbI2, is another slightly soluble salt. Its equilibrium is: PbI 2 (s ) H2O 2 Pb (aq) 2I (aq) The Solubility Product Constant • In general, the solubility product constant is the equilibrium constant for the solubility equilibrium of a slightly soluble (or nearly insoluble) ionic compound. The expression for the solubility product constant is: 2 2 K sp [Pb ][I ] Calculating Ksp from the Solubility A 1.0-L sample of a saturated calcium oxalate solution, CaC2O4, contains 0.0061-g of the salt at 25 oC. Calculate the Ksp for this salt at 25 oC. We must first convert the solubility of calcium oxalate from 0.0061 g/liter to moles per liter. 1 mol CaC2O 4 M CaC2O 4 ( 0.0061 g CaC2O 4 / L ) 128g CaC2O 4 4.8 10 5 m ol CaC2O 4 / L Calculating Ksp from the Solubility A 1.0-L sample of a saturated calcium oxalate solution, CaC2O4, contains 0.0061-g of the salt at 25 oC. Calculate the Ksp for this salt at 25 oC. When 4.8 x 10-5 mol of solid dissolve it forms 4.8 x 10-5 mol of each ion. CaC2O 4 (s ) Starting Change Equilibrium H2O 2 2 Ca (aq) C 2O 4 (aq) 0 0 +4.8 x 10-5 +4.8 x 10-5 4.8 x 10-5 4.8 x 10-5 Calculating Ksp from the Solubility A 1.0-L sample of a saturated calcium oxalate solution, CaC2O4, contains 0.0061-g of the salt at 25 oC. Calculate the Ksp for this salt at 25 oC. You can now substitute into the equilibrium-constant expression. 2 2 K sp [Ca ][C 2O 4 ] 5 5 K sp ( 4.8 10 )( 4.8 10 ) K sp 2.3 10 9 Calculating Ksp from the Solubility By experiment, it is found that 1.2 x 10-3 mol of lead(II) iodide, PbI2, dissolves in 1.0 L of water at 25 oC. What is the Ksp at this temperature? Note that in this example, you find that 1.2 x 10-3 mol of the solid dissolves to give 1.2 x 103 mol Pb2+ and 2 x (1.2 x 10-3) mol of I-. Calculating Ksp from the Solubility By experiment, it is found that 1.2 x 10-3 mol of lead(II) iodide, PbI2, dissolves in 1.0 L of water at 25 oC. What is the Ksp at this temperature? The following table summarizes. PbI 2 (s ) Starting Change Equilibrium H2O 2 Pb (aq) 2I (aq) 0 0 +1.2 x 10-3 +2 x (1.2 x 10-3) 1.2 x 10-3 2 x (1.2 x 10-3) Calculating Ksp from the Solubility By experiment, it is found that 1.2 x 10-3 mol of lead(II) iodide, PbI2, dissolves in 1.0 L of water at 25 oC. What is the Ksp at this temperature? Substituting into the equilibrium-constant expression: 2 2 K sp [Pb ][I ] 3 3 K sp (1.2 10 )( 2 (1.2 10 )) K sp 6.9 10 9 2 Calculating Ksp from the Solubility By experiment, it is found that 1.2 x 10-3 mol of lead(II) iodide, PbI2, dissolves in 1.0 L of water at 25 oC. What is the Ksp at this temperature? If the solubility product constant is known, the solubility of the compound can be calculated. Calculating the Solubility from Ksp The mineral fluorite is calcium fluoride, CaF2. Calculate the solubility (in grams per liter) of calcium fluoride in water from the Ksp (3.4 x 10-11) Let x be the molar solubility of CaF2. CaF2 (s ) Starting Change Equilibrium H2O 0 +x x 2 Ca (aq) 2F (aq) 0 +2x 2x Calculating the Solubility from Ksp The mineral fluorite is calcium fluoride, CaF2. Calculate the solubility (in grams per liter) of calcium fluoride in water from the Ksp (3.4 x 10-11) You substitute into the equilibrium-constant equation 2 2 [Ca ][F ] K sp (x)(2x) 3.4 10 2 4x 3.4 10 3 11 11 Calculating the Solubility from Ksp The mineral fluorite is calcium fluoride, CaF2. Calculate the solubility (in grams per liter) of calcium fluoride in water from the Ksp (3.4 x 10-11) You now solve for x. 3.4 10 x3 4 -11 2.0 10 4 Calculating the Solubility from Ksp The mineral fluorite is calcium fluoride, CaF2. Calculate the solubility (in grams per liter) of calcium fluoride in water from the Ksp (3.4 x 10-11) Convert to g/L (CaF2 78.1 g/mol). 78.1g CaF2 solubility 2.0 10 mol / L 1 mol CaF2 4 2 1.6 10 g CaF2 / L Solubility Equilibria – Ion Product (reaction quotient) Q is defined as the ion product (reaction quotient) Q = same as Ksp only at initial concentration I.e. AgCl ↔ Ag+ + Cl- Q = [Ag+ ]0[Cl- ]0 The subscript 0 reminds us that these are initial concentrations Criteria for Precipitation • To determine whether an equilibrium system will go in the forward or reverse direction requires that we evaluate the reaction quotient, Qc. To predict the direction of reaction, you compare Qc with Kc (Chapter 14). The reaction quotient has the same form as the Ksp expression, but the concentrations of products are starting values. Criteria for Precipitation • To determine whether an equilibrium system will go in the forward or reverse direction requires that we evaluate the reaction quotient, Qc. Consider the following equilibrium. PbCl 2 (s ) H2O 2 Pb (aq) 2Cl (aq) Criteria for Precipitation • To determine whether an equilibrium system will go in the forward or reverse direction requires that we evaluate the reaction quotient, Qc. The Qc expression is Q c [Pb 2 2 ]i [Cl ]i where initial concentration is denoted by i. Criteria for Precipitation • To determine whether an equilibrium system will go in the forward or reverse direction requires that we evaluate the reaction quotient, Qc. If Qc exceeds the Ksp, precipitation occurs. If Qc is less than Ksp, more solute can dissolve. If Qc equals the Ksp, the solution is saturated. Predicting Whether Precipitation Will Occur The concentration of calcium ion in blood plasma is 0.0025 M. If the concentration of oxalate ion is 1.0 x 10-7 M, do you expect calcium oxalate to precipitate? Ksp for calcium oxalate is 2.3 x 10-9. The ion product quotient, Qc, is: 2 2 Q c [Ca ]i [C 2O 4 ]i -7 Q c (0.0025) (1.0 10 ) Q c 2.5 10 -10 Predicting Whether Precipitation Will Occur The concentration of calcium ion in blood plasma is 0.0025 M. If the concentration of oxalate ion is 1.0 x 10-7 M, do you expect calcium oxalate to precipitate? Ksp for calcium oxalate is 2.3 x 10-9. This value is smaller than the Ksp, so you do not expect precipitation to occur. Q c 2.5 10 -10 K sp Solubility Equilibria AgCl (s) Ksp = [Ag+][Cl-] MgF2 (s) Ag2CO3 (s) Ca3(PO4)2 (s) Ag+ (aq) + Cl- (aq) Ksp is the solubility product constant Mg2+ (aq) + 2F- (aq) Ksp = [Mg2+][F-]2 2Ag+ (aq) + CO32- (aq) Ksp = [Ag+]2[CO32-] 3Ca2+ (aq) + 2PO43- (aq) Ksp = [Ca2+]3[PO43-]2 Dissolution of an ionic solid in aqueous solution: Q < Ksp Unsaturated solution Q = Ksp Saturated solution Q > Ksp Supersaturated solution No precipitate Precipitate will form Molar solubility (mol/L) is the number of moles of solute dissolved in 1 L of a saturated solution. Solubility (g/L) is the number of grams of solute dissolved in 1 L of a saturated solution. What is the solubility of silver chloride in g/L ? AgCl (s) Initial (M) Change (M) Equilibrium (M) [Ag+] = 1.3 x 10-5 M Ag+ (aq) + Cl- (aq) 0.00 0.00 +s +s s s [Cl-] = 1.3 x 10-5 M Ksp = 1.6 x 10-10 Ksp = [Ag+][Cl-] Ksp = s2 s = Ksp s = 1.3 x 10-5 1.3 x 10-5 mol AgCl 143.35 g AgCl Solubility of AgCl = x = 1.9 x 10-3 g/L 1 L soln 1 mol AgCl If 2.00 mL of 0.200 M NaOH are added to 1.00 L of 0.100 M CaCl2, will a precipitate form? The ions present in solution are Na+, OH-, Ca2+, Cl-. Only possible precipitate is Ca(OH)2 (solubility rules). Is Q > Ksp for Ca(OH)2? [Ca2+]0 = 0.100 M [OH-]0 = 4.0 x 10-4 M Q = [Ca2+]0[OH-]02 = 0.10 x (4.0 x 10-4)2 = 1.6 x 10-8 Ksp = [Ca2+][OH-]2 = 8.0 x 10-6 Q < Ksp No precipitate will form Fractional Precipitation Selective Precipitation (Mixtures of Metal Ions) • Fractional precipitation is the technique of separating two or more ions from a solution by adding a reactant that precipitates first one ion, then another, and so forth. For example, when you slowly add potassium chromate, K2CrO4, to a solution containing Ba2+ and Sr2+, barium chromate precipitates first. Fractional Precipitation • Fractional precipitation is the technique of separating two or more ions from a solution by adding a reactant that precipitates first one ion, then another, and so forth. After most of the Ba2+ ion has precipitated, strontium chromate begins to precipitate. It is therefore possible to separate Ba2+ from Sr2+ by fractional precipitation using K2CrO4. What concentration of Ag is required to precipitate ONLY AgBr in a solution that contains both Br- and Cl- at a concentration of 0.02 M? AgBr (s) Ag+ (aq) + Br- (aq) Ksp = 7.7 x 10-13 Ksp = [Ag+][Br-] -13 K 7.7 x 10 sp -11 M = = 3.9 x 10 [Ag+] = 0.020 [Br-] AgCl (s) [Ag+] Ag+ (aq) + Cl- (aq) Ksp = 1.6 x 10-10 Ksp = [Ag+][Cl-] Ksp 1.6 x 10-10 -9 M = = 8.0 x 10 = 0.020 [Cl-] 3.9 x 10-11 M < [Ag+] < 8.0 x 10-9 M Solubility and the Common-Ion Effect • In this section we will look at calculating solubilities in the presence of other ions. The importance of the Ksp becomes apparent when you consider the solubility of one salt in the solution of another having the same cation. Solubility and the Common-Ion Effect • In this section we will look at calculating solubilities in the presence of other ions. For example, suppose you wish to know the solubility of calcium oxalate in a solution of calcium chloride. Each salt contributes the same cation (Ca2+) The effect is to make calcium oxalate less soluble than it would be in pure water. A Problem To Consider What is the molar solubility of calcium oxalate in 0.15 M calcium chloride? The Ksp for calcium oxalate is 2.3 x 10-9. Note that before the calcium oxalate dissolves, there is already 0.15 M Ca2+ in the solution. H2O 2 2 CaC2O 4 (s ) Ca (aq) C 2O 4 (aq) Starting Change Equilibrium 0.15 +x 0.15+x 0 +x x A Problem To Consider What is the molar solubility of calcium oxalate in 0.15 M calcium chloride? The Ksp for calcium oxalate is 2.3 x 10-9. You substitute into the equilibrium-constant equation 2 2 [Ca ][C 2O 4 ] K sp ( 0.15 x )( x ) 2.3 10 9 A Problem To Consider What is the molar solubility of calcium oxalate in 0.15 M calcium chloride? The Ksp for calcium oxalate is 2.3 x 10-9. Now rearrange this equation to give 9 2.3 10 2.3 10 x 0.15 x 0.15 9 We expect x to be negligible compared to 0.15. A Problem To Consider What is the molar solubility of calcium oxalate in 0.15 M calcium chloride? The Ksp for calcium oxalate is 2.3 x 10-9. Now rearrange this equation to give 9 2.3 10 2.3 10 x 0.15 x 0.15 x 1.5 10 8 9 A Problem To Consider What is the molar solubility of calcium oxalate in 0.15 M calcium chloride? The Ksp for calcium oxalate is 2.3 x 10-9. Therefore, the molar solubility of calcium oxalate in 0.15 M CaCl2 is 1.5 x 10-8 M. In pure water, the molarity was 4.8 x 10-5 M, which is over 3000 times greater. The Common Ion Effect and Solubility The presence of a common ion decreases the solubility of the salt. What is the molar solubility of AgBr in (a) pure water and (b) 0.0010 M NaBr? AgBr (s) Ag+ (aq) + Br- (aq) Ksp = 7.7 x 10-13 s2 = Ksp s = 8.8 x 10-7 NaBr (s) Na+ (aq) + Br- (aq) [Br-] = 0.0010 M AgBr (s) Ag+ (aq) + Br- (aq) [Ag+] = s [Br-] = 0.0010 + s 0.0010 Ksp = 0.0010 x s s = 7.7 x 10-10 Effect of pH on Solubility • Sometimes it is necessary to account for other reactions aqueous ions might undergo. For example, if the anion is the conjugate base of a weak acid, it will react with H3O+. You should expect the solubility to be affected by pH. Effect of pH on Solubility • Sometimes it is necessary to account for other reactions aqueous ions might undergo. Consider the following equilibrium. CaC2O 4 (s ) H2O 2 2 Ca (aq) C 2O 4 (aq) Because the oxalate ion is conjugate to a weak acid (HC2O4-), it will react with H3O+. 2 C 2O 4 (aq ) H 3O (aq) H2O HC 2O 4 (aq) H 2O(l) Effect of pH on Solubility • Sometimes it is necessary to account for other reactions aqueous ions might undergo. According to Le Chatelier’s principle, as C2O42- ion is removed by the reaction with H3O+, more calcium oxalate dissolves. Therefore, you expect calcium oxalate to be more soluble in acidic solution (low pH) than in pure water. pH and Solubility • • • The presence of a common ion decreases the solubility. Insoluble bases dissolve in acidic solutions Insoluble acids dissolve in basic solutions add Mg(OH)2 (s) remove Mg2+ (aq) + 2OH- (aq) Ksp = [Mg2+][OH-]2 = 1.2 x 10-11 Ksp = (s)(2s)2 = 4s3 4s3 = 1.2 x 10-11 s = 1.4 x 10-4 M [OH-] = 2s = 2.8 x 10-4 M pOH = 3.55 pH = 10.45 At pH less than 10.45 Lower [OH-] OH- (aq) + H+ (aq) H2O (l) Increase solubility of Mg(OH)2 At pH greater than 10.45 Raise [OH-] Decrease solubility of Mg(OH)2 Complex Ion Equilibria and Solubility A complex ion is an ion containing a central metal cation bonded to one or more molecules or ions. CoCl42- (aq) Co2+ (aq) + 4Cl- (aq) The formation constant or stability constant (Kf) is the equilibrium constant for the complex ion formation. Co(H2O)2+ 6 CoCl24 Kf = [CoCl42- ] [Co2+][Cl-]4 Kf stability of complex Complex-Ion Equilibria • Many metal ions, especially transition metals, form coordinate covalent bonds with molecules or anions having a lone pair of electrons. This type of bond formation is essentially a Lewis acid-base reaction. Complex-Ion Equilibria • Many metal ions, especially transition metals, form coordinate covalent bonds with molecules or anions having a lone pair of electrons. For example, the silver ion, Ag+, can react with ammonia to form the Ag(NH3)2+ ion. Ag 2(: NH 3 ) ( H 3 N : Ag : NH 3 ) Complex-Ion Equilibria • A complex ion is an ion formed from a metal ion with a Lewis base attached to it by a coordinate covalent bond. A complex is defined as a compound containing complex ions. A ligand is a Lewis base (an electron pair donor) that bonds to a metal ion to form a complex ion. Complex-Ion Formation The aqueous silver ion forms a complex ion with ammonia in steps. Ag (aq ) NH 3 (aq) Ag ( NH 3 ) (aq ) NH 3 (aq) Ag ( NH 3 ) (aq ) Ag ( NH 3 ) 2 (aq ) When you add these equations, you get the overall equation for the formation of Ag(NH3)2+. Ag (aq ) 2 NH 3 (aq) Ag ( NH 3 ) 2 (aq ) Complex-Ion Formation The formation constant, Kf , is the equilibrium constant for the formation of a complex ion from the aqueous metal ion and the ligands. The formation constant for Ag(NH3)2+ is: [ Ag( NH 3 )2 ] Kf 2 [ Ag ][NH 3 ] The value of Kf for Ag(NH3)2+ is 1.7 x 107. Complex-Ion Formation The formation constant, Kf, is the equilibrium constant for the formation of a complex ion from the aqueous metal ion and the ligands. The large value means that the complex ion is quite stable. When a large amount of NH3 is added to a solution of Ag+, you expect most of the Ag+ ion to react to form the complex ion. Complex-Ion Formation The dissociation constant, Kd , is the reciprocal, or inverse, value of Kf. The equation for the dissociation of Ag(NH3)2+ is Ag ( NH 3 ) 2 (aq ) Ag (aq ) 2 NH 3 (aq) The equilibrium constant equation is 2 1 [ Ag ][NH 3 ] Kd K f [ Ag( NH 3 )2 ] Equilibrium Calculations with Kf What is the concentration of Ag+(aq) ion in 0.010 M AgNO3 that is also 1.00 M NH3? The Kf for Ag(NH3)2+ is 1.7 x 107. In 1.0 L of solution, you initially have 0.010 mol Ag+(aq) from AgNO3. This reacts to give 0.010 mol Ag(NH3)2+, leaving (1.00- (2 x 0.010)) = 0.98 mol NH3. You now look at the dissociation of Ag(NH3)2+. Equilibrium Calculations with Kf What is the concentration of Ag+(aq) ion in 0.010 M AgNO3 that is also 1.00 M NH3? The Kf for Ag(NH3)2+ is 1.7 x 107. The following table summarizes: Ag ( NH 3 ) 2 (aq ) Starting Change Equilibrium 0.010 -x 0.010-x Ag (aq ) 2 NH 3 (aq) 0 +x x 0.98 +2x 0.98+2x Equilibrium Calculations with Kf What is the concentration of Ag+(aq) ion in 0.010 M AgNO3 that is also 1.00 M NH3? The Kf for Ag(NH3)2+ is 1.7 x 107. The dissociation constant equation is: 2 [ Ag ][NH 3 ] 1 Kd Kf [ Ag( NH 3 )2 ] Equilibrium Calculations with Kf What is the concentration of Ag+(aq) ion in 0.010 M AgNO3 that is also 1.00 M NH3? The Kf for Ag(NH3)2+ is 1.7 x 107. Substituting into this equation gives: ( x )(0.98 2x ) 1 7 ( 0.010 x ) 1.7 10 2 Equilibrium Calculations with Kf What is the concentration of Ag+(aq) ion in 0.010 M AgNO3 that is also 1.00 M NH3? The Kf for Ag(NH3)2+ is 1.7 x 107. If we assume x is small compared with 0.010 and 0.98, then 2 ( x )(0.98) 8 5.9 10 ( 0.010) Equilibrium Calculations with Kf What is the concentration of Ag+(aq) ion in 0.010 M AgNO3 that is also 1.00 M NH3? The Kf for Ag(NH3)2+ is 1.7 x 107. and 8 x 5.9 10 ( 0.010 ) ( 0.98 ) 2 6.1 10 10 The silver ion concentration is 6.1 x 10-10 M. Qualitative Analysis • Qualitative analysis involves the determination of the identity of substances present in a mixture. In the qualitative analysis scheme for metal ions, a cation is usually detected by the presence of a characteristic precipitate. Separating the Common Cations by Selective Precipitation Copyright © Cengage Learning. All rights reserved 63 WORKED EXAMPLES Worked Example 16.8 Worked Example 16.9 Worked Example 16.10 Worked Example 16.11 Worked Example 16.12 Worked Example 16.13 Worked Example 16.14 Worked Example 16.15 Worked Example 16.16