* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Carboxylic Acid Derivatives and Nucleophilic Acyl Substitution
Survey
Document related concepts
Transcript
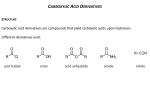
Chapter 15. Carboxylic Acid Derivatives Based on McMurry’s Organic Chemistry, 6th edition Carboxylic Compounds Acyl group bonded to Y, an electronegative atom or leaving group Includes: Y = halide (acid halides), acyloxy (anhydrides), alkoxy (esters), amine (amides), thiolate (thioesters), phosphate (acyl phosphates) 2 Naming Carboxylic Acid Derivatives Acid Halides, RCOX Derived from the carboxylic acid name by replacing the -ic acid ending with -yl or the -carboxylic acid ending with – carbonyl and specifying the halide 3 Naming Acid Anhydrides, RCO2COR' If symmetrical replace “acid” with “anhydride” based on the related carboxylic acid (for symmetrical anhydrides) From substituted monocarboxylic acids: use bis- ahead of the acid name Unsymmetrical anhydrides— cite the two acids alphabetically 4 Naming Amides, RCONH2 With unsubstituted NH2 group. replace -oic acid or -ic acid with -amide, or by replacing the -carboxylic acid ending with – carboxamide If the N is further substituted, identify the substituent groups (preceded by “N”) and then the parent amide 5 Naming Esters, RCO2R Name R’ and then, after a space, the carboxylic acid (RCOOH), with the “-ic acid” ending replaced by “-ate” 6 Nomenclature review 7 Nucleophilic Acyl Substitution Carboxylic acid derivatives have an acyl carbon bonded to a group Y that can leave A tetrahedral intermediate is formed and the leaving group is expelled to generate a new carbonyl compound, leading to substitution 8 Nucleophilic Acyl Substitution: General Reaction Pattern 9 Relative Reactivity of Carboxylic Acid Derivatives Nucleophiles react more readily with unhindered carbonyl groups More electrophilic carbonyl groups are more reactive to addition (acyl halides are most reactive, amides are least) The intermediate with the best leaving group decomposes fastest 10 Substitution in Synthesis We can readily convert a more reactive acid derivative into a less reactive one Reactions in the opposite sense are possible but require more complex approaches 11 General Reactions of Carboxylic Acid Derivatives water carboxylic acid alcohols esters ammonia or an amine an amide hydride source an aldehyde or an alcohol Grignard reagent a ketone or an alcohol 12 Mechanism of the Fischer Esterification The reaction is an acid-catalyzed, nucleophilic acyl substitution of a carboxylic acid When 18O-labeled methanol reacts with benzoic acid, the methyl benzoate produced is 18O-labeled but the water produced is unlabeled 13 Conversion of Carboxylic Acids into Acid Chlorides Reaction with thionyl chloride, SOCl2 14 Chemistry of Acid Halides Acid chlorides are prepared from carboxylic acids by reaction with SOCl2 Reaction of a carboxylic acid with PBr3 yields the acid bromide 15 Reactions of Acid Halides Nucleophilic acyl substitution Halogen replaced by OH, by OR, or by NH2 Reduction yields a primary alcohol Grignard reagent yields a tertiary alcohol 16 Hydrolysis: Conversion of Acid Halides into Acids Acid chlorides react with water to yield carboxylic acids HCl is generated during the hydrolysis: a base is added to remove the HCl 17 Conversion of Acid Halides into Esters Esters are produced in the reaction of acid chlorides with alcohols in the presence of pyridine or NaOH The reaction is better with less steric bulk 18 Aminolysis: Conversion of Acid Halides into Amides Amides result from the reaction of acid chlorides with NH3, primary (RNH2) and secondary amines (R2NH) The reaction with tertiary amines (R3N) gives an unstable species that cannot be isolated HCl is neutralized by the amine or an added base 19 Chemistry of Esters Many esters are pleasant-smelling liquids: fragrant odors of fruits and flowers Also present in fats and vegetable oils 20 Preparation of Esters Esters are usually prepared from carboxylic acids 21 Reactions of Esters Less reactive toward nucleophiles than are acid chlorides or anhydrides Cyclic esters are called lactones and react similarly to acyclic esters 22 Hydrolysis: Conversion of Esters into Carboxylic Acids An ester is hydrolyzed by aqueous base or aqueous acid to yield a carboxylic acid plus an alcohol 23 Mechanism of Base Catalyzed Ester Hydrolysis 24 Acid Catalyzed Ester Hydrolysis 25 Reduction: Conversion of Esters into Alcohols Reaction with LiAlH4 yields primary alcohols 26 Chemistry of Amides Prepared by reaction of an acid chloride with ammonia, monosubstituted amines, or disubstituted amines 27 Reactions of Amides Heating in either aqueous acid or aqueous base produces a carboxylic acid and amine Acidic hydrolysis by nucleophilic addition of water to the protonated amide, followed by loss of ammonia 28 Basic Hydrolysis of Amides Addition of hydroxide and loss of amide ion 29 Reduction: Conversion of Amides into Amines Reduced by LiAlH4 to an amine rather than an alcohol Converts C=O CH2 30 Uses of Reduction of Amides Works with cyclic and acyclic Good route to cyclic amines 31 End of chapter 15 32