* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 2672aLec17
Survey
Document related concepts
Transcript
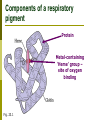
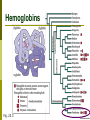
Biology 2672a: Comparative Animal Physiology Why is blood red (or green, or blue)? Gases dissolve in liquids Not the same as having air bubbles! Pliquid is proportional to Pair Amount of gas in solution depends on Temperature Salinity Gas Gases that have reacted chemically do not contribute to partial pressure in solution Blood must be thicker than water Solubility of O2 in water (especially warm salty water) not enough to provide O2 to active tissues Many organisms use respiratory pigments to bind O2 and transport it to tissues Respiratory pigments Diffusion into solution Respiratory pigment O2 in solution Air Blood O2 molecule bound to respiratory pigment is no longer in solution PO2 in blood, allows more to diffuse across (and more to bind to pigment) What it means to have a respiratory pigment Species Water Blood of an efficient bony fish Total Oxygen carrying capacity (ml O2/l) What it means to have a respiratory pigment Fig 22.4b Can not only suck a lot of O2 out of water, but can transport a lot per unit volume as well! Respiratory pigments Can be in solution or enclosed in blood cells Hematocrit Centrifuge whole blood and measure proportion of ‘solids’ (=cells) A pretty good measure of blood oxygen carrying capacity in vertebrates Components of a respiratory pigment Protein Metal-containing ‘Heme’ group – site of oxygen binding Fig. 23.1 Kinds of respiratory pigments Pigment Colour Hemoglobin Chlorocruorin Green Hemerythrin Violet/ Colourless Hemocyanin Structure O2 binding Protein + Heme + Fe2+ 1/Fe2+ Protein + Porphyrin + Fe2+ 1/2Fe2+ Protein + Cu2+ 1/2Cu2+ Chlorocruorins Found in four polychaete families: Serpulidae Sabellidae Chlorhaemidae Ampharetidae Hemerythrins Sipunculida Priapulida Brachiopoda Hemocyanins Some arthropods Many Molluscs Hemoglobins Fig. 23.3 Hb Oxygen association curve Fig. 23.4a Why is it a sigmoid curve? Cooperativity Cumulative increase in affinity as O2 binds to the heme groups Subunit changes conformation slightly, increasing affinity of other heme groups in that tetramer Subunit interaction Offloading O2 Lungs Tissues at rest Tissues in exercise Fig. 23.6 Affinity can change Fig. 23.4a The Bohr effect Exercising tissues produce CO2 and thus have pH As PCO2 increases (and pH decreases), affinity of Hb decreases This allows more O2 to be unloaded at sites where it is needed Affinity is still high at the blood-gas barrier for initial O2 uptake The Bohr Effect (~pH) Fig. 23.10b The Bohr Effect Fig. 23.11 Does CO2 bind to hemoglobin? Short answer: No Hb doesn’t drop off an O2 and pick up a CO2 to return to the lungs Most transport of CO2 is in solution (and often as carbonic acid/bicarbonate) Long answer: Yes CO2 binds to the Hb molecule (but not at the heme group) This binding alters the conformation of the protein (and contributes to the Bohr effect) But it isn’t the main means of CO2 transport. Other modulations of O2 affinity Temperature = O2 affinity Metabolic products, e.g. 2,3DPG = O2 affinity Inorganic ions? The Root Effect A change in amount of O2 bound at saturation, not (just) in affinity Used by fishes to offload O2 against gradients to fill swim bladder to supply O2 to oxygen-demanding retina Fig. 23.12 Myoglobin A monomeric globin found in muscle (esp. heart) Has a higher O2 affinity than Hb A sort of oxygen store for the cell Fig. 22.7a Neuroglobins Discovered in 2000 Monomeric, high O2 affinity Present in brain and retina of humans Protection from hypoxia Cytoglobins Discovered in 2002 Apparently present in all cells Also monomeric? Reading for Tuesday Osmoregulation in general Pp 663-679 Fish Osmoregulation Pp 681-699; Box 4.1