* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 8-Intro to Metabolism
Survey
Document related concepts
Transcript
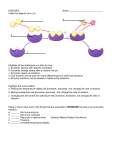
Name: ___________________________________________ Date: _____________________ Period: _____ Unit 3 Notes: Part A – Introduction to Metabolism AP Biology, Ms. Ottolini, 2012-2013 Metabolism= all the chemical reactions in an organism Catabolic Pathway (Catabolism) • release of energy by the breakdown of complex molecules to simpler compounds EX: digestive enzymes break down food Anabolic Pathway (Anabolism) 1) How are dehydration synthesis and hydrolysis related to catabolism and anabolism? • consumes energy to build complicated molecules from simpler ones EX: linking amino acids to form proteins Organisms transfer Energy (a characteristic of living organisms) Energy: capacity to do work Kinetic Energy: energy of moving objects Potential Energy: energy stored as a result of position or structure Chemical Energy- form of potential energy stored in chemical bonds in 2) Describe the forms of energy found in an apple as it grows on a tree, then falls and is digested by someone who eats it. molecules Thermodynamics: study of energy transformations that occur in matter 1st Law of Thermodynamics = Conservation of energy 3) How does the second law • energy of universe is constant; energy CAN BE transferred and transformed, but of thermodynamics help NEVER created or destroyed explain the diffusion of a 2nd Law of Thermodynamics substance across a • every energy transfer or transformation increases the entropy (disorder or membrane? randomness) in universe Equation that describes energy of system; G= H-T S - change in free energy is represented by G S = ENTROPY G = FREE ENERGY of a system (energy that is able to perform work when the temperature is uniform) H = Total energy in system T = Absolute temperature in °Kelvin You don’t need to be able to do G problems; just know that there is an equation; Endergonic Reaction- requires energy; absorbs free energy from system; not spontaneous, Energy of products is higher than energy of reactants (positive G) Exergonic Reaction- releases energy and occurs spontaneously Energy of products is lower than energy of reactants (negative G) Spontaneous Reactions: 4) Draw graphs to show the processes of endergonic and exergonic reations. x-axis: time y-axis: energy - can occur without outside help - can be harnessed to do work (objects moving down their power gradient) Cells manage their energy resources and do work by Energy Coupling (use energy from exergonic reactions to drive endergonic ones) Key role of ATP = Energy Coupling Adenosine Triphosphate = ATP = primary source of energy in all living things ADP (adenosine diphosphate) + Pi → ATP; -adding phosphate group stores energy; -removing it releases energy Activation Energy = amount of energy required to get chemical reaction started Catalyst- substance that changes the rate of a chemical reaction without being altered Enzymes = biological catalysts; most enzymes are Proteins (Ch 17 & 26: RNA enzymes = RIBOZYMES) ENZYMES work by Lowering Activation Energy; Don’t change the FREE ENERGY of reaction Substrate= Reactant enzyme acts on Active Site = region on enzyme that binds to substrate Substrate held in active site by WEAK interactions (ie. hydrogen and ionic bonds) SUBSTRATE(S) + enzyme → Enzyme-substrate complex → enzyme + PRODUCT(S) 5) Draw a graph to show how an enzyme-mediated reaction is different from a reaction that proceeds without the help of an enzyme. ENZYMES are Unchanged and Reusable Lock and Key Model: enzyme fits substrate like “lock and key” -only specific substrate will fit Induced Fit Model: once substrate binds to active site, enzyme changes shape slightly to bind the substrate more firmly placing a strain on the existing bonds in substrate lowering act energy Enzymes have Optimal Temperature for activity Higher temperatures = more collisions among the molecules so increase rate of a reaction BUT. . . above a certain temperature, activity begins to decline because the enzyme begins to Denature So rate of chemical reaction increases with temperature up to optimum, then decreases. Enzymes have own Optimal pH Different enzymes have different pH curves Extremes in pH can denature enzymes -causing them to unwind/lose their 3-D Tertiary structure -breaks hydrogen, ionic bonds; NOT covalent peptide bonds Many enzymes require Helpers Non-Protein Helper = Cofactor Ex: metal ions (zinc, iron, and copper) Organic Helpers = Coenzymes Ex: vitamins ; coenzyme A Certain molecules can prevent enzymes from performing their functions. This is called Inhibition Competitive Inhibitors: bind to the enzyme’s active site and prevent the substrate from binding (reversible) Noncompetitive Inhibitors: bind to another spot on the enzyme (not the active site), cause a shape change 6) Which temperature is optimal for the enzyme whose reaction rate is pictured to the left? 7) Pepsin is an enzyme found in the stomach, whereas trypsin is found in the small intestine. Both are digestive enzymes. Why is typsin not found in the small intestine, like pepsin? Non Competitive Inhibition is a type of Allosteric Regulation of enzyme activity REGULATORS bind to ALLOSTERIC site binding site on enzyme (not active site) binding changes shape of enzyme Activators can stimulate enzyme activity Inhibitors inhibit enzyme activity Cooperativity: If an enzyme has two or more subunits, a substrate causing induced fit in one subunit can trigger the same favorable conformational change in all the other subunits of the enzyme, amplifies the response of enzymes to substrates, one substrate molecule primes an enzyme to accept additional substrate molecules more rapidly Negative Feedback (feedback inhibition) - switches off pathway when product is plentiful - common in many enzyme reactions; - saves energy; don’t make it if you don’t need it 8) Which of the following characteristics is not associated with allosteric regulation of an enzyme’s activity? A. a mimic of the substrate competes for the active site B. a naturally occurring molecule stabilizes a catalytically active conformation C. Regulatory molecules bind to a site remote from the active site D. Inhibitors and activators may compete with one another. E. The enzyme usually has a quaternary structure. Positive Feedback – speeds up pathway - Less common EX: Chemicals released by platelets that accumulate at injury site, attract MORE platelets to the site. 9) During labor, a uterine contraction causes the release of the hormone oxytocin. This, in turn, causes a nerve stimulus that stimulates the brain’s hypothalamus to produce more oxytocin, which causes more contractions. This results in contractions increasing in amplitude and frequency. Is this an example of positive or negative feedback? Why?