* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Lanthanum has only one important oxidation state in aqueous
Hydroformylation wikipedia , lookup
Metal carbonyl wikipedia , lookup
Jahn–Teller effect wikipedia , lookup
Oxidation state wikipedia , lookup
Spin crossover wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Stability constants of complexes wikipedia , lookup
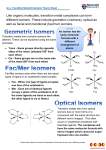
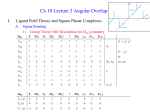
CHEM 163 Chapter 23 Spring 2009 http://www.youtube.com/watch?v=kfgtU9DDvdY Transition Metals • Large part of inorganic chemistry Electron Configurations Zn: [Ar]4s23d10 General form: Mn: [Ar]4s23d5 [noble gas] ns2 (n-1)dx n= 4 or 5 x = 1 to 10 [noble gas] ns2 (n-2)f14 (n-1)dx n= 6, or 7 Cr: [Ar]4s13d5 x = 1 to 10 Cu: [Ar]4s13d10 Transition Elements: Periodic Trends Across a period: – Atomic size: decreases, then remains constant • d e- are filling inner orbitals • Shield outer e- from nuclear pull – Electronegativity: increases slightly – Ionization Energy: increases slightly • d e- shield nuclear pull effectively Transition Elements: Periodic Trends Within a group: – Atomic size: no change • Increase in size between periods • Increase in nuclear charge (32!) decreased size – Electronegativity: increases slightly • More electronegative than elements in lower periods (increasing nuclear charge) – Ionization Energy: increases • Small increase in size; large increase in nuclear charge Density – Across a period: increase, then level off – Down a group: increase dramatically • Size constant Chemical Properties • Multiple oxidation states – Electrons close in E (all ready for bonding) – Highest oxidation state = group number for 3B(3) to 7B(7) – +2 oxidation state is common s electrons! • Metallic behavior – Lower oxidation state – ionic bonding – Higher oxidation state – covalent bonding • Reducing strength – All period 4 TMs form H2 from acid (except Cu) Color! • To absorb visible light, e- need a nearby higher E level • Main group ionic compounds have full outer shells – Next E level far away • Only colorless TM compounds include: – Sc3+, Ti4+, Zn2+ compounds of period 4 transition metals Magnetic Properties • Paramagnetic: unpaired electrons • Diamagnetic: all e- paired Attracted to an external magnetic field Unaffected by an external magnetic field • Most main-group metal ions have full shells • Compounds with TM ions typically have unpaired e• Compounds with TM ions with d0 or d10 are diamagnetic Inner Transition Elements • Lanthanides – “rare earth elements” • Not actually rare – – – – 14 elements Cerium (Z = 58) through Lutetium (Z = 71) Silvery, high-melting (800-1600 °C) Applications: • Tinted sunglasses • strongest known permanent magnet (SmCo5) • catalysts "Lanthanum has only one important oxidation state in • Actinides aqueous solution, the +3 state. With few exceptions, that statement tells the whole boring story about the other fourteen elements" - Pimental & Spratley (1971 textbook) – Radioactive! – Some never been seen/only made in labs Chromium • Protective coating of Cr2O3 forms in air • Exists in several oxidation states – CrO4 2- : yellow – Cr2O7 2- : orange – CrO3 : deep red – Cr3+ : blue/violet – Cr(OH)4 - : green • Cr (s) and Cr2+ : strong reducing agents • Cr6+ in acid: strong oxidizing agent Manganese • Used in steel alloys • more easy to work • tougher • Several oxidation states • Mn with oxidation states > +2 are good oxidizing agents • Does not easily oxidize in air • Already stable with d5 configuration Mercury • Forms bonds that can be ionic or more covalent • Can be found in the +1 oxidation state [Xe] 6s1 4f14 5d10 http://www.youtube.com/watch?v=oL0M_6bfzkU Silver • Soft – Sterling silver is alloyed with Cu to harden • Highest electrical conductivity of any element – 63.01 × 106 S/m (Cu: 59.6 × 106 S/m) • O.N. = +1 • Doesn’t form oxides in air • Tarnishes into Ag2S 2Al(s) 3Ag 2S(s) 6H2O(l ) 2Al(OH) 3 (s) 6Ag( s) 3H 2S( g ) o E 0.86V Strong reducing agent Black and White Photography Film: plastic coated in gelatin containing AgBr microcrystals Coordination Compounds TM form coordination compounds or complexes – Complex ion • central metal cation • ligands (Anions or molecules with lone pairs) – Counter ions • maintain charge neutrality Coordination compounds dissociate in water Complex Ions • Coordination Number: – # of ligand atoms bonded directly to the TM ion – Most common C.N. = 6 • Geometry: – C.N. = 2 – C.N. = 4 linear square planar tetrahedral – C.N. = 6 octahedral Ligands • Contain donor atoms – form covalent bond with metal (donates e- pair) • Monodentate: 1 donor atom • Bidentate: 2 donor atoms • Tridentate: 3 donor atoms Coordination Compound Formulas • Cation written before anion • Neutral ligands written before anionic ligands • Whole ion written in brackets – may be cationic or anionic • Charge of cation(s) balanced by charge of anion(s) Tetraaminebromochloroplatinum(IV) chloride [cation] [Pt(NH3)4BrCl] 2+ ClNeed 2Cl[Pt(NH3)4BrCl]Cl2 Potassium amminepentachloroplatinate (IV) K[Pt(NH3)Cl5] Naming Coordination Compounds • • • Name cation first, then anion Within the complex ion, ligands named (in alphabetical order) before TM Ligands – – – most molecules names stay same anions lose –ide; add –o Prefix tells how many • • • If ligand name already contains prefix, use: bis (2); tris (3); tetrakis (4) TM oxidation state in parentheses (if multiple possible) If complex ion is an anion, change to –ate K[Pt(NH3)Cl5] Isomers Compounds with same chemical formula, different properties • Constitutional Isomers: – Same atoms connected differently – [MA5B]B2 and [MA3B3]A2 • Linkage Isomers: – Same atoms, same complex ion, ligand connected differently – Nitrite ligand: NO2- Geometric Isomers • Same ligands, arranged differently around TM • cis- and trans- Optical Isomers • Physically identical except rotate polarized light differently • “enantiomers” (like hands) Bonding in Complexes • Coordinate covalent bond: – Both electrons from one atom • Complex ion is a Lewis adduct – Ligand: Lewis base – Metal: Lewis acid • Crystal Field theory – Describes d-orbital energies (TM) as ligands approach d orbitals dxy dxz dyz dz2 dx2 - y2 • In an atom- all d orbitals have same E • When part of coordination compound, E changes happen Crystal Field Splitting eg E dz2 dxy dx2 - y2 dxz ∆E dyz t2g • Large ∆E: strong field ligand • Small ∆E: weak field ligand Splitting of d orbitals • Minimized energy when ligands approach TM on axes • dxy dyz dxz lie between approaching ligands – minimal repulsion d orbital splitting dx2 - y2 E dz2 dx2 - y2 dxy dxz dxy dyz dz2 dxy dxz Octahedral dyz dz2 dx2 - y2 Tetrahedral dxz dyz Square Planar Color of TM • We see reflected/transmitted colors – Absorbed complementary color • For a given ligand, color depends on TM oxidation state • For a given metal, color depends on ligand field strength Magnetic Properties of TM [Mn(H2O)6]2+ Mn2+: d5 [Mn(CN)6]4- Mn2+: d5 E Weak-field ligand Strong-field ligand High spin Low spin