
Registration Penning electrons from atomic impurities in nonlocal
... current-voltage characteristics in which the observed spectrum of Penning electron energy (energy peaks) in a mixture of helium and argon in the pressure range of the main gas (helium) 18-100 Torr, and an impurity gas (argon) 0.1-1.7 %, as well as a mixture of helium and krypton at respective pressu ...
... current-voltage characteristics in which the observed spectrum of Penning electron energy (energy peaks) in a mixture of helium and argon in the pressure range of the main gas (helium) 18-100 Torr, and an impurity gas (argon) 0.1-1.7 %, as well as a mixture of helium and krypton at respective pressu ...
Atomic Structure
... Identify Group, Period, block, total # eWrite orbital notation/orbital diagrams to see specific quantum numbers for atoms/electrons lines/boxes with arrows (2 opposite arrows on each line) (Aufbau Principle = building up) ...
... Identify Group, Period, block, total # eWrite orbital notation/orbital diagrams to see specific quantum numbers for atoms/electrons lines/boxes with arrows (2 opposite arrows on each line) (Aufbau Principle = building up) ...
Physical Chemistry II Review Set 1
... c. The state function must always be real d. The integral of the wave function over "all space" = 1. 8. For a particle in a box of length 1nm: a. Sketch the ground state. b. Sketch the 3rd excited state. c. Using the principals of calculus, state qualitatively what you know about the derivative of t ...
... c. The state function must always be real d. The integral of the wave function over "all space" = 1. 8. For a particle in a box of length 1nm: a. Sketch the ground state. b. Sketch the 3rd excited state. c. Using the principals of calculus, state qualitatively what you know about the derivative of t ...
Precursors to Modern Physics
... atom is affected by large orbital quantum numbers? The state of an electron in an atom is completely defined by its quantum numbers. The energy of the electron is also a function of Z, the total positive charge of the nucleus. For the electrons with the same quantum numbers, what is the trend of the ...
... atom is affected by large orbital quantum numbers? The state of an electron in an atom is completely defined by its quantum numbers. The energy of the electron is also a function of Z, the total positive charge of the nucleus. For the electrons with the same quantum numbers, what is the trend of the ...
declaração - Sistema de Eventos
... that measures the TL distortion induced within a given material by laser beam, and it is related with the temperature coefficient of the optical path length change by ds/dT = (1/cp)ds/dQ, in which is the density and cp is the specific heat of the sample. This study is very important to classify t ...
... that measures the TL distortion induced within a given material by laser beam, and it is related with the temperature coefficient of the optical path length change by ds/dT = (1/cp)ds/dQ, in which is the density and cp is the specific heat of the sample. This study is very important to classify t ...
Midterm TEKS Check Review 1. Define the following terms
... 3. Identify the major contribution of each of the following to current atomic theory (be sure to include any model that they proposed and the experiment that they did): a. Dalton c. Bohr ...
... 3. Identify the major contribution of each of the following to current atomic theory (be sure to include any model that they proposed and the experiment that they did): a. Dalton c. Bohr ...
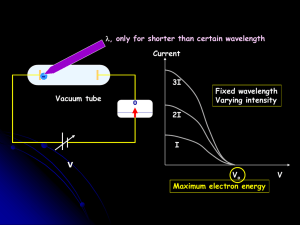
L 33 Modern Physics [1] Modern Physics
... the light is shorter than some maximum value, no matter how intense the light is, so the color ...
... the light is shorter than some maximum value, no matter how intense the light is, so the color ...
L 34 Modern Physics [1]
... orbits or states in which then do not radiate. • The electron in a high energy state can make a transition to a lower energy state by emitting a photon whose energy was the difference in energies of the two states, hf = Ei - Ef ...
... orbits or states in which then do not radiate. • The electron in a high energy state can make a transition to a lower energy state by emitting a photon whose energy was the difference in energies of the two states, hf = Ei - Ef ...
N 2
... atom must receive energy from the outside world. This can happen through a variety of mechanisms such as inelastic or semielastic collisions with other atoms and absorption of energy in the form of electromagnetic radiation (e.g., light). ...
... atom must receive energy from the outside world. This can happen through a variety of mechanisms such as inelastic or semielastic collisions with other atoms and absorption of energy in the form of electromagnetic radiation (e.g., light). ...
Document
... from Niels Bohr who explained experimentally observed discrete nature of atomic spectrum of Hydrogen. In spite of its immediate success in providing theoretical account of the spectrum and other nature of Hydrogen atom, a complete understanding of Bohr’s atom came only after de Broglie’s conjecture ...
... from Niels Bohr who explained experimentally observed discrete nature of atomic spectrum of Hydrogen. In spite of its immediate success in providing theoretical account of the spectrum and other nature of Hydrogen atom, a complete understanding of Bohr’s atom came only after de Broglie’s conjecture ...
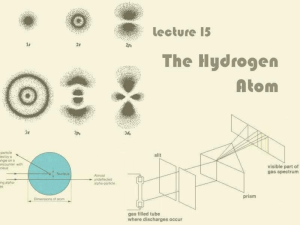
Lecture 15: The Hydrogen Atom
... It is known that accelerating charges emit radiation Thus, electron should emit radiation, lose energy and eventually fall into the nucleus! Why doesn’t this happen? Shows that something was wrong with this model of the hydrogen atom ...
... It is known that accelerating charges emit radiation Thus, electron should emit radiation, lose energy and eventually fall into the nucleus! Why doesn’t this happen? Shows that something was wrong with this model of the hydrogen atom ...
Properties of photons with similarities to waves and or particles
... This is also written as E = hf where f is the frequency of the light. Photons The modern theory of light is a merging of the wave and the particle models. Light is imagined to travel in a stream of packets or quanta of energy. The energy carried by each quantum is proportional to the frequency of li ...
... This is also written as E = hf where f is the frequency of the light. Photons The modern theory of light is a merging of the wave and the particle models. Light is imagined to travel in a stream of packets or quanta of energy. The energy carried by each quantum is proportional to the frequency of li ...
L 34 Modern Physics [1]
... orbits or states in which then do not radiate. • The electron in a high energy state can make a transition to a lower energy state by emitting a photon whose energy was the difference in energies of the two states, hf = Ei - Ef ...
... orbits or states in which then do not radiate. • The electron in a high energy state can make a transition to a lower energy state by emitting a photon whose energy was the difference in energies of the two states, hf = Ei - Ef ...
Chapter 7: Quantum Mechanical Model of Atom
... – Emission – excited electrons loses energy (usually by emitting light) and returns to a lower energy state or the ground state. – Atoms give off light when heated or otherwise excited energetically; thereby providing a clue as to their chemical makeup. ...
... – Emission – excited electrons loses energy (usually by emitting light) and returns to a lower energy state or the ground state. – Atoms give off light when heated or otherwise excited energetically; thereby providing a clue as to their chemical makeup. ...
Advantages of FTIR spectroscopy
... mid-infrared spectroscopy is is of the Fourier transform type. This is the reason why only FTIR technology will be described in the following. Bruker Optics has specialized in the field of FT-IR spectroscopy since 1974, and is one of the leading manufacturers of FT-IR, FT-NIR and FT-Raman spectromet ...
... mid-infrared spectroscopy is is of the Fourier transform type. This is the reason why only FTIR technology will be described in the following. Bruker Optics has specialized in the field of FT-IR spectroscopy since 1974, and is one of the leading manufacturers of FT-IR, FT-NIR and FT-Raman spectromet ...
APCh7MB
... Continuous spectrum – contains all the wavelengths over which the spectrum is continuous Line spectrum – contains certain specific wavelengths which are characteristics of the substances emitting those wavelengths There are specific energy levels among which an e- in a H atom can jump quantized ...
... Continuous spectrum – contains all the wavelengths over which the spectrum is continuous Line spectrum – contains certain specific wavelengths which are characteristics of the substances emitting those wavelengths There are specific energy levels among which an e- in a H atom can jump quantized ...
Mechanisms of Radio Wave Emission
... The electron and ion are initially free and moving rapidly. The ion attracts the electron but not strongly enough to capture it. As the charged particles are accelerated, they emit radio waves. ...
... The electron and ion are initially free and moving rapidly. The ion attracts the electron but not strongly enough to capture it. As the charged particles are accelerated, they emit radio waves. ...
Wavelength
... B. As electrons drop to lower levels, they give off photons C. A photon is a particle of electromagnetic radiation with no mass that carries a quantum of energy D. If the photon’s frequency corresponds the visible light range we can see different colors in the flame E. If it is not in the visible li ...
... B. As electrons drop to lower levels, they give off photons C. A photon is a particle of electromagnetic radiation with no mass that carries a quantum of energy D. If the photon’s frequency corresponds the visible light range we can see different colors in the flame E. If it is not in the visible li ...
Match the definit
... C. A fundamental substance that cannot be broken down into a simpler chemical substance D. The study and analysis of spectra E. A wave consisting of alternating electric and magnetic energy F. The theory that electromagnetic radiation may be treated as either a particle or an electromagnetic wave G. ...
... C. A fundamental substance that cannot be broken down into a simpler chemical substance D. The study and analysis of spectra E. A wave consisting of alternating electric and magnetic energy F. The theory that electromagnetic radiation may be treated as either a particle or an electromagnetic wave G. ...
Atomic and Molecular Physics for Physicists Ben-Gurion University of the Negev
... 1. Why do the helicities change upon reflection (this enables the atomchip setup)? 2. Until now we saw that all sigma (circular polatization) signs and/or helicities are the same for counter propagating beams in the MOT. Why is the above picture (also explaining a Mirror MOT) different? Hint: the en ...
... 1. Why do the helicities change upon reflection (this enables the atomchip setup)? 2. Until now we saw that all sigma (circular polatization) signs and/or helicities are the same for counter propagating beams in the MOT. Why is the above picture (also explaining a Mirror MOT) different? Hint: the en ...
L 35 Modern Physics [1] Modern Physics
... energy state to a low energy state it emits a photon Æ emission spectrum • An electron in a low energy state can absorb a photon and move up to a high energy state Æ absorption spectrum ...
... energy state to a low energy state it emits a photon Æ emission spectrum • An electron in a low energy state can absorb a photon and move up to a high energy state Æ absorption spectrum ...
X-ray fluorescence

X-ray fluorescence (XRF) is the emission of characteristic ""secondary"" (or fluorescent) X-rays from a material that has been excited by bombarding with high-energy X-rays or gamma rays. The phenomenon is widely used for elemental analysis and chemical analysis, particularly in the investigation of metals, glass, ceramics and building materials, and for research in geochemistry, forensic science and archaeology.






![L 33 Modern Physics [1] Modern Physics](http://s1.studyres.com/store/data/003217156_1-265c5a519e2bca3f33717b4abd842898-300x300.png)
![L 34 Modern Physics [1]](http://s1.studyres.com/store/data/008622077_1-047a8df5b8f51427a7d951942e25e95f-300x300.png)



![L 34 Modern Physics [1]](http://s1.studyres.com/store/data/001537103_1-dca58a96feb57d01fab60ba8bdd791ec-300x300.png)








![L 35 Modern Physics [1] - University of Iowa Physics](http://s1.studyres.com/store/data/000679677_1-b925cf8c8f031b0f2b0c09a806312d20-300x300.png)
![L 35 Modern Physics [1] Modern Physics](http://s1.studyres.com/store/data/001558975_1-84d6e03bc786b63795533f59711ce2f4-300x300.png)