Chapter 7
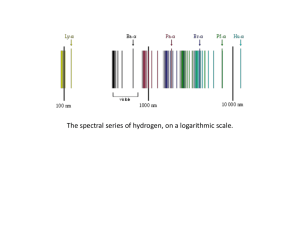
... • Heated solid metals emit light of all wavelengths, or a continuous spectrum – Would form a rainbow if sent through a prism • Heated gases emit light of only particular wavelengths, or a line spectrum – Would form only lines of particular colors if sent through a prism – These lines are associated ...
... • Heated solid metals emit light of all wavelengths, or a continuous spectrum – Would form a rainbow if sent through a prism • Heated gases emit light of only particular wavelengths, or a line spectrum – Would form only lines of particular colors if sent through a prism – These lines are associated ...
Ideas of Modern Physics
... 11. An electron is confined to a box of length L. It is in an excited state. The momentum of the particle is uncertain because a. the particle is not in the quantum ground state. b. the concept of momentum is not well-defined. c. the particle is moving in two different directions. d. the particle ha ...
... 11. An electron is confined to a box of length L. It is in an excited state. The momentum of the particle is uncertain because a. the particle is not in the quantum ground state. b. the concept of momentum is not well-defined. c. the particle is moving in two different directions. d. the particle ha ...
Electronic Structure and the Periodic Table A. Bohr Model of the
... A. Bohr Model of the Atom 1. Solar System Model 2. Created to Fit a “Quantized” Picture of Energy Transfer 3. Basis: Noncontinuous Emission Spectra of the Elements 4. Basic Postulates a. Electrons reside in certain allowed energy states b. Energy absorption and emission by atoms is caused by electro ...
... A. Bohr Model of the Atom 1. Solar System Model 2. Created to Fit a “Quantized” Picture of Energy Transfer 3. Basis: Noncontinuous Emission Spectra of the Elements 4. Basic Postulates a. Electrons reside in certain allowed energy states b. Energy absorption and emission by atoms is caused by electro ...
energy - U of L Class Index
... The higher the T, the lower the l, i.e. higher n Classical Physics predicts infinite energy emission, which violates the law of conservation of energy. Plank was able to show that the light intensity decays exponentially with wavelength allowing for energy to be conserved, and in so doing discovered ...
... The higher the T, the lower the l, i.e. higher n Classical Physics predicts infinite energy emission, which violates the law of conservation of energy. Plank was able to show that the light intensity decays exponentially with wavelength allowing for energy to be conserved, and in so doing discovered ...
Atomic Absorption Spectrometry
... radiation of the lines of the analyte are produced by the lamp; the presence of the atomic state of the analyte in the flame attenuates or reduces the intensity of the radiation. The absorbance is proportional to the concentration of the analyte, similar to ...
... radiation of the lines of the analyte are produced by the lamp; the presence of the atomic state of the analyte in the flame attenuates or reduces the intensity of the radiation. The absorbance is proportional to the concentration of the analyte, similar to ...
Complete nomenclature for electron orbitals
... be in a given place at a given time l Electron is not confined to any particular orbital distance from the nucleus but has a probability of being at various distances (with a maximum probability at the Bohr radius ao) l Think of the electron as being in an electron cloud ...
... be in a given place at a given time l Electron is not confined to any particular orbital distance from the nucleus but has a probability of being at various distances (with a maximum probability at the Bohr radius ao) l Think of the electron as being in an electron cloud ...
Notes - Photons, the Photoelectric Effect and the Compton Effect (ppt)
... • Again, experimental data shows this is not the case; increasing the intensity of the light only increases the number of electrons emitted, not their kinetic energy. • THUS the photoelectric effect is strong evidence for the photon model of light. ...
... • Again, experimental data shows this is not the case; increasing the intensity of the light only increases the number of electrons emitted, not their kinetic energy. • THUS the photoelectric effect is strong evidence for the photon model of light. ...
Nuclear Chemistry - Duluth High School
... Atoms are often converted into atoms of another element May involve protons, neutrons, and electrons Associated with large energy changes Reaction rates aren’t normally affected by temperature, pressure, or catalyst ...
... Atoms are often converted into atoms of another element May involve protons, neutrons, and electrons Associated with large energy changes Reaction rates aren’t normally affected by temperature, pressure, or catalyst ...
Quantum Theory - akugakbutuheksis
... An alternate view is that light is acting like a particle The light particle must have sufficient energy to “free” the electron from the atom. Increasing the Amplitude is simply increasing the number of light particles, but its NOT increasing the energy of each one! Increasing the Amplitude ...
... An alternate view is that light is acting like a particle The light particle must have sufficient energy to “free” the electron from the atom. Increasing the Amplitude is simply increasing the number of light particles, but its NOT increasing the energy of each one! Increasing the Amplitude ...
Problem Set 1 - MIT OpenCourseWare
... shown to be classically unstable, this is absolutely key. We will soon learn how to calculate the minimum (“ground state”) energy of such systems. iv. Use your dimensional analysis results to give a simple estimate for the ground state energy of this system. How does your estimate behave as h → 0? D ...
... shown to be classically unstable, this is absolutely key. We will soon learn how to calculate the minimum (“ground state”) energy of such systems. iv. Use your dimensional analysis results to give a simple estimate for the ground state energy of this system. How does your estimate behave as h → 0? D ...
Rutherford–Bohr model
... The Rutherford–Bohr model of the hydrogen atom (Z = 1) or a hydrogen-like ion (Z > 1), where the negatively charged electron confined to an atomic shell encircles a small, positively charged atomic nucleus and where an electron jump between orbits is accompanied by an emitted or absorbed amount of ...
... The Rutherford–Bohr model of the hydrogen atom (Z = 1) or a hydrogen-like ion (Z > 1), where the negatively charged electron confined to an atomic shell encircles a small, positively charged atomic nucleus and where an electron jump between orbits is accompanied by an emitted or absorbed amount of ...
Text Questions
... 10) What is the probability that an electron will be found within an atomic orbital? ...
... 10) What is the probability that an electron will be found within an atomic orbital? ...
7.3-Flame Test Lab
... different energies differently. Low energy red light is bent the least, and high energy violet the most. This allows us to see the various distinct colors of the emission spectrum of a sample. Finally, we will use the data we collect to identify a metal in an unknown salt solution. This process is t ...
... different energies differently. Low energy red light is bent the least, and high energy violet the most. This allows us to see the various distinct colors of the emission spectrum of a sample. Finally, we will use the data we collect to identify a metal in an unknown salt solution. This process is t ...
How electrons produce color
... • You will use the spectroscopes to see what photons are given off by the elements used in the flame test. • There will be some “background” light in your spectra. • Only focus on the bright lines! ...
... • You will use the spectroscopes to see what photons are given off by the elements used in the flame test. • There will be some “background” light in your spectra. • Only focus on the bright lines! ...
Introduction to RXS-CDW
... • If the core hole is an n=1 state, this is called a K-edge, if n=2 an L edge, if n=3 an M edge, and so on. • For example in an L-edge absorption of a transition metal one excites an electron from a full 2p shell to a 3d state. ...
... • If the core hole is an n=1 state, this is called a K-edge, if n=2 an L edge, if n=3 an M edge, and so on. • For example in an L-edge absorption of a transition metal one excites an electron from a full 2p shell to a 3d state. ...
Document
... ______ 15. Labeled each energy level in his atomic model with the principal quantum number, n. ______ 16. Wrote the equation E=h. ______ 17. The constant, h, is named for this scientist. ______ 18. Stated that you cannot observe or measure the “orbit” of an electron. Part IV: Drawing 19. Label the ...
... ______ 15. Labeled each energy level in his atomic model with the principal quantum number, n. ______ 16. Wrote the equation E=h. ______ 17. The constant, h, is named for this scientist. ______ 18. Stated that you cannot observe or measure the “orbit” of an electron. Part IV: Drawing 19. Label the ...
Quantum Physics Cumulative Review
... 4. An electron orbits the nucleus in a Bohr hydrogen atom. Calculate radius, speed and energy for each of the following orbits: a) n = 4 b) n = 8 5. Based on your previous results, what wavelength photon is produced when the electron in a hydrogen atom drops from the 8th energy level to the 4th ene ...
... 4. An electron orbits the nucleus in a Bohr hydrogen atom. Calculate radius, speed and energy for each of the following orbits: a) n = 4 b) n = 8 5. Based on your previous results, what wavelength photon is produced when the electron in a hydrogen atom drops from the 8th energy level to the 4th ene ...
Combining and Choosing Analytical Techniques
... Positive ions are formed in the ionisation chamber when an electron beam dislodges electrons from the sample atoms The positive ions are accelerated by an electric field The ions enter a magnetic field perpendicular to their path. This causes the ions to move in a curved path with a radius that depe ...
... Positive ions are formed in the ionisation chamber when an electron beam dislodges electrons from the sample atoms The positive ions are accelerated by an electric field The ions enter a magnetic field perpendicular to their path. This causes the ions to move in a curved path with a radius that depe ...
Bohr model
... • With the increase of grid potential, more electrons move to the plate and the current rises accordingly. • For mercury atoms, when V=4.9V, the electrons make inelastic collision and leave the atom jump to a high orbit (n=2). The original electrons move off with little energy and could not reach th ...
... • With the increase of grid potential, more electrons move to the plate and the current rises accordingly. • For mercury atoms, when V=4.9V, the electrons make inelastic collision and leave the atom jump to a high orbit (n=2). The original electrons move off with little energy and could not reach th ...
X-ray fluorescence

X-ray fluorescence (XRF) is the emission of characteristic ""secondary"" (or fluorescent) X-rays from a material that has been excited by bombarding with high-energy X-rays or gamma rays. The phenomenon is widely used for elemental analysis and chemical analysis, particularly in the investigation of metals, glass, ceramics and building materials, and for research in geochemistry, forensic science and archaeology.