Small Amplitude Short Period Crystal Undulators
... Ulrik I. Uggerhøj Department of Physics and Astronomy Aarhus University, Denmark On behalf of the collaborations CERN NA63 and SLAC E-212 ...
... Ulrik I. Uggerhøj Department of Physics and Astronomy Aarhus University, Denmark On behalf of the collaborations CERN NA63 and SLAC E-212 ...
The buoyant force on an object totally submerged in a fluid depends
... Predicts emission and absorption lines of hydrogen and hydrogen-like ions Predicts x-ray emissions (Moseley’s law) Gives an intuitive picture of what goes on in an atom The correspondence principle is obeyed... sort of It can’t easily be extended to more complicated atoms No prediction of rates, lin ...
... Predicts emission and absorption lines of hydrogen and hydrogen-like ions Predicts x-ray emissions (Moseley’s law) Gives an intuitive picture of what goes on in an atom The correspondence principle is obeyed... sort of It can’t easily be extended to more complicated atoms No prediction of rates, lin ...
Modern Atomic Theory (aka the electron chapter!)
... • Excited atoms can emit light. • Here the solution in a pickle is excited electrically. The Na+ ions in the pickle juice give off light characteristic of that element. ...
... • Excited atoms can emit light. • Here the solution in a pickle is excited electrically. The Na+ ions in the pickle juice give off light characteristic of that element. ...
Electrons in Atoms
... electricity through a tube filled with neon gas. Neon atoms release energy by emitting light. ...
... electricity through a tube filled with neon gas. Neon atoms release energy by emitting light. ...
Quiz 1 - sample quiz
... a) An electron jumps from a high energy orbital to a lower energy orbital when a photon of energy is emitted by an atom. b) The energy of light is directly proportional to its wavelength. c) The atomic emission spectrum consists of a series of discrete lines. d) The speed of light in a vacuum is con ...
... a) An electron jumps from a high energy orbital to a lower energy orbital when a photon of energy is emitted by an atom. b) The energy of light is directly proportional to its wavelength. c) The atomic emission spectrum consists of a series of discrete lines. d) The speed of light in a vacuum is con ...
Test 1 Guide
... 1) Neutrons have almost no mass (in amu) and no charge. 2) The fact that 2 different elements may have atoms with the same atomic mass violates Dalton’s atomic theory. 3) Deuterium differs from hydrogen in the number of protons it has. 4) In the quantum mechanical model of the atom, electrons are co ...
... 1) Neutrons have almost no mass (in amu) and no charge. 2) The fact that 2 different elements may have atoms with the same atomic mass violates Dalton’s atomic theory. 3) Deuterium differs from hydrogen in the number of protons it has. 4) In the quantum mechanical model of the atom, electrons are co ...
2.5 Bohr Model and Electron Energy
... The unit of light energy is referred to as a photon. iv. The unit of measurement for the energy lost OR gained by an atom is a quantum. ...
... The unit of light energy is referred to as a photon. iv. The unit of measurement for the energy lost OR gained by an atom is a quantum. ...
Problem Set 1 (Due January 30th by 7:00 PM) Answers to the
... Energy, Electrons and Periodic Trends (Bonus Date: January 26th) For all periodic trend problems, make sure that you can justify your answer. You will be expected to do this on an exam. 14. For each group of atoms, determine which would have a higher 1st Ionization Energy. a. Xe, Kr, Ar b. As, Cl, B ...
... Energy, Electrons and Periodic Trends (Bonus Date: January 26th) For all periodic trend problems, make sure that you can justify your answer. You will be expected to do this on an exam. 14. For each group of atoms, determine which would have a higher 1st Ionization Energy. a. Xe, Kr, Ar b. As, Cl, B ...
Chapter6_notes
... • Molecular Emission - results in a band spectrum due to the combination of the transition of electrons between atomic orbitals, molecular orbital, and vibrational states of the ...
... • Molecular Emission - results in a band spectrum due to the combination of the transition of electrons between atomic orbitals, molecular orbital, and vibrational states of the ...
Ch 16 – Quantam Physics
... The current flowing through the circuit is measured with a microammeter and the potential difference between the emitter and collector measured with a voltmeter ...
... The current flowing through the circuit is measured with a microammeter and the potential difference between the emitter and collector measured with a voltmeter ...
Chem 1st Sem Rev Ch2
... c. father of the modern atomic theory, everything made of atoms d. planetary model of the atom, electrons move around the nucleus like planets around sun. e. plum pudding model of the atom: atom looks like chocolate chip cookie f. gold foil experiment – atoms have a dense core called nucleus g. he g ...
... c. father of the modern atomic theory, everything made of atoms d. planetary model of the atom, electrons move around the nucleus like planets around sun. e. plum pudding model of the atom: atom looks like chocolate chip cookie f. gold foil experiment – atoms have a dense core called nucleus g. he g ...
electrons - TAMU Chemistry
... For electromagnetic radiation the velocity is 3.00 x 108 m/s and has the symbol c. Thus c = for electromagnetic radiation. ...
... For electromagnetic radiation the velocity is 3.00 x 108 m/s and has the symbol c. Thus c = for electromagnetic radiation. ...
Atomic Spectra - Northeast High School
... An atom’s only electron is in the fourth energy level (-2.35 eV). How many different photon energies can be emitted as this photon returns to the ground state (-15.8 eV)? What is the frequency of the photon that would be emitted if the electron returned to the ground state in a single transition? ...
... An atom’s only electron is in the fourth energy level (-2.35 eV). How many different photon energies can be emitted as this photon returns to the ground state (-15.8 eV)? What is the frequency of the photon that would be emitted if the electron returned to the ground state in a single transition? ...
Solutions to the exam itself are now available.
... would be spherical, and if you circled these, you were not penalized as long as you also circled Si and Cl.) (f) The Cr4+ ion in the compound CrO2 is exploited in magnetic audio and video recording tape. The electron configuration of Cr4+ is [Ar] 3d2. The question here is simple: which four electron ...
... would be spherical, and if you circled these, you were not penalized as long as you also circled Si and Cl.) (f) The Cr4+ ion in the compound CrO2 is exploited in magnetic audio and video recording tape. The electron configuration of Cr4+ is [Ar] 3d2. The question here is simple: which four electron ...
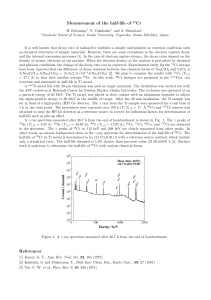
Measurement of the half-life of
... It is well known that decay rate of radioactive nuclides is usually independent on external conditions such as chemical structures of sample materials. However, there are some exceptions in the electron capture decay and the internal conversion processes [1]. In the case of electron capture decays, ...
... It is well known that decay rate of radioactive nuclides is usually independent on external conditions such as chemical structures of sample materials. However, there are some exceptions in the electron capture decay and the internal conversion processes [1]. In the case of electron capture decays, ...
CHEMISTRY 1A
... b. The shape of an atomic orbital is given by the quantum number _________. c. The maximum number of orbitals that may be associated with the following set of quantum numbers n = 5 and l = 3 is _________. d. The maximum number of electrons that may be associated with the quantum number set n = 4, l ...
... b. The shape of an atomic orbital is given by the quantum number _________. c. The maximum number of orbitals that may be associated with the following set of quantum numbers n = 5 and l = 3 is _________. d. The maximum number of electrons that may be associated with the quantum number set n = 4, l ...
484
... For practical uses, these formulae need to be worked out. Indeed, the energy dependance suggested by the explicit factor y must be corrected by taking into account h,. Beeing interested in small emittances, I shall concentrate first on the high energy part of the spectrum, where the divergence is th ...
... For practical uses, these formulae need to be worked out. Indeed, the energy dependance suggested by the explicit factor y must be corrected by taking into account h,. Beeing interested in small emittances, I shall concentrate first on the high energy part of the spectrum, where the divergence is th ...
Components of Optical Instruments
... common components which can be listed as: 1. A stable source of radiation 2. A wavelength selector to choose a single wavelength necessary for a certain absorption, emission or scattering process. ...
... common components which can be listed as: 1. A stable source of radiation 2. A wavelength selector to choose a single wavelength necessary for a certain absorption, emission or scattering process. ...
L16
... There are two major classes of interferences which can be identified in atomic absorption spectroscopy. The first class is related to spectral properties of components other than atomized analyte and is referred to as spectral interferences. The other class of interferences is related to the chemica ...
... There are two major classes of interferences which can be identified in atomic absorption spectroscopy. The first class is related to spectral properties of components other than atomized analyte and is referred to as spectral interferences. The other class of interferences is related to the chemica ...
Thursday, 1/29/09 - Liberty Union High School District
... •White light -continuous spectrum •Hydrogen atoms -line-emission spectrum •Big Question -Why did hydrogen atoms only give off specific frequency (colors) of light? ...
... •White light -continuous spectrum •Hydrogen atoms -line-emission spectrum •Big Question -Why did hydrogen atoms only give off specific frequency (colors) of light? ...
homework answers - SPHS Devil Physics
... d. Do calculations involving wavelengths of spectral lines and energy level differences e. Outline the Heisenberg Uncertainty Principle in terms of position-momentum and time-energy 3. Read section 6-5, Quantum Theory and the Uncertainty Principle (wait a minute . . . is that right? . . . no . . . y ...
... d. Do calculations involving wavelengths of spectral lines and energy level differences e. Outline the Heisenberg Uncertainty Principle in terms of position-momentum and time-energy 3. Read section 6-5, Quantum Theory and the Uncertainty Principle (wait a minute . . . is that right? . . . no . . . y ...
Physics 120 Homework Set #1 (due Sunday
... the container (the hole is very small so the probability that by chance it escapes back out is negligible). b) The spectrum is modified in such a way that its peak will be shifted towards shorter wavelengths. c) Classical theory predicted that the amount of radiation should increase indefinitely at ...
... the container (the hole is very small so the probability that by chance it escapes back out is negligible). b) The spectrum is modified in such a way that its peak will be shifted towards shorter wavelengths. c) Classical theory predicted that the amount of radiation should increase indefinitely at ...
Problem Set 05
... rotational energies of the molecule. [Result: n 2 2 /md 2 ] b) The wavelength of the photon needed to excite the molecule from the n to the n+1 rotational state. [Result: λ=2πcmd 2/(2n+1)] c) The wavelength of the photon need to excite € a nitrogen molecule (N2) from its ground state (n=0) to the ...
... rotational energies of the molecule. [Result: n 2 2 /md 2 ] b) The wavelength of the photon needed to excite the molecule from the n to the n+1 rotational state. [Result: λ=2πcmd 2/(2n+1)] c) The wavelength of the photon need to excite € a nitrogen molecule (N2) from its ground state (n=0) to the ...
X-ray fluorescence

X-ray fluorescence (XRF) is the emission of characteristic ""secondary"" (or fluorescent) X-rays from a material that has been excited by bombarding with high-energy X-rays or gamma rays. The phenomenon is widely used for elemental analysis and chemical analysis, particularly in the investigation of metals, glass, ceramics and building materials, and for research in geochemistry, forensic science and archaeology.