The Modern Nuclear Atom
... and protons but different numbers of neutrons. • Atomic number = number of protons • Mass number = the sum of the protons and neutrons in an atom’s nucleus ...
... and protons but different numbers of neutrons. • Atomic number = number of protons • Mass number = the sum of the protons and neutrons in an atom’s nucleus ...
CHM1045 - Michael Blaber
... E = h * (the relationship between energy and frequency for electromagnetic radiation En = -RH / n2 or En = -B / n2 (the relationship between the energy of an electron in Bohr's model of the hydrogen atom, and the orbit number of the electron) Elevel = RH * (1/ni2 - 1/nf2) or En = B * (1/ni2 - 1/n ...
... E = h * (the relationship between energy and frequency for electromagnetic radiation En = -RH / n2 or En = -B / n2 (the relationship between the energy of an electron in Bohr's model of the hydrogen atom, and the orbit number of the electron) Elevel = RH * (1/ni2 - 1/nf2) or En = B * (1/ni2 - 1/n ...
Chemistry 201/211 - Department of Chemistry | Oregon State
... 1.) The valve between a 5.0 L tank in which the gas pressure is 6.0 atm and a 10.0 L tank in which the pressure is 9.0 atm is opened. Assuming the connecting tube is zero volume and constant temperature throughout, the equilibrium final pressure is ...
... 1.) The valve between a 5.0 L tank in which the gas pressure is 6.0 atm and a 10.0 L tank in which the pressure is 9.0 atm is opened. Assuming the connecting tube is zero volume and constant temperature throughout, the equilibrium final pressure is ...
Chem 121 QU 78 Due in lecture
... DIRECTIONS: Write the correct answers in the appropriate blanks. Round – off answers (but not data) to 3 significant figures. Write units. Text: CHEMISTRY A molecular approach (2nd Ed.) by Tro. c= 3.00 X 108 m/s h = 6.63 X 10-34 j-s 1.00 m = 1.0 X 109 nm 1. Answer the next 4 questions concerning an ...
... DIRECTIONS: Write the correct answers in the appropriate blanks. Round – off answers (but not data) to 3 significant figures. Write units. Text: CHEMISTRY A molecular approach (2nd Ed.) by Tro. c= 3.00 X 108 m/s h = 6.63 X 10-34 j-s 1.00 m = 1.0 X 109 nm 1. Answer the next 4 questions concerning an ...
Name - Net Start Class
... 1. Define ‘extensive properties’ and give 3 examples a. Definition - a property that depends on how much matter is being considered. b. Ex. 1 Size c. Ex. 2 Mass d. Ex. 3 Volume 2. Define ‘intensive properties and give 3 examples. a. Definition - a property that does not depend on how much matter is ...
... 1. Define ‘extensive properties’ and give 3 examples a. Definition - a property that depends on how much matter is being considered. b. Ex. 1 Size c. Ex. 2 Mass d. Ex. 3 Volume 2. Define ‘intensive properties and give 3 examples. a. Definition - a property that does not depend on how much matter is ...
Absorption of Radiation
... wavelength of plane polarized light, concentration & number of symmetric molecules •circularly polarized light: the electric field vector is rotating around the axis of light propagation. • electric field vector can rotate in either the right or left direction, and the light is called right (Clockwi ...
... wavelength of plane polarized light, concentration & number of symmetric molecules •circularly polarized light: the electric field vector is rotating around the axis of light propagation. • electric field vector can rotate in either the right or left direction, and the light is called right (Clockwi ...
Document
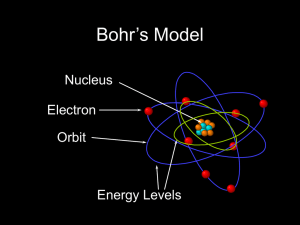
... • 1913 (Niels Bohr) proposed a new model that explained the hydrogen atom line emission spectrum • Also called planetary model • Electrons orbit nucleus at different levels • Closer to the nucleus means lower energy level; farther away is higher energy level ...
... • 1913 (Niels Bohr) proposed a new model that explained the hydrogen atom line emission spectrum • Also called planetary model • Electrons orbit nucleus at different levels • Closer to the nucleus means lower energy level; farther away is higher energy level ...
Einstein`s paper is a “bold, not to say reckless, hypothesis…which
... For very low light intensities, one can see the statistical and random nature of image formation. Use a sensitive camera that can detect single photons. ...
... For very low light intensities, one can see the statistical and random nature of image formation. Use a sensitive camera that can detect single photons. ...
Exam 3
... energy is released as fission power. 21. An energy band in a solid is a. A band connecting atoms in the crystal b. A region of high energy concentration in the crystal c. An energy range densely packed with quantum states d. A band of low-energy atoms in a crystal. e. A single quantum state on a gro ...
... energy is released as fission power. 21. An energy band in a solid is a. A band connecting atoms in the crystal b. A region of high energy concentration in the crystal c. An energy range densely packed with quantum states d. A band of low-energy atoms in a crystal. e. A single quantum state on a gro ...
Text Book: Fundamentals of Physics Authors: Halliday, Resnick
... (b)Certain features of the continuous spectrum are readily explained by the quantum theory. Thus the existence of a definite minimum wavelength can be justified if we assume that this radiation consists of x-ray photons produced by e-s which have given up all their K.E. in a single encounter with a ...
... (b)Certain features of the continuous spectrum are readily explained by the quantum theory. Thus the existence of a definite minimum wavelength can be justified if we assume that this radiation consists of x-ray photons produced by e-s which have given up all their K.E. in a single encounter with a ...
Chapter 7 Quantum Theory of the Atom
... Postulates of Bohr's Model which is the first connection between spectra of excited atoms and quantum ideas of Planck and Einstein 1) Energy level Postulate H atom has only certain allowable Energy Levels E=− ...
... Postulates of Bohr's Model which is the first connection between spectra of excited atoms and quantum ideas of Planck and Einstein 1) Energy level Postulate H atom has only certain allowable Energy Levels E=− ...
chapter-27
... wrong! (It said that an infinite amount of energy should be radiated by an object at finite temperature.) ...
... wrong! (It said that an infinite amount of energy should be radiated by an object at finite temperature.) ...
Instrumental Analysis as Applied to Architectural Materials
... date a building or element, to characterize decay mechanisms, or to evaluate materials for treatment. Most modern techniques have been used for decades. However, recent advancements have allowed for analysis of smaller samples and for quicker and more accurate interpretation. While the equipment and ...
... date a building or element, to characterize decay mechanisms, or to evaluate materials for treatment. Most modern techniques have been used for decades. However, recent advancements have allowed for analysis of smaller samples and for quicker and more accurate interpretation. While the equipment and ...
Physics 124 : Particles and Waves
... 1. Matter is stable, because there are no states lower in energy than the ground state; 2. Atoms emit and absorb a discrete spectrum of light, only photons that match the interval between stationary states can be emitted or absorbed; 3. Emission spectra can be produced by collisions; 4. Absorption w ...
... 1. Matter is stable, because there are no states lower in energy than the ground state; 2. Atoms emit and absorb a discrete spectrum of light, only photons that match the interval between stationary states can be emitted or absorbed; 3. Emission spectra can be produced by collisions; 4. Absorption w ...
Bohr Atom
... The Bohr model of the atom, like many ideas in the history of science, was at first prompted by and later partially disproved by experimentation. http://en.wikipedia.org/wiki/Category:Chemistry ...
... The Bohr model of the atom, like many ideas in the history of science, was at first prompted by and later partially disproved by experimentation. http://en.wikipedia.org/wiki/Category:Chemistry ...
File
... The radiation is divided into two beams, one of which passes through the sample while the other function as a reference beam. The reference and the sample beams are then passed alternately into a monochromator at very short intervals by means of a rotating mirror. ...
... The radiation is divided into two beams, one of which passes through the sample while the other function as a reference beam. The reference and the sample beams are then passed alternately into a monochromator at very short intervals by means of a rotating mirror. ...
Physics 324, Fall 2001 Solutions to problem set #1 Fri. 10/12/01
... We see that −ih̄∂t ψ = Eψ and −h̄2 ∇2 ψ = p2 ψ. Therefore one might be tempted to write the equation ...
... We see that −ih̄∂t ψ = Eψ and −h̄2 ∇2 ψ = p2 ψ. Therefore one might be tempted to write the equation ...
Chap. 3. Elementary Quantum Physics
... Fig. 3.3: Diffraction patterns obtained by passing X-rays through crystals can only be explained by using ideas based on the interference of waves. (a) Diffraction of X-rays from a single crystal gives a diffraction pattern of bright spots on a photographic film. (b) Diffraction of X-rays from a pow ...
... Fig. 3.3: Diffraction patterns obtained by passing X-rays through crystals can only be explained by using ideas based on the interference of waves. (a) Diffraction of X-rays from a single crystal gives a diffraction pattern of bright spots on a photographic film. (b) Diffraction of X-rays from a pow ...
Random Laser - Department of Physics
... The peak position and intensity change depends on temperature From the experiment and data that have been carried out it is found that when the temperature is increased the intensity of light emission decreases. When electrons excite the sample, other than the radiative effect (i.e. light emission) ...
... The peak position and intensity change depends on temperature From the experiment and data that have been carried out it is found that when the temperature is increased the intensity of light emission decreases. When electrons excite the sample, other than the radiative effect (i.e. light emission) ...
4PS Talk
... in time than the response. Using the delay stage to ‘scan in time’ along the response. 100 ps in time requires a path change of 15 mm. ...
... in time than the response. Using the delay stage to ‘scan in time’ along the response. 100 ps in time requires a path change of 15 mm. ...
Final “Intro Quantum Mechanics”
... (h) (T) The faster the wave function of a given particle oscillates in space, the more kinetic energy the particle has. The kinetic energy is determined by the second spatial derivative of the wave function. Cf. problem 2c. (i) (T) The x, y and z components of the spin of an electron do not commute ...
... (h) (T) The faster the wave function of a given particle oscillates in space, the more kinetic energy the particle has. The kinetic energy is determined by the second spatial derivative of the wave function. Cf. problem 2c. (i) (T) The x, y and z components of the spin of an electron do not commute ...
first chapter - damtp - University of Cambridge
... gravitational force, but this is about 10 40 times less strong, and so may be neglected. The protons and neutrons are held together in the nucleus by a di erent type of force, the nuclear force. The nuclear force is much stronger than the electrical force, and its attraction more than counteracts th ...
... gravitational force, but this is about 10 40 times less strong, and so may be neglected. The protons and neutrons are held together in the nucleus by a di erent type of force, the nuclear force. The nuclear force is much stronger than the electrical force, and its attraction more than counteracts th ...
X-ray fluorescence

X-ray fluorescence (XRF) is the emission of characteristic ""secondary"" (or fluorescent) X-rays from a material that has been excited by bombarding with high-energy X-rays or gamma rays. The phenomenon is widely used for elemental analysis and chemical analysis, particularly in the investigation of metals, glass, ceramics and building materials, and for research in geochemistry, forensic science and archaeology.