Grades 9-12 Chemistry California Content Standards
... b. the energy release per gram of material is much larger in nuclear fusion or fission reactions than in chemical reactions: change in mass (calculated by E=mc_) is small but significant in nuclear reactions. c. many naturally occurring isotopes of elements are radioactive, as are isotopes formed i ...
... b. the energy release per gram of material is much larger in nuclear fusion or fission reactions than in chemical reactions: change in mass (calculated by E=mc_) is small but significant in nuclear reactions. c. many naturally occurring isotopes of elements are radioactive, as are isotopes formed i ...
Chemistry - Gorman Learning Center
... c. energy is released when a material condenses or freezes and absorbed when a material evaporates or melts. d. how to solve problems involving heat flow and temperature changes, using known values of specific heat, and latent heat of phase change. e.* how to apply Hess's Law to calculate enthalpy c ...
... c. energy is released when a material condenses or freezes and absorbed when a material evaporates or melts. d. how to solve problems involving heat flow and temperature changes, using known values of specific heat, and latent heat of phase change. e.* how to apply Hess's Law to calculate enthalpy c ...
Compounds of Living Things
... blocks called amino acids. Most proteins are large molecules made of long-chain polymers of amino acids. Each type of protein in a cell has its own special structure. This structure depends on the way the amino acid chains fold together. Think of folding a shirt while doing laundry. There are differ ...
... blocks called amino acids. Most proteins are large molecules made of long-chain polymers of amino acids. Each type of protein in a cell has its own special structure. This structure depends on the way the amino acid chains fold together. Think of folding a shirt while doing laundry. There are differ ...
Chapter 18
... c. Vitamin E—prevents oxidation of vitamin A and polyunsaturated fatty acids and may help maintain stability of cell membranes. d. Vitamin K—needed for synthesis of prothrombin, which functions in blood clotting. Water-soluble vitamins and their functions include: e. Thiamine (Vitamin B1)—part of th ...
... c. Vitamin E—prevents oxidation of vitamin A and polyunsaturated fatty acids and may help maintain stability of cell membranes. d. Vitamin K—needed for synthesis of prothrombin, which functions in blood clotting. Water-soluble vitamins and their functions include: e. Thiamine (Vitamin B1)—part of th ...
Biology project Lz
... Western alchemists believed in vitalism. This is the theory that certain compounds could be synthesized only from their classical elements—earth, water, air, and fire—by the action of a "life-force" (vis vitalis) that only organisms possessed. Vitalism taught that these "organic" compounds were fund ...
... Western alchemists believed in vitalism. This is the theory that certain compounds could be synthesized only from their classical elements—earth, water, air, and fire—by the action of a "life-force" (vis vitalis) that only organisms possessed. Vitalism taught that these "organic" compounds were fund ...
Final Lecture Notes from 2002
... universe. This analysis is based on features of living systems on Earth (plant, animal and microbe), including those from very ...
... universe. This analysis is based on features of living systems on Earth (plant, animal and microbe), including those from very ...
The Molecular Basis of Life
... crystal, probably from before the time of the dinosaurs. In 2000, scientists revived them by giving them water and carbon-containing compounds. ...
... crystal, probably from before the time of the dinosaurs. In 2000, scientists revived them by giving them water and carbon-containing compounds. ...
The Living Planet
... However, since the nucleus of the oxygen atom (with 8 positively charged protons) attracts the negative electrons more than the one proton in the hydrogen atom, the electron pairs are not shared evenly; they are held more closely to the oxygen, so it is slightly negative… the hydrogens have their ne ...
... However, since the nucleus of the oxygen atom (with 8 positively charged protons) attracts the negative electrons more than the one proton in the hydrogen atom, the electron pairs are not shared evenly; they are held more closely to the oxygen, so it is slightly negative… the hydrogens have their ne ...
Search for Life in the Universe – What can we Learn from our own
... are considered to be indispensable, as well as the presence of liquid H2 O (e.g., Oro et al. 1982). The biogenic elements, which make up the bulk of terrestrial biomass, are among the most abundant elements in the Universe. Whether the organic starting material relevant to the origin of life came fr ...
... are considered to be indispensable, as well as the presence of liquid H2 O (e.g., Oro et al. 1982). The biogenic elements, which make up the bulk of terrestrial biomass, are among the most abundant elements in the Universe. Whether the organic starting material relevant to the origin of life came fr ...
Biochemistry notes File
... living in or close to water habitat. The denser water at 4oC allows animals to survive at the bottom of lakes during the winter. The ice floats and insulates the water below. Mammals sweat, and plants in deserts increase their transpiration, for thermoregulation and preventing overheating. Habitat f ...
... living in or close to water habitat. The denser water at 4oC allows animals to survive at the bottom of lakes during the winter. The ice floats and insulates the water below. Mammals sweat, and plants in deserts increase their transpiration, for thermoregulation and preventing overheating. Habitat f ...
Preview Sample 1
... A) excess hydrogen ions can change the shape of large complex molecules, rendering them nonfunctional. B) excess hydrogen ions can disrupt tissue functions. C) excess hydrogen ions can break chemical bonds. D) all of the above E) A and B only ...
... A) excess hydrogen ions can change the shape of large complex molecules, rendering them nonfunctional. B) excess hydrogen ions can disrupt tissue functions. C) excess hydrogen ions can break chemical bonds. D) all of the above E) A and B only ...
KS3 Biology - Science at St. Dominics
... • Know the ratio which describes carbohydrates? • Know the ratio of oxygen to hydrogen in carbohydrates? • Know the three types of carbohydrates and give examples of them? • Describe how to do a test for the presence of ...
... • Know the ratio which describes carbohydrates? • Know the ratio of oxygen to hydrogen in carbohydrates? • Know the three types of carbohydrates and give examples of them? • Describe how to do a test for the presence of ...
Ch 41 Notes
... Meat, eggs, and cheese provide all the essential amino acids and are thus “complete” proteins ...
... Meat, eggs, and cheese provide all the essential amino acids and are thus “complete” proteins ...
Key - UCSB CLAS
... to maximize H-bonds in the backbone) tertiary ⇒ the 3D structure of the entire protein quaternary ⇒ if a protein has more than one polypeptide chain (aka subunit) the quaternary structure is the way the individual subunits arrange themselves with respect to one another 13. Descibe denaturation and g ...
... to maximize H-bonds in the backbone) tertiary ⇒ the 3D structure of the entire protein quaternary ⇒ if a protein has more than one polypeptide chain (aka subunit) the quaternary structure is the way the individual subunits arrange themselves with respect to one another 13. Descibe denaturation and g ...
Evolution #1
... sugars, glycoproteins, and fats The theory that chemical evolution proceeded biological life is now generally accepted ...
... sugars, glycoproteins, and fats The theory that chemical evolution proceeded biological life is now generally accepted ...
16-1 The Importance of Food
... Many foods are good sources of one or more vitamins, especially vegetables, whole grains and meats ...
... Many foods are good sources of one or more vitamins, especially vegetables, whole grains and meats ...
Lesson 2.4 Biogeochemical Cycles
... cannot use this form of nitrogen. • Certain bacteria in the soil can change nitrogen gas (N2) into ammonia (NH3) by combing it with hydrogen gas (H2). This process is termed nitrogen fixation. • Plants take in nitrogen compounds through their roots. They use the nitrogen compounds to make proteins. ...
... cannot use this form of nitrogen. • Certain bacteria in the soil can change nitrogen gas (N2) into ammonia (NH3) by combing it with hydrogen gas (H2). This process is termed nitrogen fixation. • Plants take in nitrogen compounds through their roots. They use the nitrogen compounds to make proteins. ...
ch08
... developed under anoxic, aqueous conditions. It is possible that life arrived to this planet aboard a meteorite or a comet. The more likely option is that life arose on Earth. If so, it must have evolved in a place where contact with free oxygen of the atmosphere was limited. This “primordial soup” c ...
... developed under anoxic, aqueous conditions. It is possible that life arrived to this planet aboard a meteorite or a comet. The more likely option is that life arose on Earth. If so, it must have evolved in a place where contact with free oxygen of the atmosphere was limited. This “primordial soup” c ...
The Chemistry of Living Systems
... when scientists look for life on other planets, the first thing they look for is water. We believe Mars either had or has water on its surface or beneath its surface. That raises the tantalizing possibility that life may exist there. There are three important characteristics of water that make it es ...
... when scientists look for life on other planets, the first thing they look for is water. We believe Mars either had or has water on its surface or beneath its surface. That raises the tantalizing possibility that life may exist there. There are three important characteristics of water that make it es ...
The chemical building blocks of life Carbon
... Nitrogen has 3 covalent bonds which tend to form planar structures - Carbon is capable of forming complex molecules not only with itself, but also with H, O and N This is because the bonds C-C, C-H, C-O, and C-N have similar energies For instance, N can replace C in ring structures ...
... Nitrogen has 3 covalent bonds which tend to form planar structures - Carbon is capable of forming complex molecules not only with itself, but also with H, O and N This is because the bonds C-C, C-H, C-O, and C-N have similar energies For instance, N can replace C in ring structures ...
The Imprecise Search for Habitability
... to mirror that of the star, but not the volatiles. In other words, we expect the rocks of the exoplanet to mirror the metals in the star, but not its gas. This expectation is certainly met by Earth, whose nitrogen-dominated atmosphere hardly resembles the hydrogendominated Sun. On Earth, the amount ...
... to mirror that of the star, but not the volatiles. In other words, we expect the rocks of the exoplanet to mirror the metals in the star, but not its gas. This expectation is certainly met by Earth, whose nitrogen-dominated atmosphere hardly resembles the hydrogendominated Sun. On Earth, the amount ...
Unit 1 Powerpoint
... Scientists show the composition of compounds by a kind of shorthand known as a chemical formula. Water, which contains two atoms of hydrogen for each atom of oxygen, has the chemical formula =H2O. The formula for table salt, NaCl, indicates that the elements that make up table salt—sodium and chlori ...
... Scientists show the composition of compounds by a kind of shorthand known as a chemical formula. Water, which contains two atoms of hydrogen for each atom of oxygen, has the chemical formula =H2O. The formula for table salt, NaCl, indicates that the elements that make up table salt—sodium and chlori ...
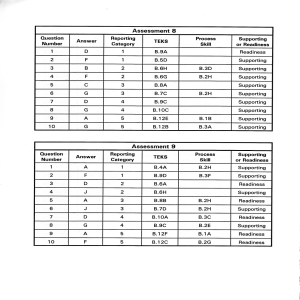
Assessment 8 Assessment I
... During rare rain events, the ocotillo rapidly grows small, green leaves and flowers. After it stops blooming, the ocotillo's leaves dry and fall off, and the ocotillo returns to its bare state. This behavior of the ocotillo is an example of ...
... During rare rain events, the ocotillo rapidly grows small, green leaves and flowers. After it stops blooming, the ocotillo's leaves dry and fall off, and the ocotillo returns to its bare state. This behavior of the ocotillo is an example of ...
rtf
... stars offers an incomparably more probable setting for the origin of life than any that was available on the early Earth. ...
... stars offers an incomparably more probable setting for the origin of life than any that was available on the early Earth. ...
urbano, mariajose
... • Have specific chemical and physical properties. • Are the regions of organic molecules which are commonly chemically reactive. • Behave consistently from one organic molecule to another. • Depending upon their number and arrangement, determine unique chemical properties of organic molecules in whi ...
... • Have specific chemical and physical properties. • Are the regions of organic molecules which are commonly chemically reactive. • Behave consistently from one organic molecule to another. • Depending upon their number and arrangement, determine unique chemical properties of organic molecules in whi ...
Abiogenesis

Abiogenesis (Brit.: /ˌeɪbaɪ.ɵˈdʒɛnɨsɪs/ AY-by-oh-JEN-ə-siss U.S. English pronunciation: /ˌeɪˌbaɪoʊˈdʒɛnᵻsɪs/), or biopoiesis, is the natural process of life arising from non-living matter, such as simple organic compounds. It is thought to have occurred on Earth between 3.8 and 4 billion years ago, and is studied through a combination of laboratory experiments and extrapolation from the genetic information of modern organisms in order to make reasonable conjectures about what pre-life chemical reactions may have given rise to a living system.The study of abiogenesis involves three main types of considerations: the geophysical, the chemical, and the biological, with more recent approaches attempting a synthesis of all three. Many approaches investigate how self-replicating molecules, or their components, came into existence. It is generally accepted that current life on Earth descended from an RNA world, although RNA-based life may not have been the first life to have existed. The Miller–Urey experiment and similar experiments demonstrated that most amino acids, basic chemicals of life, can be synthesized from inorganic compounds in conditions intended to be similar to early Earth. Several mechanisms have been investigated, including lightning and radiation. Other approaches (""metabolism first"" hypotheses) focus on understanding how catalysis in chemical systems in the early Earth might have provided the precursor molecules necessary for self-replication. Complex organic molecules have been found in the Solar System and in interstellar space, and these molecules may have provided starting material for the development of life on Earth.According to the panspermia hypothesis, microscopic life—distributed by meteoroids, asteroids and other small Solar System bodies—may exist throughout the Universe. It is speculated that the biochemistry of life may have begun shortly after the Big Bang, 13.8 billion years ago, during a habitable epoch when the age of the universe was only 10–17 million years.Nonetheless, Earth is the only place in the Universe known to harbor life. The age of the Earth is about 4.54 billion years. The earliest undisputed evidence of life on Earth dates at least from 3.5 billion years ago, during the Eoarchean Era after a geological crust started to solidify following the earlier molten Hadean Eon. There are microbial mat fossils found in 3.48 billion-year-old sandstone discovered in Western Australia. Other early physical evidence of a biogenic substance is graphite in 3.7 billion-year-old metasedimentary rocks discovered in southwestern Greenland.