one dimensional steady state heat conduction
... In each equation the dependent variable, T, is a function of 4 independent variables, (x,y,z,τ); (r, ,z,τ); (r,φ,θ,τ) and is a 2nd order, partial differential equation. The solution of such equations will normally require a numerical solution. For the present, we shall simply look at the simplific ...
... In each equation the dependent variable, T, is a function of 4 independent variables, (x,y,z,τ); (r, ,z,τ); (r,φ,θ,τ) and is a 2nd order, partial differential equation. The solution of such equations will normally require a numerical solution. For the present, we shall simply look at the simplific ...
module 2
... In each equation the dependent variable, T, is a function of 4 independent variables, (x,y,z,τ); (r, ,z,τ); (r,φ,θ,τ) and is a 2nd order, partial differential equation. The solution of such equations will normally require a numerical solution. For the present, we shall simply look at the simplific ...
... In each equation the dependent variable, T, is a function of 4 independent variables, (x,y,z,τ); (r, ,z,τ); (r,φ,θ,τ) and is a 2nd order, partial differential equation. The solution of such equations will normally require a numerical solution. For the present, we shall simply look at the simplific ...
thermochemistry - Pace University Webspace
... • Gibbs Free Energy is a measure of nonpV work that must go into a reaction to make it occur (when G is positive) or work that a reaction can do (when negative). It can be shown that the change of Gibbs Free Energy over temperature in a reaction is the change of the total entropy of the reacting sys ...
... • Gibbs Free Energy is a measure of nonpV work that must go into a reaction to make it occur (when G is positive) or work that a reaction can do (when negative). It can be shown that the change of Gibbs Free Energy over temperature in a reaction is the change of the total entropy of the reacting sys ...
Thermo-regulation - Learning Central
... How the body conserves and produces heat (e.g. when in cold environment) Endocrine system response Cooling of specific area in hypothalamus (preoptic area) increase in secretion of thyrotropin releasing hormone (TRH - from hypothalamus) stimulates secretion of thyroid stimulating hormone (TSH - ...
... How the body conserves and produces heat (e.g. when in cold environment) Endocrine system response Cooling of specific area in hypothalamus (preoptic area) increase in secretion of thyrotropin releasing hormone (TRH - from hypothalamus) stimulates secretion of thyroid stimulating hormone (TSH - ...
FIXED TEMPERATURE HEAT DETECTOR 70°C WATER
... Ease of installation and wiring connection reduces installation costs ...
... Ease of installation and wiring connection reduces installation costs ...
L14
... to allow it to cool down the rest of the way until you’re back where you started. The net result is that the heat you added to the compressed gas was partially converted to work, and partially released to the environment as waste heat. The efficiency of the energy is the work divided by the heat add ...
... to allow it to cool down the rest of the way until you’re back where you started. The net result is that the heat you added to the compressed gas was partially converted to work, and partially released to the environment as waste heat. The efficiency of the energy is the work divided by the heat add ...
Allow the sun`s warm rays in and heat up energy
... Allow the sun’s warm rays in and heat up energy savings. When the cold of winter comes, nothing feels better than the sun’s warming rays through big, bright windows. The new low-E from Guardian – ClimaGuard 72/57 – incorporates a passive solar heat concept to provide a powerful level of insulation a ...
... Allow the sun’s warm rays in and heat up energy savings. When the cold of winter comes, nothing feels better than the sun’s warming rays through big, bright windows. The new low-E from Guardian – ClimaGuard 72/57 – incorporates a passive solar heat concept to provide a powerful level of insulation a ...
ppt
... Conservation of Energy Latent heat is the energy given up or taken up by a system to cause a change of phase, such as water vapor condensing into liquid water. It is a key to understanding weather because latent heat is a major source of energy for thunderstorms and hurricanes. For evaporation, ene ...
... Conservation of Energy Latent heat is the energy given up or taken up by a system to cause a change of phase, such as water vapor condensing into liquid water. It is a key to understanding weather because latent heat is a major source of energy for thunderstorms and hurricanes. For evaporation, ene ...
Name
... 1. A 2-kg beaker of water has a temperature of 15°C. If 2 more kg of water at 15°C are added, the temperature becomes 30°C. 2. Thermal Energy only moves from particles at a lower temperature to particles at a higher temperature. 3. Friction converts thermal energy into mechanical energy. 4. Heat can ...
... 1. A 2-kg beaker of water has a temperature of 15°C. If 2 more kg of water at 15°C are added, the temperature becomes 30°C. 2. Thermal Energy only moves from particles at a lower temperature to particles at a higher temperature. 3. Friction converts thermal energy into mechanical energy. 4. Heat can ...
Unit 4: Themodynamics
... Endo v. Exo Pre Lab Questions Endo v. Exo Lab Notes on Specific Heat ...
... Endo v. Exo Pre Lab Questions Endo v. Exo Lab Notes on Specific Heat ...
Heat And Thermodynamics - Figure B
... Heat is a form of energy which appears when two bodies at different temperature are placed into thermal contact. It can flow from high temperature to low temperature till temperature of the two bodies becomes same. Thus, we can say that heat is the energy in transit. Heat is not property of system, ...
... Heat is a form of energy which appears when two bodies at different temperature are placed into thermal contact. It can flow from high temperature to low temperature till temperature of the two bodies becomes same. Thus, we can say that heat is the energy in transit. Heat is not property of system, ...
Specific Heat
... Example: Calculate the joules of energy required to heat 454 g of water from 5.4 degrees Celsius to 98.6 degrees Celsius. ...
... Example: Calculate the joules of energy required to heat 454 g of water from 5.4 degrees Celsius to 98.6 degrees Celsius. ...
Measuring the Specific Heat Capacity of Water
... Energy to heat 1kg (1000ml) of water by 1˚C = __________ × 10 = __________ J ...
... Energy to heat 1kg (1000ml) of water by 1˚C = __________ × 10 = __________ J ...
Worksheet- Calculations involving Specific Heat
... Worksheet- Calculations involving Specific Heat 1. For q= m ●c ● Δ T : identify each variables by name & the units associated with it. q = amount of heat (J) m = mass (grams) c = specific heat (J/g°C) ΔT = change in temperature (°C) 2. Heat is not the same as temperature, yet they are related. Expla ...
... Worksheet- Calculations involving Specific Heat 1. For q= m ●c ● Δ T : identify each variables by name & the units associated with it. q = amount of heat (J) m = mass (grams) c = specific heat (J/g°C) ΔT = change in temperature (°C) 2. Heat is not the same as temperature, yet they are related. Expla ...
Chapter 7 Thermal and Energy Systems
... • The physical property that quantifies the amount of heat that must flow into or out of a material to produce a phase change is called the latent heat. • Transfer of Heat • heat as energy that is being transferred from one location to another because of a temperature difference. • The three mechan ...
... • The physical property that quantifies the amount of heat that must flow into or out of a material to produce a phase change is called the latent heat. • Transfer of Heat • heat as energy that is being transferred from one location to another because of a temperature difference. • The three mechan ...
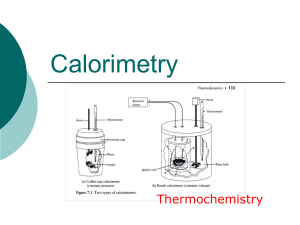
Thermochemistry - hrsbstaff.ednet.ns.ca
... one another (insulation), then filled with a specific quantity of water and covered with another cup as a cover. A chemical reaction or phase change takes place inside and a thermometer is placed within to measure any change in temperature that occurs to the system. ...
... one another (insulation), then filled with a specific quantity of water and covered with another cup as a cover. A chemical reaction or phase change takes place inside and a thermometer is placed within to measure any change in temperature that occurs to the system. ...
Chapter2 The First Law of Thermodynamics
... Energy can be neither created nor destroyed;it can only change forms 3-1-2 The First Law of Thermodynamics Neither heat nor work can be destroyed;they can only change from one to another, that is: ...
... Energy can be neither created nor destroyed;it can only change forms 3-1-2 The First Law of Thermodynamics Neither heat nor work can be destroyed;they can only change from one to another, that is: ...
Lessons 3 and 4 Thermodynamics
... Most systems, when left, tend towards more disorder (think of your bedroom! This is why heat spreads from hot to cold. Entropy can decrease in a small part of a system ...
... Most systems, when left, tend towards more disorder (think of your bedroom! This is why heat spreads from hot to cold. Entropy can decrease in a small part of a system ...
Heat wave

A heat wave is a prolonged period of excessively hot weather, which may be accompanied by high humidity, especially in oceanic climate countries. While definitions vary, a heat wave is measured relative to the usual weather in the area and relative to normal temperatures for the season. Temperatures that people from a hotter climate consider normal can be termed a heat wave in a cooler area if they are outside the normal climate pattern for that area.The term is applied both to routine weather variations and to extraordinary spells of heat which may occur only once a century. Severe heat waves have caused catastrophic crop failures, thousands of deaths from hyperthermia, and widespread power outages due to increased use of air conditioning. A heat wave is considered extreme weather, and a danger because heat and sunlight may overheat the human body.