state of matter - Mayfield City Schools
... thermal energy enters your hand because the stove is warmer than your hand. When you touch a piece of ice, thermal energy passes out of your hand and into the colder ice. ...
... thermal energy enters your hand because the stove is warmer than your hand. When you touch a piece of ice, thermal energy passes out of your hand and into the colder ice. ...
Energy Worksheet - MICDS Intranet Menu
... A 5.0 kilogram block of ice at -10.0oC is placed in a container of warm water. The entire block of ice is warmed to 0.0oC, and 4.0 kilograms of ice remains unmelted. At this point, how many joules were transferred from the warm water completely? q = mct = 5000gr x 2.09 J/gr oC x (-10oC - 0oC) = - 10 ...
... A 5.0 kilogram block of ice at -10.0oC is placed in a container of warm water. The entire block of ice is warmed to 0.0oC, and 4.0 kilograms of ice remains unmelted. At this point, how many joules were transferred from the warm water completely? q = mct = 5000gr x 2.09 J/gr oC x (-10oC - 0oC) = - 10 ...
Currents experiment
... minutes and observe what happens to the solutions. You should be asking yourself what does this have to ...
... minutes and observe what happens to the solutions. You should be asking yourself what does this have to ...
Thermodynamic functions - Phase Transformations Group
... the specimen. Since CVL = dU/dT , it follows that the lattice specific heat capacity at constant volume can be specified in terms of the Debye temperature and the Debye function (equation 12). The theory does not provide a complete description of the lattice specific heat since TD is found to vary s ...
... the specimen. Since CVL = dU/dT , it follows that the lattice specific heat capacity at constant volume can be specified in terms of the Debye temperature and the Debye function (equation 12). The theory does not provide a complete description of the lattice specific heat since TD is found to vary s ...
Thermodynamics
... Carnot Efficiency Carnot a believed that there was an absolute zero of temperature, from which he figured out that on being cooled to absolute zero, the fluid would give up all its heat energy. Therefore, if it falls only half way to absolute zero from its beginning temperature, it will give up hal ...
... Carnot Efficiency Carnot a believed that there was an absolute zero of temperature, from which he figured out that on being cooled to absolute zero, the fluid would give up all its heat energy. Therefore, if it falls only half way to absolute zero from its beginning temperature, it will give up hal ...
Measurements - WordPress.com
... ◦ Q = heat measured in joules ◦ m = mass of the object in grams ◦ c = specific heat of an object in ◦ ∆T = change in temperature ...
... ◦ Q = heat measured in joules ◦ m = mass of the object in grams ◦ c = specific heat of an object in ◦ ∆T = change in temperature ...
Exercises – Chapter 8
... E.18 The hotter the water, the more thermal energy can be harnessed and the greater the temperature difference between the hot water and the colder surrounding, the more efficient the hurricane is at converting that thermal energy into mechanical work. 19. On a clear sunny day, the ground is heated ...
... E.18 The hotter the water, the more thermal energy can be harnessed and the greater the temperature difference between the hot water and the colder surrounding, the more efficient the hurricane is at converting that thermal energy into mechanical work. 19. On a clear sunny day, the ground is heated ...
Heat Recovery for Commercial Buildings
... The savings were substantial, $110,700 was saved with the ...
... The savings were substantial, $110,700 was saved with the ...
BUOYANCY-DRIVEN TURBULENT CONVECTION IN A BUNDLE
... Buoyant, turbulent convective heat transfer around cylindrical rods arranged in bundles is a technically relevant heat transfer configuration which finds application in steam generators, cooling of reactor core fuel assemblies and heat exchangers in general. Most of the research performed so far con ...
... Buoyant, turbulent convective heat transfer around cylindrical rods arranged in bundles is a technically relevant heat transfer configuration which finds application in steam generators, cooling of reactor core fuel assemblies and heat exchangers in general. Most of the research performed so far con ...
The Earth`s Heat
... important role than conduction. In relatively uncon strained systems, convection occurs by means o f convection cells, in w hi ch a rising fl uid carries heat upward and transfers it to cooler material at the top of the ce ll. As the fluid ri ses, it cools, becomes more dense and sinks to the botto ...
... important role than conduction. In relatively uncon strained systems, convection occurs by means o f convection cells, in w hi ch a rising fl uid carries heat upward and transfers it to cooler material at the top of the ce ll. As the fluid ri ses, it cools, becomes more dense and sinks to the botto ...
Chapter Summary
... a whole satisfies the first law of thermodynamics, as does each of its processes. The change in internal energy for any cycle is always zero, because the system returns to its initial state, and the area of the enclosed region on the P-V diagram is the net work done in the cycle. Entropy and the Sec ...
... a whole satisfies the first law of thermodynamics, as does each of its processes. The change in internal energy for any cycle is always zero, because the system returns to its initial state, and the area of the enclosed region on the P-V diagram is the net work done in the cycle. Entropy and the Sec ...
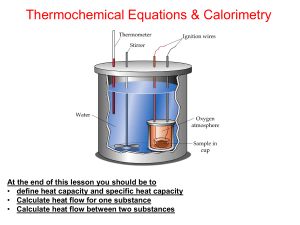
U3 S1 L2 q=mct
... 1. Calculate the heat change involved when 2.00 L of water is heated from 20.0°C to 99.7°C in an electric kettle. 2. Calculate the heat change associated with cooling a 350.0 g aluminum bar from 70.0°C to 25.0°C. Is the change endothermic or exothermic? Why? (Hint: what is the sign of your answer?) ...
... 1. Calculate the heat change involved when 2.00 L of water is heated from 20.0°C to 99.7°C in an electric kettle. 2. Calculate the heat change associated with cooling a 350.0 g aluminum bar from 70.0°C to 25.0°C. Is the change endothermic or exothermic? Why? (Hint: what is the sign of your answer?) ...
Power & Heat
... Vpk in ts seconds while the current starts from Ipk and drops linearly to 0 in the same time. Then the instantaneous power dissipated in the switching device is ...
... Vpk in ts seconds while the current starts from Ipk and drops linearly to 0 in the same time. Then the instantaneous power dissipated in the switching device is ...
Radiant Barrier Training 7-2013 - Fi-Foil
... Heat is conducted through roofing materials and reradiated to the mass insulation and ceiling below. Attic air temperatures climb to superheated levels…typically 140 degrees Fahrenheit in the summer. Radiant heat transfers into air conditioning ducts increasing energy costs. Attic structure and cont ...
... Heat is conducted through roofing materials and reradiated to the mass insulation and ceiling below. Attic air temperatures climb to superheated levels…typically 140 degrees Fahrenheit in the summer. Radiant heat transfers into air conditioning ducts increasing energy costs. Attic structure and cont ...
Part II. Convection Currents and the Mantle
... A. Go to Convection, Conduction and Radiation and Heat Transfer websites. 1. How is heat transferred by conduction? ____________________________________________________ _____________________________________________________________________________________ 2. Explain the process of convection.________ ...
... A. Go to Convection, Conduction and Radiation and Heat Transfer websites. 1. How is heat transferred by conduction? ____________________________________________________ _____________________________________________________________________________________ 2. Explain the process of convection.________ ...
Atmospheric - Penicuik High School
... The Earth’s orbit Shape of the orbit (rugby ball) Angle of tilt to the sun Time of year when earth is closest to the sun ...
... The Earth’s orbit Shape of the orbit (rugby ball) Angle of tilt to the sun Time of year when earth is closest to the sun ...
AGU Fall Meeting 08 - Global Heat Flow Database
... continents, oceanic heat flow remains nearly constant. The change in slope of the oceanic T-z curve is due to the change in thermal conductivity at the base of the crust. Extreme scatter in heat flow in the ocean basin indicates extreme hydrothermal circulation. Diagram 2b shows 4457 heat flow obser ...
... continents, oceanic heat flow remains nearly constant. The change in slope of the oceanic T-z curve is due to the change in thermal conductivity at the base of the crust. Extreme scatter in heat flow in the ocean basin indicates extreme hydrothermal circulation. Diagram 2b shows 4457 heat flow obser ...
Heat wave

A heat wave is a prolonged period of excessively hot weather, which may be accompanied by high humidity, especially in oceanic climate countries. While definitions vary, a heat wave is measured relative to the usual weather in the area and relative to normal temperatures for the season. Temperatures that people from a hotter climate consider normal can be termed a heat wave in a cooler area if they are outside the normal climate pattern for that area.The term is applied both to routine weather variations and to extraordinary spells of heat which may occur only once a century. Severe heat waves have caused catastrophic crop failures, thousands of deaths from hyperthermia, and widespread power outages due to increased use of air conditioning. A heat wave is considered extreme weather, and a danger because heat and sunlight may overheat the human body.























