Mole Concept Balancing - The Gurukul Institute
... 6. The percentage of oxygen in CuSO4. 5H2O is……….. 7. An oxide of nitrogen with molecular mass 92 has 30.4% nitrogen. Then molecular formula is………… 8. ……..grams of HCI are required to prepare 4 litres of 5M HCI in water. 9. 100 mL on 10 M HCI are diluted with distilled water to volume of 2 litres. T ...
... 6. The percentage of oxygen in CuSO4. 5H2O is……….. 7. An oxide of nitrogen with molecular mass 92 has 30.4% nitrogen. Then molecular formula is………… 8. ……..grams of HCI are required to prepare 4 litres of 5M HCI in water. 9. 100 mL on 10 M HCI are diluted with distilled water to volume of 2 litres. T ...
Second exam 2014 with answers
... Last Name: ____________________________________________ First Name: _____________________________________________ Note: There are 10 questions in this exam (check both sides of the sheet). Fill in your answer in the blank space provided immediately following each question. 1/2 point will be subtract ...
... Last Name: ____________________________________________ First Name: _____________________________________________ Note: There are 10 questions in this exam (check both sides of the sheet). Fill in your answer in the blank space provided immediately following each question. 1/2 point will be subtract ...
Solids and Fluids
... • SI units of pressure are the pascal (1 Pa = 1 N/m2) • Although pressure and force are used interchangeably in everyday life, they are quite different in physics – Pressure is a scalar quantity – Fluid pressure acts perpendicular to any surface in the fluid, no matter how the surface is oriented – ...
... • SI units of pressure are the pascal (1 Pa = 1 N/m2) • Although pressure and force are used interchangeably in everyday life, they are quite different in physics – Pressure is a scalar quantity – Fluid pressure acts perpendicular to any surface in the fluid, no matter how the surface is oriented – ...
Dynamic Planet Test
... 26. Which of the following would be first to crystallize as magma cools? a. Amphibole b. Pyroxene c. Olivine d. Quartz 27. What is the primary cause of delamination? a. The instability caused by the difference in density of the lithosphere and asthenosphere b. Accretion due to interplate forces c. ...
... 26. Which of the following would be first to crystallize as magma cools? a. Amphibole b. Pyroxene c. Olivine d. Quartz 27. What is the primary cause of delamination? a. The instability caused by the difference in density of the lithosphere and asthenosphere b. Accretion due to interplate forces c. ...
Dynamic Planet Test 1. Label the plates on the map: (1 point each) A
... 26. Which of the following would be first to crystallize as magma cools? a. Amphibole b. Pyroxene c. Olivine d. Quartz 27. What is the primary cause of delamination? a. The instability caused by the difference in density of the lithosphere and asthenosphere b. Accretion due to interplate forces c. ...
... 26. Which of the following would be first to crystallize as magma cools? a. Amphibole b. Pyroxene c. Olivine d. Quartz 27. What is the primary cause of delamination? a. The instability caused by the difference in density of the lithosphere and asthenosphere b. Accretion due to interplate forces c. ...
CHEM WKST: EQUILIBRIUM / LE CHATELIER`S PRINCIPLE
... 6) For the reaction CH4(g) + H2O(g) ⇄ CO(g) + 3H2(g) + heat tell in which direction the equilibrium will shift for each of the following cases. a) Some H2O(g) is removed. shifts ← b) The temperature is increased. shifts ← c) An inert gas is added. no shift d) The pressure increases. shifts ← e) Some ...
... 6) For the reaction CH4(g) + H2O(g) ⇄ CO(g) + 3H2(g) + heat tell in which direction the equilibrium will shift for each of the following cases. a) Some H2O(g) is removed. shifts ← b) The temperature is increased. shifts ← c) An inert gas is added. no shift d) The pressure increases. shifts ← e) Some ...
110 EXAM IV MATERIAL Tro Spr 2015
... 4. Smelling salts contain ammonium carbonate, which can decompose to form ammonia, a mild heart stimulant. The ammonium carbonate decomposes according to the following reaction: (NH4)2CO3(s) 2NH3(g) + CO2(g) + H2O(l) ...
... 4. Smelling salts contain ammonium carbonate, which can decompose to form ammonia, a mild heart stimulant. The ammonium carbonate decomposes according to the following reaction: (NH4)2CO3(s) 2NH3(g) + CO2(g) + H2O(l) ...
CP Chemistry Final Review – Chap. 10-19
... 2. Describe the significance of the coefficients in a balanced equation (what do they stand for?). 3. Show the process of determining the amount of product made/reactant required given a quantity of reactant. 4. Describe the terms limiting reactant and excess reactant and their significance in chemi ...
... 2. Describe the significance of the coefficients in a balanced equation (what do they stand for?). 3. Show the process of determining the amount of product made/reactant required given a quantity of reactant. 4. Describe the terms limiting reactant and excess reactant and their significance in chemi ...
Instrumental Analysis as Applied to Architectural Materials
... date a building or element, to characterize decay mechanisms, or to evaluate materials for treatment. Most modern techniques have been used for decades. However, recent advancements have allowed for analysis of smaller samples and for quicker and more accurate interpretation. While the equipment and ...
... date a building or element, to characterize decay mechanisms, or to evaluate materials for treatment. Most modern techniques have been used for decades. However, recent advancements have allowed for analysis of smaller samples and for quicker and more accurate interpretation. While the equipment and ...
Scanning Electron Microscopy / Electron Probe X
... constituent chemical elements. From the energy or wavelength and intensity distribution of these X-rays the local chemical composition can be derived not only qualitatively, but also quantitatively. An essential feature of EPMA is the localized excitation of a small area of the sample surface with t ...
... constituent chemical elements. From the energy or wavelength and intensity distribution of these X-rays the local chemical composition can be derived not only qualitatively, but also quantitatively. An essential feature of EPMA is the localized excitation of a small area of the sample surface with t ...
FINAL EXAM REVIEW PROBLEMS
... For each of the following reactions, predict the direction the equilibrium will shift when the volume of the container is increased. a. H2 (g) + F2 (g) 2 HF (g) b. CO (g) + 2 H2 (g) CH3OH (g) c. 2 SO3 (g) 2 SO2 (g) + O2 (g) 57. For the reaction: 2 SO2 (g) + O2 (g) 2 SO3 (g) a. If the reactio ...
... For each of the following reactions, predict the direction the equilibrium will shift when the volume of the container is increased. a. H2 (g) + F2 (g) 2 HF (g) b. CO (g) + 2 H2 (g) CH3OH (g) c. 2 SO3 (g) 2 SO2 (g) + O2 (g) 57. For the reaction: 2 SO2 (g) + O2 (g) 2 SO3 (g) a. If the reactio ...
origin of stylolites in upper permian zechstein anhydrite
... stylolites thus must be attributed to the same time interval. INTRODUCTION Chemical compaction (Lloyd 1977) by pressure solution is a weIl known feature of diagenesis. It is most important in carbonate rocks and sandstones, where it causes reduction of porosity by generation of autochthonous cement ...
... stylolites thus must be attributed to the same time interval. INTRODUCTION Chemical compaction (Lloyd 1977) by pressure solution is a weIl known feature of diagenesis. It is most important in carbonate rocks and sandstones, where it causes reduction of porosity by generation of autochthonous cement ...
Document
... • is lower at high altitudes where the density of air is less. • is higher on a rainy day than on a sunny day. ...
... • is lower at high altitudes where the density of air is less. • is higher on a rainy day than on a sunny day. ...
CHEM 101 Final (Term 141)
... C) The pressure at the triple point for substance A is higher than that of substance B, but the normal boiling and normal melting point for substance A are lower than those of substance B. D) The pressure at the triple point, normal boiling and normal melting point for substance B and for substance ...
... C) The pressure at the triple point for substance A is higher than that of substance B, but the normal boiling and normal melting point for substance A are lower than those of substance B. D) The pressure at the triple point, normal boiling and normal melting point for substance B and for substance ...
Etching of Antimonide-Based Semiconductor Materials
... References: [1] Brain R. Bennett, et al. Antimonide-based compound semiconductors for electronic devices: A review. Solid-State Electronics 49 (2005) pp.1875–1895 [2] Zhang G., et al. Inductively coupled plasma-reactive ion etching of InSb using CH4/H2/Ar plasma. J. Vac. Sci. Technol. A, Vol. 27, No ...
... References: [1] Brain R. Bennett, et al. Antimonide-based compound semiconductors for electronic devices: A review. Solid-State Electronics 49 (2005) pp.1875–1895 [2] Zhang G., et al. Inductively coupled plasma-reactive ion etching of InSb using CH4/H2/Ar plasma. J. Vac. Sci. Technol. A, Vol. 27, No ...
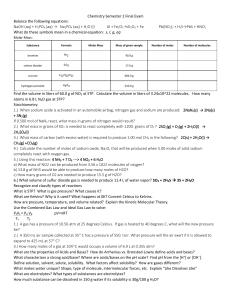
Chemistry Semester 2 Final Exam Chemistry Semester 2 Final Exam
... be? 10.0 atm 2.) A 350 mL air sample collected at 35 C has a pressure of 550. torr. What pressure will the air exert if it is allowed to expand to 425 mL at 57 C? 485 torr 3.) How many moles of a gas at 104 oC would occupy a volume of 6.9 L at 0.355 atm? 0.0791 mol What are the properties of Acids ...
... be? 10.0 atm 2.) A 350 mL air sample collected at 35 C has a pressure of 550. torr. What pressure will the air exert if it is allowed to expand to 425 mL at 57 C? 485 torr 3.) How many moles of a gas at 104 oC would occupy a volume of 6.9 L at 0.355 atm? 0.0791 mol What are the properties of Acids ...
SampleTest3
... ____12. The vapor pressure of a liquid A) decreases with increasing temperature B) is independent of temperature C) is equal to one atmosphere at the normal boiling point D) cannot be measured ____13. Which of the following is not a standard condition (STP) for gas measurements? A) 0OC B) 1.00 atm C ...
... ____12. The vapor pressure of a liquid A) decreases with increasing temperature B) is independent of temperature C) is equal to one atmosphere at the normal boiling point D) cannot be measured ____13. Which of the following is not a standard condition (STP) for gas measurements? A) 0OC B) 1.00 atm C ...
Are You suprised ?
... A system at equilibrium is described by the equation: Heat + SO2Cl2(l ) ⇌ SO2(g) + Cl2(g) One of the following sentences is correct. A) Adding Cl2 will increase heat. B) The equilibrium will move to the left when we remove Cl2. C) Increasing the pressure has no effect on this system. D) Adding catal ...
... A system at equilibrium is described by the equation: Heat + SO2Cl2(l ) ⇌ SO2(g) + Cl2(g) One of the following sentences is correct. A) Adding Cl2 will increase heat. B) The equilibrium will move to the left when we remove Cl2. C) Increasing the pressure has no effect on this system. D) Adding catal ...
Chapter 5 Gases
... moveable piston produces 0.621 mol of a gaseous product. If the cylinder contianed 0.120 mol of gas before the reaction and had an initial volume of 2.18L, what was its volume after reaction? ...
... moveable piston produces 0.621 mol of a gaseous product. If the cylinder contianed 0.120 mol of gas before the reaction and had an initial volume of 2.18L, what was its volume after reaction? ...
Phy 211: General Physics I
... this is a consequence of Pascal’s Principle. At sea level the atmospheric pressure is ...
... this is a consequence of Pascal’s Principle. At sea level the atmospheric pressure is ...
CHM1 Exam 16 Name 2222222222222222222222222222 Multiple
... (1) 2 Na + 2 H2O 2 NaOH + H2 (2) CaCO3 CaO + CO2 (3) LiOH + HCl LiCl + H2O (4) CH4 + 2 O2 CO2 + 2 H2O 15. For the following balanced chemical reaction, what is the sum of the stoichiometric ...
... (1) 2 Na + 2 H2O 2 NaOH + H2 (2) CaCO3 CaO + CO2 (3) LiOH + HCl LiCl + H2O (4) CH4 + 2 O2 CO2 + 2 H2O 15. For the following balanced chemical reaction, what is the sum of the stoichiometric ...
P1.192 Sample holder design for e ective thermal
... Since for the functional materials of solid type breeding blanket a pebble-bed form is mainly adopted instead of a bulk form such as a block or a disk, it should be needed to measure the thermal conductivity of pebble-bed. In this study, the effective thermal conductivity of pebble-bed is measured b ...
... Since for the functional materials of solid type breeding blanket a pebble-bed form is mainly adopted instead of a bulk form such as a block or a disk, it should be needed to measure the thermal conductivity of pebble-bed. In this study, the effective thermal conductivity of pebble-bed is measured b ...
Acids and bases
... Look up Rubidium (RB) on your periodic table to find the atomic number which goes on the bottom. Rubidium’s atomic number is 37. ...
... Look up Rubidium (RB) on your periodic table to find the atomic number which goes on the bottom. Rubidium’s atomic number is 37. ...
CHEMISTRY: Practice Spring Final
... Note: Do not JUST study this practice exam; it does not contain every topic that may appear on your final exam. Be sure to look at your review guide to see a list of topics you are responsible for. Also, this practice test is broken up by topic; your final exam will not be. CHEMICAL REACTIONS 1) Cla ...
... Note: Do not JUST study this practice exam; it does not contain every topic that may appear on your final exam. Be sure to look at your review guide to see a list of topics you are responsible for. Also, this practice test is broken up by topic; your final exam will not be. CHEMICAL REACTIONS 1) Cla ...
Diamond anvil cell
A diamond anvil cell (DAC) is a device used in scientific experiments. It allows compressing a small (sub-millimeter-sized) piece of material to extreme pressures, which can exceed 600 gigapascals (6,000,000 bars / 6 million atmospheres).The device has been used to recreate the pressure existing deep inside planets, creating materials and phases not observed under normal conditions. Notable examples include the non-molecular ice X, polymeric nitrogen and metallic xenon (an inert gas at lower pressures).A DAC consists of two opposing diamonds with a sample compressed between the culets (tips). Pressure may be monitored using a reference material whose behavior under pressure is known. Common pressure standards include ruby fluorescence, and various structurally simple metals, such as copper or platinum. The uniaxial pressure supplied by the DAC may be transformed into uniform hydrostatic pressure using a pressure transmitting medium, such as argon, xenon, hydrogen, helium, paraffin oil or a mixture of methanol and ethanol. The pressure-transmitting medium is enclosed by a gasket and the two diamond anvils. The sample can be viewed through the diamonds and illuminated by X-rays and visible light. In this way, X-ray diffraction and fluorescence; optical absorption and photoluminescence; Mössbauer, Raman and Brillouin scattering; positron annihilation and other signals can be measured from materials under high pressure. Magnetic and microwave fields can be applied externally to the cell allowing nuclear magnetic resonance, electron paramagnetic resonance and other magnetic measurements. Attaching electrodes to the sample allows electrical and magnetoelectrical measurements as well as heating up the sample to a few thousand degrees. Much higher temperatures (up to 7000 K) can be achieved with laser-induced heating, and cooling down to millikelvins has been demonstrated.