Powerpoint Presentation Physical Geology, 10/e
... generally increases with increasing pressure Decompression melting can occur when hot mantle rock moves upward and pressure is reduced enough to drop melting point to the temperature of the rising rock body ...
... generally increases with increasing pressure Decompression melting can occur when hot mantle rock moves upward and pressure is reduced enough to drop melting point to the temperature of the rising rock body ...
Hydraulic Press tonnage calculation Diameter (inches) Pressure
... Hydraulic Press tonnage calculation ...
... Hydraulic Press tonnage calculation ...
LECTURE W3-L7-9 - Partial Melting
... uplift and extensional collapse of orogenically thickened continental crust. Subduction leads to thickened crust by either continental collision (a1) or compression of the continental arc (a2), each with its characteristic orogenic magmatism. Both mechanisms lead to a thickened crust, and probably t ...
... uplift and extensional collapse of orogenically thickened continental crust. Subduction leads to thickened crust by either continental collision (a1) or compression of the continental arc (a2), each with its characteristic orogenic magmatism. Both mechanisms lead to a thickened crust, and probably t ...
Thermal Stability of Mineral-Wool Heat-Insulating
... currents, which correspond to acetylene (m/z = 26), propene (m/z = 42), and butene (m/z = 56). When heated, the binder polymers undergo destruction, with backbone bonds ruptured and low-molecular gaseous and liquid volatile products formed. Despite the complex composition of the volatile products, t ...
... currents, which correspond to acetylene (m/z = 26), propene (m/z = 42), and butene (m/z = 56). When heated, the binder polymers undergo destruction, with backbone bonds ruptured and low-molecular gaseous and liquid volatile products formed. Despite the complex composition of the volatile products, t ...
Melting and Magma Generation
... temperature. At the melting point (and only at that temperature), the solid and liquid phases are in equilibrium. Equilibrium implies dG = 0. Recall that dG = VdP-SdT For a reaction or phase change, such as melting, this equation may be written as d∆G = ∆VdP - ∆SdT where ∆ designates the change in t ...
... temperature. At the melting point (and only at that temperature), the solid and liquid phases are in equilibrium. Equilibrium implies dG = 0. Recall that dG = VdP-SdT For a reaction or phase change, such as melting, this equation may be written as d∆G = ∆VdP - ∆SdT where ∆ designates the change in t ...
Effects of antioxidants for the degradation of flame
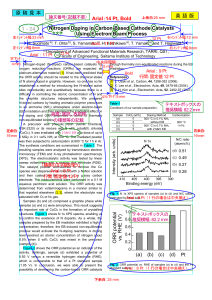
... The synthesis conditions are summarized in Table 1. The resulting samples were analyzed by transmission electron microscopy (TEM) and X-ray photoelectron spectroscopy (XPS). The electrocatalytic activity was tested by linear sweep voltammetry with a rotating disk electrode (RDE). The catalyst powder ...
... The synthesis conditions are summarized in Table 1. The resulting samples were analyzed by transmission electron microscopy (TEM) and X-ray photoelectron spectroscopy (XPS). The electrocatalytic activity was tested by linear sweep voltammetry with a rotating disk electrode (RDE). The catalyst powder ...
CORRELATING THE GEOLOGY OF SHINYANGA DISTRICT
... garnet, ilmenite and spinel. The kimberlites are also poor in heavy minerals known to be indicative of diamond bearing kimberlites. A geochemical study of their composition showed no diamonds were present. Nevertheless in 1931 during mining in Kakamega goldfield, south of Siaya, a few diamonds were ...
... garnet, ilmenite and spinel. The kimberlites are also poor in heavy minerals known to be indicative of diamond bearing kimberlites. A geochemical study of their composition showed no diamonds were present. Nevertheless in 1931 during mining in Kakamega goldfield, south of Siaya, a few diamonds were ...
Atomic Theory (2
... 1.) What are the 5 characteristics of ideal gases? 2.) What is the volume of one mole of any gas at STP? 3.) At what temperature would 2.10 moles of N2 gas have a pressure of 1.25 atm and in a 25.0 L tank? 4.) What volume is occupied by 5.03 g of O2 at 28°C and a pressure of 0.998atm? 5.) What is th ...
... 1.) What are the 5 characteristics of ideal gases? 2.) What is the volume of one mole of any gas at STP? 3.) At what temperature would 2.10 moles of N2 gas have a pressure of 1.25 atm and in a 25.0 L tank? 4.) What volume is occupied by 5.03 g of O2 at 28°C and a pressure of 0.998atm? 5.) What is th ...
2013 us national chemistry olympiad
... d. Nuclear magnetic resonance (NMR) spectroscopy can be used to distinguish between atoms in different environments in a molecule. State and account for the number of unique F-19 signals if 19F NMR were carried out on SF3Cl at i. a low temperature (e.g. –80 °C) ii. room temperature in solution. e. W ...
... d. Nuclear magnetic resonance (NMR) spectroscopy can be used to distinguish between atoms in different environments in a molecule. State and account for the number of unique F-19 signals if 19F NMR were carried out on SF3Cl at i. a low temperature (e.g. –80 °C) ii. room temperature in solution. e. W ...
Chemistry Name: LeChâtlier`s Principle Date: Chemical Equilibrium
... Total pressure is increased (or volume decreased),toward side with less moles of gas Total pressure is decreased (or volume increased),toward side with more moles of gas Increase the temperature, shift away from heat Decrease the temperature, shift toward heat Endothermic = heat is a reactant (+∆H) ...
... Total pressure is increased (or volume decreased),toward side with less moles of gas Total pressure is decreased (or volume increased),toward side with more moles of gas Increase the temperature, shift away from heat Decrease the temperature, shift toward heat Endothermic = heat is a reactant (+∆H) ...
A theoretical model of the explosive fragmentation of vesicular magma
... was improved and modified several times to suit different scientific problems (Spieler et al. 2004; Scheu et al. 2006, 2008a,b), yet the basic principle remains the same: a cylindrical sample (in this study 12 mm radius and 60 mm length) of a porous volcanic rock is glued into a sample holder and plac ...
... was improved and modified several times to suit different scientific problems (Spieler et al. 2004; Scheu et al. 2006, 2008a,b), yet the basic principle remains the same: a cylindrical sample (in this study 12 mm radius and 60 mm length) of a porous volcanic rock is glued into a sample holder and plac ...
File
... solvent because the solution has a lower vapor pressure than the water (Raoult’s Law) . the temperature of the solution has be higher to produce enough vapor pressure to equal the atmospheric pressure (i.e., boiling) (ii) the amount of boiling point elevation depends on the number of non-volatile pa ...
... solvent because the solution has a lower vapor pressure than the water (Raoult’s Law) . the temperature of the solution has be higher to produce enough vapor pressure to equal the atmospheric pressure (i.e., boiling) (ii) the amount of boiling point elevation depends on the number of non-volatile pa ...
PPT
... Kelvin scale of temperature is defined by assigning the number 273.16 to the temperature of a mixture of pure ice, water and water vapor in equilibrium. ...
... Kelvin scale of temperature is defined by assigning the number 273.16 to the temperature of a mixture of pure ice, water and water vapor in equilibrium. ...
WELCOME TO AP CHEMISTRY
... I am very much looking forward to meeting you in September and teaching you second-year Chemistry in preparation for the AP Test in May, 2014. In September we will start out running and then proceed to a gallop as the months go by! The summer assignment is attached. The purpose of the assignment is ...
... I am very much looking forward to meeting you in September and teaching you second-year Chemistry in preparation for the AP Test in May, 2014. In September we will start out running and then proceed to a gallop as the months go by! The summer assignment is attached. The purpose of the assignment is ...
Final Exam Practice Problems: R = 0.0821 Latm/molK NA = 6.022
... 2. A substance that can't be chemically broken down into simpler substances is considered to be A) a homogeneous mixture. B) an element. C) a heterogeneous mixture. D) a compound. E) an electron. 3. Which of the following are examples of a chemical change? A) coffee brewing B) water boiling C) leave ...
... 2. A substance that can't be chemically broken down into simpler substances is considered to be A) a homogeneous mixture. B) an element. C) a heterogeneous mixture. D) a compound. E) an electron. 3. Which of the following are examples of a chemical change? A) coffee brewing B) water boiling C) leave ...
Methane Production from Municipal Solid Waste
... overestimates the biodegradability of paper. Paper products have a very high volatile solids content. Newsprint, office paper, and cardboard have VS of 94%, 96.4%, and 94% respectively (Tchobanoglous, Theisen et al. 1993). Paper products also can have a high content of lignocellulosic components tha ...
... overestimates the biodegradability of paper. Paper products have a very high volatile solids content. Newsprint, office paper, and cardboard have VS of 94%, 96.4%, and 94% respectively (Tchobanoglous, Theisen et al. 1993). Paper products also can have a high content of lignocellulosic components tha ...
2014 Abstract Booklet
... excipient, type of drug polymorph present (amorphous and crystalline forms), and the mechanisms by which the drug elutes from the delivery system are revealed using a variety of methods such as Raman imaging, atomic force microscopy (AFM) and x-ray diffraction. In one example, an ophthalmic device w ...
... excipient, type of drug polymorph present (amorphous and crystalline forms), and the mechanisms by which the drug elutes from the delivery system are revealed using a variety of methods such as Raman imaging, atomic force microscopy (AFM) and x-ray diffraction. In one example, an ophthalmic device w ...
ELAB: One of the Most Potent Amino Acid Analysis
... wide temperature range. Injection using a syringe can also result in discrimination for such substances [1]. On-column injection [2, 3] or "cold"- injection systems can reduce the problem. o Separation of the sample in the chromatographic system: irreversible and reversible adsorptions distort the a ...
... wide temperature range. Injection using a syringe can also result in discrimination for such substances [1]. On-column injection [2, 3] or "cold"- injection systems can reduce the problem. o Separation of the sample in the chromatographic system: irreversible and reversible adsorptions distort the a ...
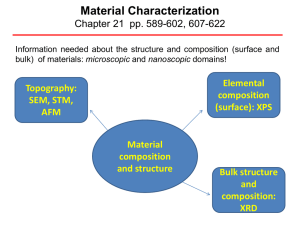
Material Characterization
... X-ray Photoelectron Spectroscopy (XPS) XPS is a surface chemical analysis technique that can be used to analyze the surface chemistry of a material in its "as received" state, or after some treatment XPS detects all elements with an atomic number (Z) of 3 (lithium) and above. It cannot detect hyd ...
... X-ray Photoelectron Spectroscopy (XPS) XPS is a surface chemical analysis technique that can be used to analyze the surface chemistry of a material in its "as received" state, or after some treatment XPS detects all elements with an atomic number (Z) of 3 (lithium) and above. It cannot detect hyd ...
X-ray Diffraction
... laureate in Physics1901), their exact nature was not known. The question posed at the time was "are x-rays particles or are they waves like visible light?" Showing that xrays could diffract would prove that x-rays have a wave-like nature. If x-rays were waves, then their wavelengths had already been ...
... laureate in Physics1901), their exact nature was not known. The question posed at the time was "are x-rays particles or are they waves like visible light?" Showing that xrays could diffract would prove that x-rays have a wave-like nature. If x-rays were waves, then their wavelengths had already been ...
Mass-Mass Stoichiometry
... Chem Regular Second Semester Final Exam Review This review is simply a guide. Anything we’ve covered is considered “fair game” on the final. However, we did try to give example problems from all the topics we’ve studied. So if you run across something during your studying that is not found on this s ...
... Chem Regular Second Semester Final Exam Review This review is simply a guide. Anything we’ve covered is considered “fair game” on the final. However, we did try to give example problems from all the topics we’ve studied. So if you run across something during your studying that is not found on this s ...
Name
... 66. I have a balloon that can hold 100 liters of air. If I blow up this balloon with 3 moles of oxygen at a pressure of 1.00 atm, what is the temperature of the balloon? SHOW YOUR WORK AND LABEL ALL YOUR UNITS! ...
... 66. I have a balloon that can hold 100 liters of air. If I blow up this balloon with 3 moles of oxygen at a pressure of 1.00 atm, what is the temperature of the balloon? SHOW YOUR WORK AND LABEL ALL YOUR UNITS! ...
Practice Exam I solutions
... of nitrate in drinking water samples. Both labs were given a National Institute of Standards and Technology (NIST) standard to analyze and results of 5 measurements by each lab are shown below. Shaky Hands Testing: 3.50, 3.57, 3.38, 3.47, 3.41 ppm High Accuracy, Inc.: 3.23, 3.21, 3.29, 3.30, 3.24 pp ...
... of nitrate in drinking water samples. Both labs were given a National Institute of Standards and Technology (NIST) standard to analyze and results of 5 measurements by each lab are shown below. Shaky Hands Testing: 3.50, 3.57, 3.38, 3.47, 3.41 ppm High Accuracy, Inc.: 3.23, 3.21, 3.29, 3.30, 3.24 pp ...
Metathesis Problems (and Some Solutions) Identified Through
... • Most gases are less soluble in water at higher temperatures (bubbles that appear when heating water) • Most ionic solids are more soluble in water at higher temperatures – Some have very little change, like NaCl – Some are less soluble in higher temperatures • Heat of solution: heat absorbed or re ...
... • Most gases are less soluble in water at higher temperatures (bubbles that appear when heating water) • Most ionic solids are more soluble in water at higher temperatures – Some have very little change, like NaCl – Some are less soluble in higher temperatures • Heat of solution: heat absorbed or re ...
Exam Review
... 6. Sodium metal reacts with chlorine gas to produce sodium chloride. 2 Na(s) + Cl2(g) 2 NaCl a) What mass of sodium is needed to completely react with 15.00 g of chlorine gas? (9.72 g of Na) b) What mass of sodium is required to produce, in excess chlorine, 8.00 g of magnesium chloride? (3.14 g Na ...
... 6. Sodium metal reacts with chlorine gas to produce sodium chloride. 2 Na(s) + Cl2(g) 2 NaCl a) What mass of sodium is needed to completely react with 15.00 g of chlorine gas? (9.72 g of Na) b) What mass of sodium is required to produce, in excess chlorine, 8.00 g of magnesium chloride? (3.14 g Na ...
Diamond anvil cell
A diamond anvil cell (DAC) is a device used in scientific experiments. It allows compressing a small (sub-millimeter-sized) piece of material to extreme pressures, which can exceed 600 gigapascals (6,000,000 bars / 6 million atmospheres).The device has been used to recreate the pressure existing deep inside planets, creating materials and phases not observed under normal conditions. Notable examples include the non-molecular ice X, polymeric nitrogen and metallic xenon (an inert gas at lower pressures).A DAC consists of two opposing diamonds with a sample compressed between the culets (tips). Pressure may be monitored using a reference material whose behavior under pressure is known. Common pressure standards include ruby fluorescence, and various structurally simple metals, such as copper or platinum. The uniaxial pressure supplied by the DAC may be transformed into uniform hydrostatic pressure using a pressure transmitting medium, such as argon, xenon, hydrogen, helium, paraffin oil or a mixture of methanol and ethanol. The pressure-transmitting medium is enclosed by a gasket and the two diamond anvils. The sample can be viewed through the diamonds and illuminated by X-rays and visible light. In this way, X-ray diffraction and fluorescence; optical absorption and photoluminescence; Mössbauer, Raman and Brillouin scattering; positron annihilation and other signals can be measured from materials under high pressure. Magnetic and microwave fields can be applied externally to the cell allowing nuclear magnetic resonance, electron paramagnetic resonance and other magnetic measurements. Attaching electrodes to the sample allows electrical and magnetoelectrical measurements as well as heating up the sample to a few thousand degrees. Much higher temperatures (up to 7000 K) can be achieved with laser-induced heating, and cooling down to millikelvins has been demonstrated.