effect of thermal stresses along crack surface on ultrasonic response
... !!!!!The crack opening was monitored by a video camera during the loading. Figure 13 shows the crack opening measured from the video image as a function of time together with UT amplitude and calculated strains. The crack closes during heating (surface under compressive strains) and opens during coo ...
... !!!!!The crack opening was monitored by a video camera during the loading. Figure 13 shows the crack opening measured from the video image as a function of time together with UT amplitude and calculated strains. The crack closes during heating (surface under compressive strains) and opens during coo ...
CHEMISTRY 102A/102C Spring 2014 Hour Exam II Page _____ For
... I. Equal masses of ideal gases at the same temperature and pressure contain equal numbers of molecules. II. On average, a N2 molecule will possess the same kinetic energy as a CO2 molecule at the same temperature. III. On average, an H2 molecule has a faster average velocity than a N2 molecule at th ...
... I. Equal masses of ideal gases at the same temperature and pressure contain equal numbers of molecules. II. On average, a N2 molecule will possess the same kinetic energy as a CO2 molecule at the same temperature. III. On average, an H2 molecule has a faster average velocity than a N2 molecule at th ...
Sample pages 2 PDF
... Additionally, if we collect experimental data on any reaction at a given temperature (or pressure), we can determine the slope and extrapolate to other conditions, rather than doing more time-consuming experiments. Clapeyron equation helps determining thermodynamic values for reactions or phases. Wh ...
... Additionally, if we collect experimental data on any reaction at a given temperature (or pressure), we can determine the slope and extrapolate to other conditions, rather than doing more time-consuming experiments. Clapeyron equation helps determining thermodynamic values for reactions or phases. Wh ...
How Do Diamonds Form?
... Coal has rarely played a role in the formation of diamonds. In fact, most diamonds that have been dated are much older than Earth's first land plants the source material of coal! That alone should be enough evidence to shut down the idea that Earth's diamond deposits were formed from coal. Another p ...
... Coal has rarely played a role in the formation of diamonds. In fact, most diamonds that have been dated are much older than Earth's first land plants the source material of coal! That alone should be enough evidence to shut down the idea that Earth's diamond deposits were formed from coal. Another p ...
Reactor#4, ALD Films
... 1. Isolate the gas lines in sections by closing the appropriate valves when possible. 2. Pressurize the line and check for bubbling while spraying a few drops of a soapy solution such as "Snoop". 3. If that does not work, spray helium around the suspect connection and look for any pressure change in ...
... 1. Isolate the gas lines in sections by closing the appropriate valves when possible. 2. Pressurize the line and check for bubbling while spraying a few drops of a soapy solution such as "Snoop". 3. If that does not work, spray helium around the suspect connection and look for any pressure change in ...
effect of coupling agent on the interfacial properties of 3d ramie
... remained at a low level. However, thanks to SCA and ethanol treatment much more matrix has been found on the surface of ramie fibers and between fibers. Hence, it is not surprising that the mechanical properties of Sample C show better results than Sample A and B due to better dipersion of matrix. I ...
... remained at a low level. However, thanks to SCA and ethanol treatment much more matrix has been found on the surface of ramie fibers and between fibers. Hence, it is not surprising that the mechanical properties of Sample C show better results than Sample A and B due to better dipersion of matrix. I ...
Mercury in a box – in the Planetary Emissivity Laboratory (PEL) at
... Introduction: Analyzing the surface composition of Mercury's regolith from remote-sensing measurements is a challenging task. In support of the National Aeronautics and Space Agency's MErcury Surface, Space ENvironment, GEochemistry and Ranging (MESSENGER) mission and especially in preparation for t ...
... Introduction: Analyzing the surface composition of Mercury's regolith from remote-sensing measurements is a challenging task. In support of the National Aeronautics and Space Agency's MErcury Surface, Space ENvironment, GEochemistry and Ranging (MESSENGER) mission and especially in preparation for t ...
Gases - Chemistry 504
... 3. Gases don't experience intermolecular forces. The attractive and repulsive forces are negligible (NOT TRUE but they are so small that they can be safely ignored) ...
... 3. Gases don't experience intermolecular forces. The attractive and repulsive forces are negligible (NOT TRUE but they are so small that they can be safely ignored) ...
Origin of Magma
... migrates to the Earth’s surface forming a basaltic lava flow. (Ape Cave at Mt. St. Helens formed as a lava tube in a basaltic lava flow from St. Helens.) The basalt generated in the subduction zone may also melt continental crust. Recall that the melting temperature of the higher silica igneous rock ...
... migrates to the Earth’s surface forming a basaltic lava flow. (Ape Cave at Mt. St. Helens formed as a lava tube in a basaltic lava flow from St. Helens.) The basalt generated in the subduction zone may also melt continental crust. Recall that the melting temperature of the higher silica igneous rock ...
THERMOCHEMISTRY or Thermodynamics
... • Be careful not to confuse these values with STP. Thermochemical standard states of matter – For pure substances in their liquid or solid phase the standard state is the pure liquid or solid. – For gases the standard state is the gas at 1.00 atm of pressure. • For gaseous mixtures the partial press ...
... • Be careful not to confuse these values with STP. Thermochemical standard states of matter – For pure substances in their liquid or solid phase the standard state is the pure liquid or solid. – For gases the standard state is the gas at 1.00 atm of pressure. • For gaseous mixtures the partial press ...
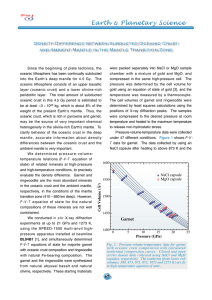
Density Difference between Subducted Oceanic Crust - SPring-8
... layer (oceanic crust) and a lower olivine-rich peridotitic layer. The total amount of subducted oceanic crust in this 4.5 Gy period is estimated to be at least ~3 × 1023 kg, which is about 8% of the weight of the present Earth’s mantle. Thus, the oceanic crust, which is rich in pyroxene and garnet, ...
... layer (oceanic crust) and a lower olivine-rich peridotitic layer. The total amount of subducted oceanic crust in this 4.5 Gy period is estimated to be at least ~3 × 1023 kg, which is about 8% of the weight of the present Earth’s mantle. Thus, the oceanic crust, which is rich in pyroxene and garnet, ...
Thin Film Deposition, Formation of Nanoparticles
... devices and for enabling a variety of investigations of fundamental physical principles. There are many techniques for controllably depositing thin films onto a substrate with thicknesses as small as a few nm. In this paper, we will focus on one deposition technique- thermal evaporation- and produce ...
... devices and for enabling a variety of investigations of fundamental physical principles. There are many techniques for controllably depositing thin films onto a substrate with thicknesses as small as a few nm. In this paper, we will focus on one deposition technique- thermal evaporation- and produce ...
First stop – the CRUST
... temperatures are greatest there, the intense pressure keeps the inner core solid. The outer core, with less pressure, behaves as a liquid due to the intense heat. The mesosphere, or mantle, acts as a highly viscous solid, which means it is capable of flow. Heat from the core rises, causing convectio ...
... temperatures are greatest there, the intense pressure keeps the inner core solid. The outer core, with less pressure, behaves as a liquid due to the intense heat. The mesosphere, or mantle, acts as a highly viscous solid, which means it is capable of flow. Heat from the core rises, causing convectio ...
kinds of metamorphism
... IGNEOUS FLUIDS AND PEGMATITES: The most spectacular hydrothermal metamorphism takes place as an after effect of igneous activity. Magmas have lots of water with dissolved minerals, but as the magma crystallizes the mineral laden water is driven off into the surrounding country rock where it seeps in ...
... IGNEOUS FLUIDS AND PEGMATITES: The most spectacular hydrothermal metamorphism takes place as an after effect of igneous activity. Magmas have lots of water with dissolved minerals, but as the magma crystallizes the mineral laden water is driven off into the surrounding country rock where it seeps in ...
Metamorphism What and Why
... Because atoms at the surface of a substance are in a different environment than those in the interior, there is an excess energy at the surface, called the interfacial free energy. (Surface tension is a manifestation of this energy.) Ratio of surface area to volume decreases with increasing grain si ...
... Because atoms at the surface of a substance are in a different environment than those in the interior, there is an excess energy at the surface, called the interfacial free energy. (Surface tension is a manifestation of this energy.) Ratio of surface area to volume decreases with increasing grain si ...
Bellin College Homework Supplement
... 23.Classify each of the following as a pure substance (element or compound) or a mixture (homogeneous or heterogeneous): a. copper in copper wire b. a chocolate-chip cookie c. nitrox, a combination of oxygen and nitrogen used to fill scuba tanks 24. Classify each of the following as a physical or ch ...
... 23.Classify each of the following as a pure substance (element or compound) or a mixture (homogeneous or heterogeneous): a. copper in copper wire b. a chocolate-chip cookie c. nitrox, a combination of oxygen and nitrogen used to fill scuba tanks 24. Classify each of the following as a physical or ch ...
Print out Reviews # 1 through # 17
... Part 3 – Solubility – Write (s) or (aq) next to each product. Then, write the net ionic equation. 8. ___ Mg(NO3)2 (aq) + ___ Na2O (aq) ___ NaNO3 ( ) + ___ MgO ( ) 9. ___ Zn(ClO3)2 (aq) + ___ K2CO4 (aq) ___ KClO3 ( ) + ___ ZnCO3 ( ) EOC REVIEW #13 1. Given the reaction at equilibrium: 2 N2 (g) + ...
... Part 3 – Solubility – Write (s) or (aq) next to each product. Then, write the net ionic equation. 8. ___ Mg(NO3)2 (aq) + ___ Na2O (aq) ___ NaNO3 ( ) + ___ MgO ( ) 9. ___ Zn(ClO3)2 (aq) + ___ K2CO4 (aq) ___ KClO3 ( ) + ___ ZnCO3 ( ) EOC REVIEW #13 1. Given the reaction at equilibrium: 2 N2 (g) + ...
MM409-coating
... carbide particles which improves cutting performance. • An interesting area of current development is the use of laser glazing, in combination with powder or reactive gasses to make surface compositional changes as well as structural ones. ...
... carbide particles which improves cutting performance. • An interesting area of current development is the use of laser glazing, in combination with powder or reactive gasses to make surface compositional changes as well as structural ones. ...
Gas review
... Kinetic Molecular Theory of Ideal Gases 1. A gas is composed of molecules that are separated from each other by distances far greater than their own dimensions. The molecules can be considered to be points; that is, they possess mass but have negligible volume. 2. Gas molecules are in constant moti ...
... Kinetic Molecular Theory of Ideal Gases 1. A gas is composed of molecules that are separated from each other by distances far greater than their own dimensions. The molecules can be considered to be points; that is, they possess mass but have negligible volume. 2. Gas molecules are in constant moti ...
Rapid Microwave Synthesis, Characterization and Reactivity
... Simultaneous thermal analysis (thermogravimetric and differential thermal analysis; TG-DTA) was performed using a NETZSCH STA 409PC thermobalance coupled to a HIDEN HPR20 mass spectrometer (MS). Approximately 30 mg of Li4NH was placed in an alumina pan and heated from ambient temperature to either 7 ...
... Simultaneous thermal analysis (thermogravimetric and differential thermal analysis; TG-DTA) was performed using a NETZSCH STA 409PC thermobalance coupled to a HIDEN HPR20 mass spectrometer (MS). Approximately 30 mg of Li4NH was placed in an alumina pan and heated from ambient temperature to either 7 ...
Question 1 - JustAnswer
... temperature is raised? Answer The equilibrium position is shifted to the right, and the value for K increases. The equilibrium position is shifted to the right, and the value for K decreases. The equilibrium position is shifted to the left, and the value for K decreases. The equilibrium position is ...
... temperature is raised? Answer The equilibrium position is shifted to the right, and the value for K increases. The equilibrium position is shifted to the right, and the value for K decreases. The equilibrium position is shifted to the left, and the value for K decreases. The equilibrium position is ...
June 2010 Regents Exam Part C Questions
... (2) absorb energy as they move to higher energy states (3) release energy as they move to lower energy states (4) release energy as they move to higher energy states Q8 An atom of which element has the greatest attraction for electrons in a chemical bond? ( note Q8-10 are on Bonding ; see items ...
... (2) absorb energy as they move to higher energy states (3) release energy as they move to lower energy states (4) release energy as they move to higher energy states Q8 An atom of which element has the greatest attraction for electrons in a chemical bond? ( note Q8-10 are on Bonding ; see items ...
ppt
... • A 6-ns long Nd:YAG laser pulse is made to impinge over a f3mm area on a 0.5 mm thick aluminum film that is sandwiched between the back surface of a substrate disk and a 10–20 mm thick layer of SiO2. • The melting-induced expansion of the aluminum layer under confinement generates a compressive str ...
... • A 6-ns long Nd:YAG laser pulse is made to impinge over a f3mm area on a 0.5 mm thick aluminum film that is sandwiched between the back surface of a substrate disk and a 10–20 mm thick layer of SiO2. • The melting-induced expansion of the aluminum layer under confinement generates a compressive str ...
Geology 101 Origin of Magma From our discussions of the structure
... Sometimes this basalt migrates to the Earth’s surface forming a basaltic lava flow. (Ape Cave at Mt. St. Helens formed as a lava tube in a basaltic lava flow from St. Helens.) The basalt generated in the subduction zone may also melt continental crust. Recall that the melting temperature of the high ...
... Sometimes this basalt migrates to the Earth’s surface forming a basaltic lava flow. (Ape Cave at Mt. St. Helens formed as a lava tube in a basaltic lava flow from St. Helens.) The basalt generated in the subduction zone may also melt continental crust. Recall that the melting temperature of the high ...
Carefully detach the last page. It is the Data Sheet.
... The vapour pressure of water is 3.17 kPa at 298 K. ...
... The vapour pressure of water is 3.17 kPa at 298 K. ...
Diamond anvil cell
A diamond anvil cell (DAC) is a device used in scientific experiments. It allows compressing a small (sub-millimeter-sized) piece of material to extreme pressures, which can exceed 600 gigapascals (6,000,000 bars / 6 million atmospheres).The device has been used to recreate the pressure existing deep inside planets, creating materials and phases not observed under normal conditions. Notable examples include the non-molecular ice X, polymeric nitrogen and metallic xenon (an inert gas at lower pressures).A DAC consists of two opposing diamonds with a sample compressed between the culets (tips). Pressure may be monitored using a reference material whose behavior under pressure is known. Common pressure standards include ruby fluorescence, and various structurally simple metals, such as copper or platinum. The uniaxial pressure supplied by the DAC may be transformed into uniform hydrostatic pressure using a pressure transmitting medium, such as argon, xenon, hydrogen, helium, paraffin oil or a mixture of methanol and ethanol. The pressure-transmitting medium is enclosed by a gasket and the two diamond anvils. The sample can be viewed through the diamonds and illuminated by X-rays and visible light. In this way, X-ray diffraction and fluorescence; optical absorption and photoluminescence; Mössbauer, Raman and Brillouin scattering; positron annihilation and other signals can be measured from materials under high pressure. Magnetic and microwave fields can be applied externally to the cell allowing nuclear magnetic resonance, electron paramagnetic resonance and other magnetic measurements. Attaching electrodes to the sample allows electrical and magnetoelectrical measurements as well as heating up the sample to a few thousand degrees. Much higher temperatures (up to 7000 K) can be achieved with laser-induced heating, and cooling down to millikelvins has been demonstrated.