B. Basic Concepts of Metabolism
... fashion in which the atoms of the raw materials are rearranged, often one at a time, until the formation of the final product takes place. Each step requires its own enzyme. The sequence of enzymatically-catalyzed steps from a starting raw material to final end products is called an enzymatic pathwa ...
... fashion in which the atoms of the raw materials are rearranged, often one at a time, until the formation of the final product takes place. Each step requires its own enzyme. The sequence of enzymatically-catalyzed steps from a starting raw material to final end products is called an enzymatic pathwa ...
10/31
... Ammonia is easily incorporated because it is more highly reduced than other forms of nitrogen Can be combined with pyruvate to form alanine or ketoglutarate to form glutamate ...
... Ammonia is easily incorporated because it is more highly reduced than other forms of nitrogen Can be combined with pyruvate to form alanine or ketoglutarate to form glutamate ...
Chapter 3
... The biosphere is the region within which all living things are found on Earth, extending from the bottom of the oceans to the upper atmosphere. The biosphere is but one of the four separate components of the geochemical model along with the lithosphere, hydrosphere, and atmosphere. The Gaia Hypoth ...
... The biosphere is the region within which all living things are found on Earth, extending from the bottom of the oceans to the upper atmosphere. The biosphere is but one of the four separate components of the geochemical model along with the lithosphere, hydrosphere, and atmosphere. The Gaia Hypoth ...
Higher Tier, Unit C2: Chemistry
... (c) Temporary hardness in water can be softened by boiling. All hard water can be softened by ion exchange. Explain how both these methods work by setting out clearly what happens and why the water ends up being soft. ...
... (c) Temporary hardness in water can be softened by boiling. All hard water can be softened by ion exchange. Explain how both these methods work by setting out clearly what happens and why the water ends up being soft. ...
Upon completion of Chapter 7, you should be able to
... Copper (II) nitrate: molar mass 187.6 g/mol Sulfur dioxide: molar mass 64.06 g/mol 1. If you had 12.3 g of copper (II) nitrate, how many moles would you have? ...
... Copper (II) nitrate: molar mass 187.6 g/mol Sulfur dioxide: molar mass 64.06 g/mol 1. If you had 12.3 g of copper (II) nitrate, how many moles would you have? ...
FST 123 - Enzymology Homework IS `13
... 3. The course website contains a link to a Kinemage file depicting the structures of four proteins. Download them, view them using Mage or King (http://kinemage.biochem.duke.edu/software/index.php), and classify them according to Chothia’s four categories. 4. A buffer was made by dissolving 18.92 g ...
... 3. The course website contains a link to a Kinemage file depicting the structures of four proteins. Download them, view them using Mage or King (http://kinemage.biochem.duke.edu/software/index.php), and classify them according to Chothia’s four categories. 4. A buffer was made by dissolving 18.92 g ...
Chapter 14 cycles
... Open ocean Coastal seawater Upwelling area Coral reef Corn field Rice paddy Sugarcane field ...
... Open ocean Coastal seawater Upwelling area Coral reef Corn field Rice paddy Sugarcane field ...
Topic 16 Some non-metals and their compounds notes
... Carbon dioxide is causing the global temperature of the Earth to rise. This rise in temperature affects the climate all over the planet in ways which are hard to predict. Some areas may become wetter or dryer, other areas may become hotter. The main (predictable) directions of wind and ocean current ...
... Carbon dioxide is causing the global temperature of the Earth to rise. This rise in temperature affects the climate all over the planet in ways which are hard to predict. Some areas may become wetter or dryer, other areas may become hotter. The main (predictable) directions of wind and ocean current ...
Strategies for the Allocation of Resources under Sulfur Limitation in
... of Rubisco protein to total protein could facilitate the allocation of reduced sulfur and carbon skeletons to proteins specifically induced by sulfur limitation without an aggravation in sulfur and carbon consumption, as has been suggested for acclimation of wheat leaves to sulfate deprivation (Gilb ...
... of Rubisco protein to total protein could facilitate the allocation of reduced sulfur and carbon skeletons to proteins specifically induced by sulfur limitation without an aggravation in sulfur and carbon consumption, as has been suggested for acclimation of wheat leaves to sulfate deprivation (Gilb ...
Biogeochemical+sulfur+cycle
... o Sulfur is released into atmosphere as sulfur dioxide by volcanic eruptions, bacterial-byproduct sea spray releases, human activity of burning fossil fuels. o The sulfuric acid falls back to earth through rain in terrestrial & aquatic. o Sulfur falls to bottom of ocean and geological activity mixes ...
... o Sulfur is released into atmosphere as sulfur dioxide by volcanic eruptions, bacterial-byproduct sea spray releases, human activity of burning fossil fuels. o The sulfuric acid falls back to earth through rain in terrestrial & aquatic. o Sulfur falls to bottom of ocean and geological activity mixes ...
The Impact of Yeast on Wine Aroma and Flavor: The Good, the Bad
... What factors are associated with appearance of the taint? Always found with a specific vineyard? Associated with unsound fruit? Associated with specific processing? ...
... What factors are associated with appearance of the taint? Always found with a specific vineyard? Associated with unsound fruit? Associated with specific processing? ...
tutorial on carbohydrates
... 12. The polymer chains of glycosaminoglycans are widely spread apart and bind large amount of water. a. What 2 functional groups of the polymer make this binding of water possible? b. What type of binding is involved? 13. In glycoproteins, what are the 3 amino acids to which the carbohydrate groups ...
... 12. The polymer chains of glycosaminoglycans are widely spread apart and bind large amount of water. a. What 2 functional groups of the polymer make this binding of water possible? b. What type of binding is involved? 13. In glycoproteins, what are the 3 amino acids to which the carbohydrate groups ...
Diapositiva 1 - cloudfront.net
... environment with serious impact on human health. It is, nowadays in a small percentage used in the industrial process to produce sulfuric acid. • Olive wastes and orange residues are recalcitrant biomass, pollutant for the environment. ...
... environment with serious impact on human health. It is, nowadays in a small percentage used in the industrial process to produce sulfuric acid. • Olive wastes and orange residues are recalcitrant biomass, pollutant for the environment. ...
Variant 3 - Egypt IG Student Room
... C6H5SO3H. Benzenesulfonic acid is a strong acid which ionises to produce hydrogen ions, H+, and benzenesulfonate ions, C6H5SO3–. ...
... C6H5SO3H. Benzenesulfonic acid is a strong acid which ionises to produce hydrogen ions, H+, and benzenesulfonate ions, C6H5SO3–. ...
Chapter 11 - Geobiology
... Volcanoes release hydrogen sulfide gas. Tectonic processes uplift rocks, and weathering breaks down sulfurbearing minerals. ...
... Volcanoes release hydrogen sulfide gas. Tectonic processes uplift rocks, and weathering breaks down sulfurbearing minerals. ...
weekly schedule and topics
... CHEMICAL ASPECTS OF INDUSTRIAL METALLURTY (ChapterI4)Iron, copper, aluminum, magnesium, gold, and uranium (printed material will be ...
... CHEMICAL ASPECTS OF INDUSTRIAL METALLURTY (ChapterI4)Iron, copper, aluminum, magnesium, gold, and uranium (printed material will be ...
Elements, Compounds and Mixtures.
... similar to those of the substances are different to those of the in a mixture. elements which reacted to form it. There are practically no energy Heat is usually given out or changes when a mixture is made taken in when a compound is formed. It is usually easy to separate the components of a mixture ...
... similar to those of the substances are different to those of the in a mixture. elements which reacted to form it. There are practically no energy Heat is usually given out or changes when a mixture is made taken in when a compound is formed. It is usually easy to separate the components of a mixture ...
2 Unit Biogeochemical Cycling Quiz
... STATE two ways in which nitrogen is made available to life STATE the driving force (energy) that powers the water cycle STATE the two most limiting nutrients of many plants DEFINE transpiration LIST 5 common examples of plants characterized as legumes IDENTIFY organisms that could increase global ox ...
... STATE two ways in which nitrogen is made available to life STATE the driving force (energy) that powers the water cycle STATE the two most limiting nutrients of many plants DEFINE transpiration LIST 5 common examples of plants characterized as legumes IDENTIFY organisms that could increase global ox ...
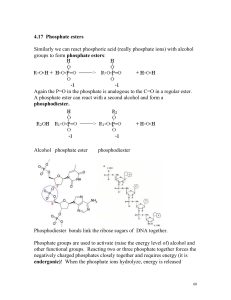
4.17 Phosphate esters Similarly we can react phosphoric acid (really
... Phosphate groups are used to activate (raise the energy level of) alcohol and other functional groups. Reacting two or three phosphate together forces the negatively charged phosphates closely together and requires energy (it is endergonic)! When the phosphate ions hydrolyze, energy is released ...
... Phosphate groups are used to activate (raise the energy level of) alcohol and other functional groups. Reacting two or three phosphate together forces the negatively charged phosphates closely together and requires energy (it is endergonic)! When the phosphate ions hydrolyze, energy is released ...
Sulfur Cycle
... organic matter. As the microorganisms consume the plant matter, they release some of the plant's carbon into the atmosphere in the form of CO2, and some is used in the body. These animals then return more of the carbon to the atmosphere as CO2 through respiration, although some will be stored within ...
... organic matter. As the microorganisms consume the plant matter, they release some of the plant's carbon into the atmosphere in the form of CO2, and some is used in the body. These animals then return more of the carbon to the atmosphere as CO2 through respiration, although some will be stored within ...
Chapter 11: The rise of oxygen and ozone – ppt
... This indicates that no preferential sulfur isotope was being incorporated into rocks. The process was probably photolosys of SO2, which does not occur today because of ozone. This indicates much lower ozone, and therefore oxygen, values prior to 2.3 bya. Also, in a low-oxygen atmosphere, sulfur unde ...
... This indicates that no preferential sulfur isotope was being incorporated into rocks. The process was probably photolosys of SO2, which does not occur today because of ozone. This indicates much lower ozone, and therefore oxygen, values prior to 2.3 bya. Also, in a low-oxygen atmosphere, sulfur unde ...
Sulfur cycle

The sulfur cycle is the collection of processes by which sulfur moves to and from minerals (including the waterways) and living systems. Such biogeochemical cycles are important in geology because they affect many minerals. Biogeochemical cycles are also important for life because sulfur is an essential element, being a constituent of many proteins and cofactors.Steps of the sulfur cycle are: Mineralization of organic sulfur into inorganic forms, such as hydrogen sulfide (H2S), elemental sulfur, as well as sulfide minerals. Oxidation of hydrogen sulfide, sulfide, and elemental sulfur (S) to sulfate (SO42−). Reduction of sulfate to sulfide. Incorporation of sulfide into organic compounds (including metal-containing derivatives).These are often termed as follows:Assimilative sulfate reduction (see also sulfur assimilation) in which sulfate (SO42−) is reduced by plants, fungi and various prokaryotes. The oxidation states of sulfur are +6 in sulfate and –2 in R–SH.Desulfurization in which organic molecules containing sulfur can be desulfurized, producing hydrogen sulfide gas (H2S, oxidation state = –2). An analogous process for organic nitrogen compounds is deamination.Oxidation of hydrogen sulfide produces elemental sulfur (S8), oxidation state = 0. This reaction occurs in the photosynthetic green and purple sulfur bacteria and some chemolithotrophs. Often the elemental sulfur is stored as polysulfides.Oxidation of elemental sulfur by sulfur oxidizers produces sulfate.Dissimilative sulfur reduction in which elemental sulfur can be reduced to hydrogen sulfide.Dissimilative sulfate reduction in which sulfate reducers generate hydrogen sulfide from sulfate.↑