General Concepts and Biogeochemistry
... productivity in soil and marine ecosystems (limitation of energy-yielding organic carbon). ...
... productivity in soil and marine ecosystems (limitation of energy-yielding organic carbon). ...
The Role of Organosulfur Compounds in the Growth of PbS
... = 189.17 g/mol) at the mass-to-charge ratio m/z = 189 Da in both spectra. We used the CHCA molecular ion peak to calibrate the instrument; we performed no post-acquisition corrections to the spectra.A molecular ion peak for ODE (mw = 252.48 g/mol) is not observed, but we do observe two principal fra ...
... = 189.17 g/mol) at the mass-to-charge ratio m/z = 189 Da in both spectra. We used the CHCA molecular ion peak to calibrate the instrument; we performed no post-acquisition corrections to the spectra.A molecular ion peak for ODE (mw = 252.48 g/mol) is not observed, but we do observe two principal fra ...
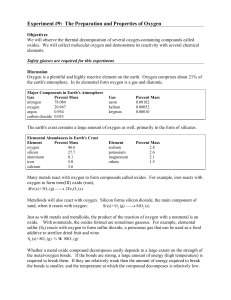
Experiment # 9 Properties of Oxygen
... Experiment #9: The Preparation and Properties of Oxygen Objectives We will observe the thermal decomposition of several oxygen-containing compounds called oxides. We will collect molecular oxygen and demonstrate its reactivity with several chemical elements. Safety glasses are required for this expe ...
... Experiment #9: The Preparation and Properties of Oxygen Objectives We will observe the thermal decomposition of several oxygen-containing compounds called oxides. We will collect molecular oxygen and demonstrate its reactivity with several chemical elements. Safety glasses are required for this expe ...
Oxidation-Reduction Processes in Natural Waters
... Photosynthesis by photoautotrophs is the ultimate origin of nearly all of the organic carbon and energy available in natural ecosystems. This is true even if there are chemoautotrophic bacteria present which grow by oxidizing inorganic chemicals (such as H2S) and assimilating inorganic C as a carbon ...
... Photosynthesis by photoautotrophs is the ultimate origin of nearly all of the organic carbon and energy available in natural ecosystems. This is true even if there are chemoautotrophic bacteria present which grow by oxidizing inorganic chemicals (such as H2S) and assimilating inorganic C as a carbon ...
Proton n.m.r, spectroscopic evidence for sulfur
... bChemiC'JI shift difference between the aromatic signals. ...
... bChemiC'JI shift difference between the aromatic signals. ...
rethinking sulfidization and the role of hydrogen sulfide
... Chesapeake Bay tidal marsh in which sulfidization occurs. The man likely detected the odor of hydrogen sulfide emanating from the marsh. Tidal flushing supplies sulfate, removes bicarbonate and may supply small amounts of oxygen. ...
... Chesapeake Bay tidal marsh in which sulfidization occurs. The man likely detected the odor of hydrogen sulfide emanating from the marsh. Tidal flushing supplies sulfate, removes bicarbonate and may supply small amounts of oxygen. ...
Nomenclature hand out
... Name of the polyatomic ions written as such. For monoatomic ions, i) the metal ion’s name is written as the name of the element. ii) the nonmetal ion is written with part of the nonmetal name with ‘ide’ added to it. ...
... Name of the polyatomic ions written as such. For monoatomic ions, i) the metal ion’s name is written as the name of the element. ii) the nonmetal ion is written with part of the nonmetal name with ‘ide’ added to it. ...
Seasonal changes in Iron species and soluble Iron
... degree of Fe dissolution is influenced by the magnitude of anthropogenic activity, such as fossil fuel combustion. The fitting of XAFS spectra coupled with micro Xray fluorescence analysis (XRF) determined the main Fe species in aerosols in Tsukuba to be illite, ferrihydrite, hornblende, and Fe(III) ...
... degree of Fe dissolution is influenced by the magnitude of anthropogenic activity, such as fossil fuel combustion. The fitting of XAFS spectra coupled with micro Xray fluorescence analysis (XRF) determined the main Fe species in aerosols in Tsukuba to be illite, ferrihydrite, hornblende, and Fe(III) ...
Sulfur Assimilatory Metabolism. The Long and
... environment, sulfate ion in soil and sulfur dioxide gas in the air, is fixed into Cys by the sulfur assimilation pathway in plants (Saito, 2000). Thereafter, Cys is converted to Met. Animals, however, do not have the assimilatory mechanisms for inorganic sulfur; they require Met as an essential amin ...
... environment, sulfate ion in soil and sulfur dioxide gas in the air, is fixed into Cys by the sulfur assimilation pathway in plants (Saito, 2000). Thereafter, Cys is converted to Met. Animals, however, do not have the assimilatory mechanisms for inorganic sulfur; they require Met as an essential amin ...
Sulfur Part II: Sulfur and Sulfur Compounds in the Human Body
... ** T – indicates high level of Thiol content, I – indicates high level of Isothiocyanate content. From the list above the most important source of organosulfur compounds in the human diet is from proteins. The organosulfur compounds found in protein come from the sulfur containing amino acids methio ...
... ** T – indicates high level of Thiol content, I – indicates high level of Isothiocyanate content. From the list above the most important source of organosulfur compounds in the human diet is from proteins. The organosulfur compounds found in protein come from the sulfur containing amino acids methio ...
APES Review Packet 1: Unit 1/Unit 2
... In energy transactions, you can’t break even—conversion always involves a loss of higher-quality energy to lower-quality energy b. You cannot change matter in any fashion—molecules never chemically change. c. Energy flows cannot be prevented. Energy is never really contained, just used and passed on ...
... In energy transactions, you can’t break even—conversion always involves a loss of higher-quality energy to lower-quality energy b. You cannot change matter in any fashion—molecules never chemically change. c. Energy flows cannot be prevented. Energy is never really contained, just used and passed on ...
Vitamins and Minerals
... of body I- is in thyroid T4 (thyroxine) T3 (triiodothyronine)-3 times more active than T4 Controls rate of energy metabolism in cells ...
... of body I- is in thyroid T4 (thyroxine) T3 (triiodothyronine)-3 times more active than T4 Controls rate of energy metabolism in cells ...
Amphetamine sulfate solubility
... practically odorless and produces a mucilaginous sensation on the tongue. Insoluble in alcohol. Optical rotation varies depending on the source of Acacia. Solute pKa, Solvent pH, and Solubility. According to the Henderson-Hasselbach equation, the relationship between pH, pKa, and relative concentrat ...
... practically odorless and produces a mucilaginous sensation on the tongue. Insoluble in alcohol. Optical rotation varies depending on the source of Acacia. Solute pKa, Solvent pH, and Solubility. According to the Henderson-Hasselbach equation, the relationship between pH, pKa, and relative concentrat ...
Chapter 5 Powerpoint Slides
... – First, rainfall scrubs CO2 out of the air, producing carbonic acid (H2CO3), a weak acid. – Next, this solution reacts on contact with silicate rocks to release calcium and other cations and leave behind carbonate and biocarbonate ions dissolved in the water. This solution is washed into the ocea ...
... – First, rainfall scrubs CO2 out of the air, producing carbonic acid (H2CO3), a weak acid. – Next, this solution reacts on contact with silicate rocks to release calcium and other cations and leave behind carbonate and biocarbonate ions dissolved in the water. This solution is washed into the ocea ...
biogeochemical cycles PP
... – First, rainfall scrubs CO2 out of the air, producing carbonic acid (H2CO3), a weak acid. – Next, this solution reacts on contact with silicate rocks to release calcium and other cations and leave behind carbonate and biocarbonate ions dissolved in the water. This solution is washed into the ocea ...
... – First, rainfall scrubs CO2 out of the air, producing carbonic acid (H2CO3), a weak acid. – Next, this solution reacts on contact with silicate rocks to release calcium and other cations and leave behind carbonate and biocarbonate ions dissolved in the water. This solution is washed into the ocea ...
Thiorhodospira sibirica gen. nov., sp. nov., a new alkaliphilic purple
... plant mass in the near-shore area of the soda lake Malyi Kasytui (pH 9 5 , 0 2 % salinity) located in the steppe of the Chita region of south-east Siberia. Single cells were vibrioid- or spiral-shaped ( 3 4 pm wide and 7-20 pm long) and motile by means of a polar tuft of flagella. Internal photosynt ...
... plant mass in the near-shore area of the soda lake Malyi Kasytui (pH 9 5 , 0 2 % salinity) located in the steppe of the Chita region of south-east Siberia. Single cells were vibrioid- or spiral-shaped ( 3 4 pm wide and 7-20 pm long) and motile by means of a polar tuft of flagella. Internal photosynt ...
Diversity of Prokaryotic Organisms
... (25%) deeper water with the less salty (5-12 %) surface water. (Produce H2S) Shown against the green phytoplankton of the surface water. (Use H2S as an electron source for energy production) ...
... (25%) deeper water with the less salty (5-12 %) surface water. (Produce H2S) Shown against the green phytoplankton of the surface water. (Use H2S as an electron source for energy production) ...
Trans-Tonoplast Transport of the Sulfur Containing
... protoplasts. Uptake rates were similar in the light and in the dark and roughly linear with time. In the presence of 1 mM substrate concentrations, GSH was taken up at a rate of 0.11 and 0.15 umol 10? protoplasts-1 h-i in the light and in the dark, respectively. The corresponding rates for GSSG were ...
... protoplasts. Uptake rates were similar in the light and in the dark and roughly linear with time. In the presence of 1 mM substrate concentrations, GSH was taken up at a rate of 0.11 and 0.15 umol 10? protoplasts-1 h-i in the light and in the dark, respectively. The corresponding rates for GSSG were ...
Lecture 8. Biogeochemical Cycles
... emerged because nitrogen was a limiting element for microbial growth. Although molecular nitrogen was abundant in the atmosphere, microbial cells could not directly utilize nitrogen as N2 gas. Cells require organic nitrogen compounds or reduced inorganic forms of nitrogen for growth. Therefore, unde ...
... emerged because nitrogen was a limiting element for microbial growth. Although molecular nitrogen was abundant in the atmosphere, microbial cells could not directly utilize nitrogen as N2 gas. Cells require organic nitrogen compounds or reduced inorganic forms of nitrogen for growth. Therefore, unde ...
8492_Chemichal Weapons Production Indicators
... Phosgene is used as an intermediate in the manufacture of many organic chemicals. The largest amount (approximately 80% of world production) is used to produce toluene diisocyanate and other isocyanates. ...
... Phosgene is used as an intermediate in the manufacture of many organic chemicals. The largest amount (approximately 80% of world production) is used to produce toluene diisocyanate and other isocyanates. ...
Biogeochemical Cycles
... farm waste and discharge of sewage. This stimulates growth of algae. Breakdown of algae by aerobic decomposers depletes water of oxygen. ...
... farm waste and discharge of sewage. This stimulates growth of algae. Breakdown of algae by aerobic decomposers depletes water of oxygen. ...
Ch 11
... Nitrogen such as ammonia and nitrite Important in the breakdown of ammonia containing waste Nitrogen polluted waters become hypoxic As nitrogen is oxidized oxygen is consumed Nitrifiers encompass two metabolically distinctive groups Ammonia oxidizers Nitrite oxidizers ...
... Nitrogen such as ammonia and nitrite Important in the breakdown of ammonia containing waste Nitrogen polluted waters become hypoxic As nitrogen is oxidized oxygen is consumed Nitrifiers encompass two metabolically distinctive groups Ammonia oxidizers Nitrite oxidizers ...
Sulfur cycle

The sulfur cycle is the collection of processes by which sulfur moves to and from minerals (including the waterways) and living systems. Such biogeochemical cycles are important in geology because they affect many minerals. Biogeochemical cycles are also important for life because sulfur is an essential element, being a constituent of many proteins and cofactors.Steps of the sulfur cycle are: Mineralization of organic sulfur into inorganic forms, such as hydrogen sulfide (H2S), elemental sulfur, as well as sulfide minerals. Oxidation of hydrogen sulfide, sulfide, and elemental sulfur (S) to sulfate (SO42−). Reduction of sulfate to sulfide. Incorporation of sulfide into organic compounds (including metal-containing derivatives).These are often termed as follows:Assimilative sulfate reduction (see also sulfur assimilation) in which sulfate (SO42−) is reduced by plants, fungi and various prokaryotes. The oxidation states of sulfur are +6 in sulfate and –2 in R–SH.Desulfurization in which organic molecules containing sulfur can be desulfurized, producing hydrogen sulfide gas (H2S, oxidation state = –2). An analogous process for organic nitrogen compounds is deamination.Oxidation of hydrogen sulfide produces elemental sulfur (S8), oxidation state = 0. This reaction occurs in the photosynthetic green and purple sulfur bacteria and some chemolithotrophs. Often the elemental sulfur is stored as polysulfides.Oxidation of elemental sulfur by sulfur oxidizers produces sulfate.Dissimilative sulfur reduction in which elemental sulfur can be reduced to hydrogen sulfide.Dissimilative sulfate reduction in which sulfate reducers generate hydrogen sulfide from sulfate.↑