chapter 5 ecosystems and the physical environment
... proteins and nucleic acids – when animals consume plant tissues, they assimilate nitrogen by taking in plant nitrogen compounds (amino acids) and converting them to animal compounds (proteins) • d. ammonification – conversion of biological nitrogen compounds into ammonia and ammonium ions – produce ...
... proteins and nucleic acids – when animals consume plant tissues, they assimilate nitrogen by taking in plant nitrogen compounds (amino acids) and converting them to animal compounds (proteins) • d. ammonification – conversion of biological nitrogen compounds into ammonia and ammonium ions – produce ...
PChem Data 7-9 Data Talk Version 2
... • Mn does not bind sulfide (and therefore the protein must bind it with N and O) • It can hold H2O close until energy from light/photons knocks off the protons and oxidizes it to O, which will combine with another nearby O to make oxygen ...
... • Mn does not bind sulfide (and therefore the protein must bind it with N and O) • It can hold H2O close until energy from light/photons knocks off the protons and oxidizes it to O, which will combine with another nearby O to make oxygen ...
Prelab Discussion
... Materials needed per team of two students: The 3 incubated SIM tubes from the last session. ...
... Materials needed per team of two students: The 3 incubated SIM tubes from the last session. ...
Life and Biogeochemical Cycles
... structure and function of proteins. In its fully oxidized state, sulfur exists as sulfate and is the major cause of acidity in both natural and polluted rainwater. This link to acidity makes sulfur important to geochemical, atmospheric, and biological processes such as the natural weathering of rock ...
... structure and function of proteins. In its fully oxidized state, sulfur exists as sulfate and is the major cause of acidity in both natural and polluted rainwater. This link to acidity makes sulfur important to geochemical, atmospheric, and biological processes such as the natural weathering of rock ...
*Coal often contains sulfur as an impurity that is released as
... Btus per metric ton, determine the number of kg of coal needed to produce 1 million Btus of heat. ...
... Btus per metric ton, determine the number of kg of coal needed to produce 1 million Btus of heat. ...
dynamate - Animal Science Products, Inc.
... and magnesium, dietary essentials for animals and poultry. • DYNAMATE is slowly soluble, thereby supplying sulfur, potassium and magnesium at a controlled rate for optimum use by rumen micro-organisms. • DYNAMATE sulfur is utilized by rumen bacteria to produce essential amino acids. High energy rati ...
... and magnesium, dietary essentials for animals and poultry. • DYNAMATE is slowly soluble, thereby supplying sulfur, potassium and magnesium at a controlled rate for optimum use by rumen micro-organisms. • DYNAMATE sulfur is utilized by rumen bacteria to produce essential amino acids. High energy rati ...
Give your alfalfa the sulfur and calcium it needs for top
... GYPSOIL BLENDABLE is calcium sulfate dihydrate (CaSO4 • 2H2O) It is a new pelleted gypsum product from GYPSOIL, the leader in supplying agricultural gypsum sold throughout the upper Midwest, Plains and Southeast. GYPSOIL BLENDABLE is a highly pure material that’s free of contaminants. It blends easi ...
... GYPSOIL BLENDABLE is calcium sulfate dihydrate (CaSO4 • 2H2O) It is a new pelleted gypsum product from GYPSOIL, the leader in supplying agricultural gypsum sold throughout the upper Midwest, Plains and Southeast. GYPSOIL BLENDABLE is a highly pure material that’s free of contaminants. It blends easi ...
HYDROTHERMAL VENT PPT
... • Sulfur bacteria serve as the primary producers. They harvest energy from hydrogen sulfide discharged from cracks in Earth’s crust. This process is called chemosynthesis. • Animals of the vent communities feed on these bacteria. Some animals consume them directly. ...
... • Sulfur bacteria serve as the primary producers. They harvest energy from hydrogen sulfide discharged from cracks in Earth’s crust. This process is called chemosynthesis. • Animals of the vent communities feed on these bacteria. Some animals consume them directly. ...
E. Natural Biochemical Cycles Carbon Cycle The carbon cycle is
... oxidizes to sulfate (SO4). Sulfate can be reduced to hydrogen sulfide by anaerobic respiration (no use of oxygen). Organic sulfur is a main component to proteins. Most of it is found in sedimentary rocks and minerals (Earth’s Crust). Sulfur cycles mainly through decomposition and assimilation. Plant ...
... oxidizes to sulfate (SO4). Sulfate can be reduced to hydrogen sulfide by anaerobic respiration (no use of oxygen). Organic sulfur is a main component to proteins. Most of it is found in sedimentary rocks and minerals (Earth’s Crust). Sulfur cycles mainly through decomposition and assimilation. Plant ...
Role of microorganisms in the cycling of elements
... • Plants obtain their sulfur from sulfur compounds, animals feeding on plant materials sulfur is found mostly as a component of sulfur containing amino acids such • Dead organic matter contains large molecules. • Decomposers excrete digestive enzymes. • Enzymes convert large molecules into small one ...
... • Plants obtain their sulfur from sulfur compounds, animals feeding on plant materials sulfur is found mostly as a component of sulfur containing amino acids such • Dead organic matter contains large molecules. • Decomposers excrete digestive enzymes. • Enzymes convert large molecules into small one ...
Hershey Chase Experiment
... Must employ criteria of the natural world Must not be biased toward either hypothesis (Could support either) ...
... Must employ criteria of the natural world Must not be biased toward either hypothesis (Could support either) ...
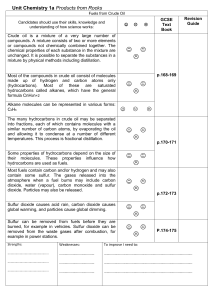
Unit_Chemistry_1a_Oil
... unchanged. It is possible to separate the substances in a mixture by physical methods including distillation. ...
... unchanged. It is possible to separate the substances in a mixture by physical methods including distillation. ...
APES Unit 5: Ecosystem Ecology (Ch. 4) Study Guide
... 35. What is eutrophication? What is the result of eutrophication? accumulation of nutrients , resulting in algal blooms, and then eventually oxygen depletion 36. What is a dead zone? area of water (like Gulf of Mexico at the mouth of the Mississippi) where there is not oxygen and no marine life 37. ...
... 35. What is eutrophication? What is the result of eutrophication? accumulation of nutrients , resulting in algal blooms, and then eventually oxygen depletion 36. What is a dead zone? area of water (like Gulf of Mexico at the mouth of the Mississippi) where there is not oxygen and no marine life 37. ...
Toxicology8 Dr. Ali Ibrahim
... 4. Sulfur:Excessive sulfur intake can result in polioencephalomalacia (PEM) in ruminants. ...
... 4. Sulfur:Excessive sulfur intake can result in polioencephalomalacia (PEM) in ruminants. ...
Carbon Cycle
... help return the Sulfur for the next generation of phosphorus in the soil. 4. Some of the phosphorus in soils can be washed away into water ___________. 5. Another source of phosphorus in water comes from man-made _____________. 6. Too much phosphorus in water leads to plant ________________, strangl ...
... help return the Sulfur for the next generation of phosphorus in the soil. 4. Some of the phosphorus in soils can be washed away into water ___________. 5. Another source of phosphorus in water comes from man-made _____________. 6. Too much phosphorus in water leads to plant ________________, strangl ...
Sulfur - SOIL 5813
... between polypeptide chains; component of other S-containing substances, including S-adenosylmethionine, formylmethionine, lipoic acid, and sulfolipid; about 2% of the organic reduced sulfur in the plant is present in the water soluble thiol (-SH) fraction; vital part of ferredoxin; responsible for t ...
... between polypeptide chains; component of other S-containing substances, including S-adenosylmethionine, formylmethionine, lipoic acid, and sulfolipid; about 2% of the organic reduced sulfur in the plant is present in the water soluble thiol (-SH) fraction; vital part of ferredoxin; responsible for t ...
Quiz 2
... 1. The EPA limit for CO is 9 ppm. Express this number as a percentage. A. 90% B. 9% C. 0.09% D. 0.0009% Percent is parts per hundred. One hundred is 10,000 times less than one million. 2. The burning of coal produces sulfur dioxide, SO2, a pollutant that slowly reacts in air to form SO3. Sulfur trio ...
... 1. The EPA limit for CO is 9 ppm. Express this number as a percentage. A. 90% B. 9% C. 0.09% D. 0.0009% Percent is parts per hundred. One hundred is 10,000 times less than one million. 2. The burning of coal produces sulfur dioxide, SO2, a pollutant that slowly reacts in air to form SO3. Sulfur trio ...
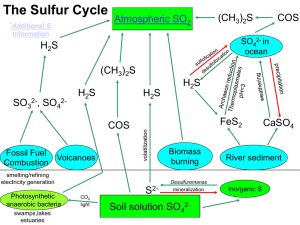
THE SULFUR CYCLE
... pedosphere, large amounts of sulfur, originally meant to maintain in deep soil and sediments, have been released into the environment. There is an increase of oxidized sulfur (SO4) in the global cycle at the expense of the storage of reduced sulfur in the Earth’s crust. ☺ SO2 is released as an air p ...
... pedosphere, large amounts of sulfur, originally meant to maintain in deep soil and sediments, have been released into the environment. There is an increase of oxidized sulfur (SO4) in the global cycle at the expense of the storage of reduced sulfur in the Earth’s crust. ☺ SO2 is released as an air p ...
The Sulfur Cycle
... hormones. It is critical in the environment, climate and the health of ecosystems. Random facts: it can also be referred to as brimstone. it’s the tenth most abundant element in the universe ...
... hormones. It is critical in the environment, climate and the health of ecosystems. Random facts: it can also be referred to as brimstone. it’s the tenth most abundant element in the universe ...
Chemistry Review - Woodlawn School Wiki
... and treated with an excess of aqueous barium chloride, resulting in the precipitation of all the sulfate ions as barium sulfate. The precipitate was collected, dried, and found to weigh 2.33 g. Determine the atomic mass of M, and identify M. (Hints: What is charge on M? Make M a variable in a calcul ...
... and treated with an excess of aqueous barium chloride, resulting in the precipitation of all the sulfate ions as barium sulfate. The precipitate was collected, dried, and found to weigh 2.33 g. Determine the atomic mass of M, and identify M. (Hints: What is charge on M? Make M a variable in a calcul ...
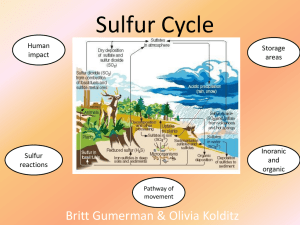
Sulfur cycle

The sulfur cycle is the collection of processes by which sulfur moves to and from minerals (including the waterways) and living systems. Such biogeochemical cycles are important in geology because they affect many minerals. Biogeochemical cycles are also important for life because sulfur is an essential element, being a constituent of many proteins and cofactors.Steps of the sulfur cycle are: Mineralization of organic sulfur into inorganic forms, such as hydrogen sulfide (H2S), elemental sulfur, as well as sulfide minerals. Oxidation of hydrogen sulfide, sulfide, and elemental sulfur (S) to sulfate (SO42−). Reduction of sulfate to sulfide. Incorporation of sulfide into organic compounds (including metal-containing derivatives).These are often termed as follows:Assimilative sulfate reduction (see also sulfur assimilation) in which sulfate (SO42−) is reduced by plants, fungi and various prokaryotes. The oxidation states of sulfur are +6 in sulfate and –2 in R–SH.Desulfurization in which organic molecules containing sulfur can be desulfurized, producing hydrogen sulfide gas (H2S, oxidation state = –2). An analogous process for organic nitrogen compounds is deamination.Oxidation of hydrogen sulfide produces elemental sulfur (S8), oxidation state = 0. This reaction occurs in the photosynthetic green and purple sulfur bacteria and some chemolithotrophs. Often the elemental sulfur is stored as polysulfides.Oxidation of elemental sulfur by sulfur oxidizers produces sulfate.Dissimilative sulfur reduction in which elemental sulfur can be reduced to hydrogen sulfide.Dissimilative sulfate reduction in which sulfate reducers generate hydrogen sulfide from sulfate.↑