Amino Acid Metabolism - Breakdown Other metabolic
... lots protein in diet = carbon skeletons used for fuel, lots of urea starvation = breakdown muscle protein for energy, lots of urea All enzymes (CPS-I and 4 in cycle) synthesized at higher rates in starving animals and animals on high protein diets 2. Carbomyl phosphate synthetase I allosterically ac ...
... lots protein in diet = carbon skeletons used for fuel, lots of urea starvation = breakdown muscle protein for energy, lots of urea All enzymes (CPS-I and 4 in cycle) synthesized at higher rates in starving animals and animals on high protein diets 2. Carbomyl phosphate synthetase I allosterically ac ...
Organic Molecules: The Molecules of Life
... fatty tissue also covers our organs and protects them from injury lipids do not dissolve in water It is possible for us to digest fats because we emulsify them. Bile from our gall bladder is secreted into our digestive tract and causes fat droplets to be broken down into small drops of fat which mak ...
... fatty tissue also covers our organs and protects them from injury lipids do not dissolve in water It is possible for us to digest fats because we emulsify them. Bile from our gall bladder is secreted into our digestive tract and causes fat droplets to be broken down into small drops of fat which mak ...
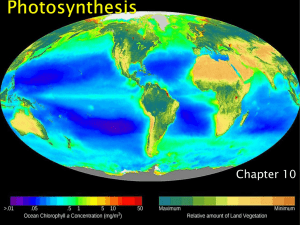
Photosynthesis - Lake Stevens School District
... Reactions are organized into two photosystems (protein complexes that absorb energy from sunlight) ◦ photosystem II and photosystem I ...
... Reactions are organized into two photosystems (protein complexes that absorb energy from sunlight) ◦ photosystem II and photosystem I ...
Unit 2 Review Sheet - Discover more about NYLearns.org
... What is the equation for photosynthesis? What ORGANELLE does photosynthesis take place in? ___________________________________________________________________________________________ What is the equation for cellular respiration? What ORGANELLE does cellular respiration take place in? ______________ ...
... What is the equation for photosynthesis? What ORGANELLE does photosynthesis take place in? ___________________________________________________________________________________________ What is the equation for cellular respiration? What ORGANELLE does cellular respiration take place in? ______________ ...
Carbon and Macromolecules
... made of three fatty acid chains bonded to a glycerol molecule. • When a carboxylic acid and an alcohol react, a water molecule is removed, and an ester linkage is formed • Triglycerides make “the fat” of our bodies. In animals, they are stored as droplets in fat cells or adipocytes. ...
... made of three fatty acid chains bonded to a glycerol molecule. • When a carboxylic acid and an alcohol react, a water molecule is removed, and an ester linkage is formed • Triglycerides make “the fat” of our bodies. In animals, they are stored as droplets in fat cells or adipocytes. ...
Review Guide for Third Exam in Biochemistry 507 (1997)
... 3. The reaction catalyzed by glutamate dehydrogenase 4. The reactions in the synthesis of carbamoyl phosphate 5. The sequence of steps, and the names (not structures) of the intermediates of the urea cycle 6. The reaction catalyzed by arginase (structures) 7. Role of the urea cycle in mammals Lectur ...
... 3. The reaction catalyzed by glutamate dehydrogenase 4. The reactions in the synthesis of carbamoyl phosphate 5. The sequence of steps, and the names (not structures) of the intermediates of the urea cycle 6. The reaction catalyzed by arginase (structures) 7. Role of the urea cycle in mammals Lectur ...
L01_2002
... 1. Carbohydrates (sugars) 2. Lipids (fats) 3. Amino acids (proteins) 4. Nucleic acids (DNA and RNA—not used for major nutritional purposes in humans, but are metabolized for energy purposes by some animals such as ruminants) ...
... 1. Carbohydrates (sugars) 2. Lipids (fats) 3. Amino acids (proteins) 4. Nucleic acids (DNA and RNA—not used for major nutritional purposes in humans, but are metabolized for energy purposes by some animals such as ruminants) ...
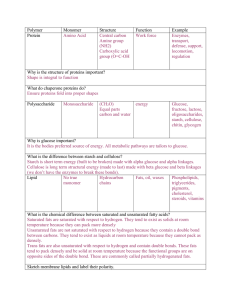
Biochemistry: Monomers and Polymers
... – Our bodies are able to make 12 of the 20 amino acids, the rest come from what you eat. – The amino acid monomers are linked together by peptide bonds to form protein polymers. ...
... – Our bodies are able to make 12 of the 20 amino acids, the rest come from what you eat. – The amino acid monomers are linked together by peptide bonds to form protein polymers. ...
Jordan University of Science and Technology Faculty of Medicine
... 6. Lipids and Proteins are Associated in Biological Membranes (Chapter 8) a. What is the definition of a lipid? b. What are the chemical natures of the lipid types? c. What is the nature of biological membranes? d. What are some common types of membrane proteins? e. What is the Fluid-Mosaic model of ...
... 6. Lipids and Proteins are Associated in Biological Membranes (Chapter 8) a. What is the definition of a lipid? b. What are the chemical natures of the lipid types? c. What is the nature of biological membranes? d. What are some common types of membrane proteins? e. What is the Fluid-Mosaic model of ...
123 - Jordan University of Science and Technology
... 6. Lipids and Proteins are Associated in Biological Membranes (Chapter 8) a. What is the definition of a lipid? b. What are the chemical natures of the lipid types? c. What is the nature of biological membranes? d. What are some common types of membrane proteins? e. What is the Fluid-Mosaic model of ...
... 6. Lipids and Proteins are Associated in Biological Membranes (Chapter 8) a. What is the definition of a lipid? b. What are the chemical natures of the lipid types? c. What is the nature of biological membranes? d. What are some common types of membrane proteins? e. What is the Fluid-Mosaic model of ...
123 biochemistry - Jordan University of Science and Technology
... 6. Lipids and Proteins are Associated in Biological Membranes (Chapter 8) a. What is the definition of a lipid? b. What are the chemical natures of the lipid types? c. What is the nature of biological membranes? d. What are some common types of membrane proteins? e. What is the Fluid-Mosaic model of ...
... 6. Lipids and Proteins are Associated in Biological Membranes (Chapter 8) a. What is the definition of a lipid? b. What are the chemical natures of the lipid types? c. What is the nature of biological membranes? d. What are some common types of membrane proteins? e. What is the Fluid-Mosaic model of ...
File
... – rained down on _________________ from outer space; – were synthesized at ___________________ vents on the ocean floor. Chemical evolution • The formation (synthesis) of complex __________________ molecules from simpler inorganic molecules through chemical reactions in the oceans during the early h ...
... – rained down on _________________ from outer space; – were synthesized at ___________________ vents on the ocean floor. Chemical evolution • The formation (synthesis) of complex __________________ molecules from simpler inorganic molecules through chemical reactions in the oceans during the early h ...
Citric Acid Cycle 2
... What is the thermodynamic driving force for formation of citrate? High levels of oxaloacetate C-C bond formation through condensation Loss of carbon dioxide Hydrolysis of a high energy bond ...
... What is the thermodynamic driving force for formation of citrate? High levels of oxaloacetate C-C bond formation through condensation Loss of carbon dioxide Hydrolysis of a high energy bond ...
Carbohydrate Metabolism Updated
... • the final common pathway for the oxidation of carbohydrates, lipids, and proteins are all metabolized to acetyl-CoA or intermediates of the cycle. • an amphibolic process.Citric acid cycle has a dual function, it (catabolism and anabolism). pathways originate from the cycle: • Gluconeogenesis:All ...
... • the final common pathway for the oxidation of carbohydrates, lipids, and proteins are all metabolized to acetyl-CoA or intermediates of the cycle. • an amphibolic process.Citric acid cycle has a dual function, it (catabolism and anabolism). pathways originate from the cycle: • Gluconeogenesis:All ...
Microbial Metabolism Lipids and Proteins - ASAB-NUST
... • Some bacteria and fungi particularly pathogenic, food spoilage, and soil microorganisms can use proteins as their source of carbon and energy. • They secrete protease enzymes that hydrolyze proteins and polypeptides to amino acids, which are transported into the cell and catabolized ...
... • Some bacteria and fungi particularly pathogenic, food spoilage, and soil microorganisms can use proteins as their source of carbon and energy. • They secrete protease enzymes that hydrolyze proteins and polypeptides to amino acids, which are transported into the cell and catabolized ...
babkapres
... • If a peptide bond is formed, water is released • In a reverse reaction, the peptide bond can hence be cleaved by water (hydrolysis) ...
... • If a peptide bond is formed, water is released • In a reverse reaction, the peptide bond can hence be cleaved by water (hydrolysis) ...
(Semester VI) Paper 15: PLANT METABOLISM THEORY Unit 1
... Unit 4: Carbon Oxidation Glycolysis, fate of pyruvate, regulation of glycolysis, oxidative pentose phosphate pathway, oxidative decarboxylation of pyruvate, regulation of PDH, NADH shuttle; TCA cycle, amphibolic role, anaplerotic reactions, regulation of the cycle, mitochondrial electron transport, ...
... Unit 4: Carbon Oxidation Glycolysis, fate of pyruvate, regulation of glycolysis, oxidative pentose phosphate pathway, oxidative decarboxylation of pyruvate, regulation of PDH, NADH shuttle; TCA cycle, amphibolic role, anaplerotic reactions, regulation of the cycle, mitochondrial electron transport, ...
Quiz Next Tuesday (09/18) - Chemistry at Winthrop University
... The Coplanar Nature of the Peptide Bond Six atoms of the peptide group lie in a plane! ...
... The Coplanar Nature of the Peptide Bond Six atoms of the peptide group lie in a plane! ...
BIOMOLECULES. I. (up to proteins) Basic Molecules of Terrestrial
... basic problem in understanding the origin of life. They include: carbohydrates (used for food and structural materials) fats (store and transport energy) lipids (e.g. cell membranes) – these have a crucial “amphiphilic/amphiphobic” property due to their structure and that of water. These are importa ...
... basic problem in understanding the origin of life. They include: carbohydrates (used for food and structural materials) fats (store and transport energy) lipids (e.g. cell membranes) – these have a crucial “amphiphilic/amphiphobic” property due to their structure and that of water. These are importa ...
optional activity key File
... Trans fats are also unsaturated with respect to hydrogen and contain double bonds. These fats tend to pack densely and be solid at room temperature because the functional groups are on opposite sides of the double bond. These are commonly called partially hydrogenated fats. Sketch membrane lipids an ...
... Trans fats are also unsaturated with respect to hydrogen and contain double bonds. These fats tend to pack densely and be solid at room temperature because the functional groups are on opposite sides of the double bond. These are commonly called partially hydrogenated fats. Sketch membrane lipids an ...
Anatomy and Physiology Chapter #4
... Energy can be mechanical, chemical, and thermal. The function of ATP is to store energy in its terminal phosphate bond. ...
... Energy can be mechanical, chemical, and thermal. The function of ATP is to store energy in its terminal phosphate bond. ...
3.2 Proteins - Biology with Radjewski
... of 20 or fewer amino acids (some hormones and signaling molecules) • Polypeptides or proteins range in size from insulin, which has 51 amino acids, to huge molecules such as the muscle protein titin, with 34,350 amino acids. ...
... of 20 or fewer amino acids (some hormones and signaling molecules) • Polypeptides or proteins range in size from insulin, which has 51 amino acids, to huge molecules such as the muscle protein titin, with 34,350 amino acids. ...
Metabolism

Metabolism (from Greek: μεταβολή metabolē, ""change"") is the set of life-sustaining chemical transformations within the cells of living organisms. These enzyme-catalyzed reactions allow organisms to grow and reproduce, maintain their structures, and respond to their environments. The word metabolism can also refer to all chemical reactions that occur in living organisms, including digestion and the transport of substances into and between different cells, in which case the set of reactions within the cells is called intermediary metabolism or intermediate metabolism.Metabolism is usually divided into two categories: catabolism, the breaking down of organic matter by way of cellular respiration, and anabolism, the building up of components of cells such as proteins and nucleic acids. Usually, breaking down releases energy and building up consumes energy.The chemical reactions of metabolism are organized into metabolic pathways, in which one chemical is transformed through a series of steps into another chemical, by a sequence of enzymes. Enzymes are crucial to metabolism because they allow organisms to drive desirable reactions that require energy that will not occur by themselves, by coupling them to spontaneous reactions that release energy. Enzymes act as catalysts that allow the reactions to proceed more rapidly. Enzymes also allow the regulation of metabolic pathways in response to changes in the cell's environment or to signals from other cells.The metabolic system of a particular organism determines which substances it will find nutritious and which poisonous. For example, some prokaryotes use hydrogen sulfide as a nutrient, yet this gas is poisonous to animals. The speed of metabolism, the metabolic rate, influences how much food an organism will require, and also affects how it is able to obtain that food.A striking feature of metabolism is the similarity of the basic metabolic pathways and components between even vastly different species. For example, the set of carboxylic acids that are best known as the intermediates in the citric acid cycle are present in all known organisms, being found in species as diverse as the unicellular bacterium Escherichia coli and huge multicellular organisms like elephants. These striking similarities in metabolic pathways are likely due to their early appearance in evolutionary history, and their retention because of their efficacy.