video slide - Blue Valley Schools
... Enzymes that digest starch by hydrolyzing alpha linkages can’t hydrolyze beta linkages in cellulose. Cellulose in human food passes through the digestive tract as insoluble fiber. Some microbes use enzymes to digest cellulose. Many herbivores, from cows to termites, have symbiotic relationshi ...
... Enzymes that digest starch by hydrolyzing alpha linkages can’t hydrolyze beta linkages in cellulose. Cellulose in human food passes through the digestive tract as insoluble fiber. Some microbes use enzymes to digest cellulose. Many herbivores, from cows to termites, have symbiotic relationshi ...
Table S2. Functional classification of differentially expressed genes
... Transport of small molecules ...
... Transport of small molecules ...
Delivery of Nutrients to Cells
... required glucagon produced in the pancreas converts glycogen back to glucose. Lipid: Cholesterol required for cell membrane structure and synthesis of steroids. Liver synthesises cholesterol if not enough obtained through dietary intake. Excess cholesterol excreted by liver and passed out of body as ...
... required glucagon produced in the pancreas converts glycogen back to glucose. Lipid: Cholesterol required for cell membrane structure and synthesis of steroids. Liver synthesises cholesterol if not enough obtained through dietary intake. Excess cholesterol excreted by liver and passed out of body as ...
Macromolecule PowerPoint
... Main energy transport molecule & source for metabolism All digested carbohydrates broken down to this for absorption in the small intestine. Only fuel used by the brain/nervous tissue ...
... Main energy transport molecule & source for metabolism All digested carbohydrates broken down to this for absorption in the small intestine. Only fuel used by the brain/nervous tissue ...
BIO 220 Chapter 5 lecture outline Metabolism definition Collision
... 14. Compare and contrast substrate-level phosphorylation and oxidative phosphorylation. What is chemiosmosis? 15. Describe the similarities and differences between aerobic and anaerobic metabolism. 16. What does ATP stand for? Describe its general structure. 17. What are the steps of aerobic cellula ...
... 14. Compare and contrast substrate-level phosphorylation and oxidative phosphorylation. What is chemiosmosis? 15. Describe the similarities and differences between aerobic and anaerobic metabolism. 16. What does ATP stand for? Describe its general structure. 17. What are the steps of aerobic cellula ...
Bio 210 Cell Chemistry Lecture 8 “Glycolysis”
... these molecules involved, we can be fairly certain that oxidation and reduction reactions are going on. The name of one essential carrier is NAD+: nicotinamide adenine dinucleotide. The molecule consists of two nucleotides joined together. Fig. 9.4. Oxidized NAD+ can carry two electrons and a proto ...
... these molecules involved, we can be fairly certain that oxidation and reduction reactions are going on. The name of one essential carrier is NAD+: nicotinamide adenine dinucleotide. The molecule consists of two nucleotides joined together. Fig. 9.4. Oxidized NAD+ can carry two electrons and a proto ...
BIOCHEMISTRY NOTES
... a. An increase in temperature (to a point) INCREASES the rate of an enzymecatalyzed reaction b. At a certain temperature (which is different for each enzyme, but is usually 100 degrees C) the temperature gets so high that the enzyme is moving so fast that its H bonds are broken. 1) The enzyme loses ...
... a. An increase in temperature (to a point) INCREASES the rate of an enzymecatalyzed reaction b. At a certain temperature (which is different for each enzyme, but is usually 100 degrees C) the temperature gets so high that the enzyme is moving so fast that its H bonds are broken. 1) The enzyme loses ...
Human Physiology Quiz Questions: 1) Purines degrade into what
... 4) What is ‘de novo synthesis’ of nucleic acids and where does it take place? 5) What is the difference between a nucleoside and a nucleotide? 6) What is the ‘salvage pathway’ for nucleic acids? 7) What is the primary enzyme that catalyzes glycogenesis? 8) What two membrane transporters absorb monos ...
... 4) What is ‘de novo synthesis’ of nucleic acids and where does it take place? 5) What is the difference between a nucleoside and a nucleotide? 6) What is the ‘salvage pathway’ for nucleic acids? 7) What is the primary enzyme that catalyzes glycogenesis? 8) What two membrane transporters absorb monos ...
Cell Processes and Energy Study Guide The products of
... 21. The raw materials for respiration are the ___________________ of the end products for photosynthesis. opposite 22. In ___________ transport, materials move from a higher concentration to a lower concentration through the cell membrane. passive 23. Carbon dioxide enters plants through the _______ ...
... 21. The raw materials for respiration are the ___________________ of the end products for photosynthesis. opposite 22. In ___________ transport, materials move from a higher concentration to a lower concentration through the cell membrane. passive 23. Carbon dioxide enters plants through the _______ ...
Cellular_Respiration_overviewap
... Electron Transport Chain: Along the inner membrane of the mitochondria The final step of aerobic cellular respiration is called the electron transport chain (ETC). The ETC works with the 10 NADH’s and 2 FADH2’s produced from glycolysis and the Krebs cycle. The electrons stored by NADH and FADH2 get ...
... Electron Transport Chain: Along the inner membrane of the mitochondria The final step of aerobic cellular respiration is called the electron transport chain (ETC). The ETC works with the 10 NADH’s and 2 FADH2’s produced from glycolysis and the Krebs cycle. The electrons stored by NADH and FADH2 get ...
Revised Chapter 4 and 5
... • Genetic material that stores information for its own replication and for the sequence of amino acids in proteins. ...
... • Genetic material that stores information for its own replication and for the sequence of amino acids in proteins. ...
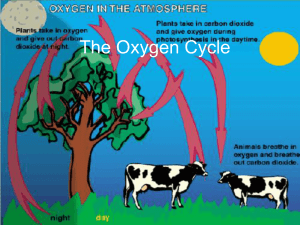
The Oxygen Cycle
... microorganisms in the pond begin to break down organic matter, consuming oxygen in the process. ...
... microorganisms in the pond begin to break down organic matter, consuming oxygen in the process. ...
1. Metabolic pathways 2. Basic enzyme kinetics 3. Metabolic
... » Electrons are transported from NADH & FADH through the electron transport chain to oxygen » Electron transport causes protons to be released into the intermembrane space » These electrons can be transported back into mitochondrial matrix by a proton conducting ATP-synthase » The detailed mechanist ...
... » Electrons are transported from NADH & FADH through the electron transport chain to oxygen » Electron transport causes protons to be released into the intermembrane space » These electrons can be transported back into mitochondrial matrix by a proton conducting ATP-synthase » The detailed mechanist ...
Abiogenesis – Students should know basic problems a successful
... machine to perform many necessary functions. Each of these proteins is made up of a long chain of amino acids ordered in a very specific sequence. A typical chain length is 400 specific amino acids. To have a specific sequence of amino acids occur by random processes is difficult to justify so let’s ...
... machine to perform many necessary functions. Each of these proteins is made up of a long chain of amino acids ordered in a very specific sequence. A typical chain length is 400 specific amino acids. To have a specific sequence of amino acids occur by random processes is difficult to justify so let’s ...
Document
... Prokaryotic and eukaryotic organisms alive today all have structures based on the cell. Cell structure must have originated from chemicals present on early Earth. For this to happen four essential steps are required. ...
... Prokaryotic and eukaryotic organisms alive today all have structures based on the cell. Cell structure must have originated from chemicals present on early Earth. For this to happen four essential steps are required. ...
Learning Objectives
... You should be able to recognize (in a picture), the nucleus, the cytoplasm, and the mitochondria in a eukaryotic cell. You should also know the main functions of these organelles Why is ATP called the energy currency of the cell? Why does hydrolyzing ATP release so much energy? What are the three ma ...
... You should be able to recognize (in a picture), the nucleus, the cytoplasm, and the mitochondria in a eukaryotic cell. You should also know the main functions of these organelles Why is ATP called the energy currency of the cell? Why does hydrolyzing ATP release so much energy? What are the three ma ...
1. Introduction
... constricted at these two steps on the basis of the limited availability of these two substrates. Replenishment of compounds necessary to drive the citric acid cycle, such as oxaloacetate and αketoglutarate, are: fixation of CO2 to pyruvate by the actions of the enzymes pyruvate carboxylase ATPdepe ...
... constricted at these two steps on the basis of the limited availability of these two substrates. Replenishment of compounds necessary to drive the citric acid cycle, such as oxaloacetate and αketoglutarate, are: fixation of CO2 to pyruvate by the actions of the enzymes pyruvate carboxylase ATPdepe ...
Pantesin HF55
... Pantesin is a high-quality pharmaceutical grade branded form of Pantethine that is a biological active form of Vitamin B5. Pantethine forms the reactive component of Coenzyme A (CoA) and the acyl-carrier protein (ACP). CoA and ACP are extensively involved in carbohydrate, lipid and amino acid metabo ...
... Pantesin is a high-quality pharmaceutical grade branded form of Pantethine that is a biological active form of Vitamin B5. Pantethine forms the reactive component of Coenzyme A (CoA) and the acyl-carrier protein (ACP). CoA and ACP are extensively involved in carbohydrate, lipid and amino acid metabo ...
Biochemistry Review Reteach
... (a.) calcium (b.) phosphorus (c.) nitrogen (d.) oxygen 10. Structurally lipids are a very diverse group but they are all placed in one group because of what property? (a.) They are composed of glycerol and fatty acids. (b.) They are all relatively insoluble in water. (c.) They all contain four inter ...
... (a.) calcium (b.) phosphorus (c.) nitrogen (d.) oxygen 10. Structurally lipids are a very diverse group but they are all placed in one group because of what property? (a.) They are composed of glycerol and fatty acids. (b.) They are all relatively insoluble in water. (c.) They all contain four inter ...
Chapter 2b
... • Increasing [OH] increases alkalinity. • Most organisms grow best between pH 6.5 and 8.5. ...
... • Increasing [OH] increases alkalinity. • Most organisms grow best between pH 6.5 and 8.5. ...
1199734spontaneous generation-km
... but: then how did life on this planet start in the first place? ...
... but: then how did life on this planet start in the first place? ...
Macromolecules
... The Atoms of Life The most frequently found atoms in the body are “CHNOPS” (not in this order). Carbon (C) Hydrogen (H) What are other Nitrogen (N) elements you Oxygen (O) expected to be on Phosphorous (P) this list? Sulfur (S) ...
... The Atoms of Life The most frequently found atoms in the body are “CHNOPS” (not in this order). Carbon (C) Hydrogen (H) What are other Nitrogen (N) elements you Oxygen (O) expected to be on Phosphorous (P) this list? Sulfur (S) ...
Practice exam #1 review
... 18.) What of the following is/ are the product(s) of the citric acid cyclea. CO2 b. NAPH and FAPH2 c. ATP d. H2O e. all the above Modified True or False Write T or F at each question and if false correct then make it true. 1. ATP is an energy intermediate T F 2. ATP releases energy when the bond un ...
... 18.) What of the following is/ are the product(s) of the citric acid cyclea. CO2 b. NAPH and FAPH2 c. ATP d. H2O e. all the above Modified True or False Write T or F at each question and if false correct then make it true. 1. ATP is an energy intermediate T F 2. ATP releases energy when the bond un ...
Metabolism

Metabolism (from Greek: μεταβολή metabolē, ""change"") is the set of life-sustaining chemical transformations within the cells of living organisms. These enzyme-catalyzed reactions allow organisms to grow and reproduce, maintain their structures, and respond to their environments. The word metabolism can also refer to all chemical reactions that occur in living organisms, including digestion and the transport of substances into and between different cells, in which case the set of reactions within the cells is called intermediary metabolism or intermediate metabolism.Metabolism is usually divided into two categories: catabolism, the breaking down of organic matter by way of cellular respiration, and anabolism, the building up of components of cells such as proteins and nucleic acids. Usually, breaking down releases energy and building up consumes energy.The chemical reactions of metabolism are organized into metabolic pathways, in which one chemical is transformed through a series of steps into another chemical, by a sequence of enzymes. Enzymes are crucial to metabolism because they allow organisms to drive desirable reactions that require energy that will not occur by themselves, by coupling them to spontaneous reactions that release energy. Enzymes act as catalysts that allow the reactions to proceed more rapidly. Enzymes also allow the regulation of metabolic pathways in response to changes in the cell's environment or to signals from other cells.The metabolic system of a particular organism determines which substances it will find nutritious and which poisonous. For example, some prokaryotes use hydrogen sulfide as a nutrient, yet this gas is poisonous to animals. The speed of metabolism, the metabolic rate, influences how much food an organism will require, and also affects how it is able to obtain that food.A striking feature of metabolism is the similarity of the basic metabolic pathways and components between even vastly different species. For example, the set of carboxylic acids that are best known as the intermediates in the citric acid cycle are present in all known organisms, being found in species as diverse as the unicellular bacterium Escherichia coli and huge multicellular organisms like elephants. These striking similarities in metabolic pathways are likely due to their early appearance in evolutionary history, and their retention because of their efficacy.