Holt Chemistry – Guided Notes, Chapter 1
... • Set up conversion factors, and use them in calculations. • Identify and describe physical properties, including density. • Identify chemical properties. When working with numbers, be careful to distinguish between a quantity and its unit. • _____________ describes something that has magnitude, siz ...
... • Set up conversion factors, and use them in calculations. • Identify and describe physical properties, including density. • Identify chemical properties. When working with numbers, be careful to distinguish between a quantity and its unit. • _____________ describes something that has magnitude, siz ...
1.2 Properties and Changes of Matter
... You must wear safety glasses for this lab. Please, be very careful with the chemicals. You may work in groups of three. ...
... You must wear safety glasses for this lab. Please, be very careful with the chemicals. You may work in groups of three. ...
Simple Chemical Reactions
... 40 cm3 Industrial denatured alcohol (IDA is highly flammable) If you are planning on using alternative fuels contact SSERC first for advice. ...
... 40 cm3 Industrial denatured alcohol (IDA is highly flammable) If you are planning on using alternative fuels contact SSERC first for advice. ...
Notes matter energy
... number and type of atoms in a molecule. For example, H2SO4 (sulfuric acid) is the formula for a molecule because it consists of only nonmetals. The molecule is made up of 2 hydrogen atoms, 1 sulfur atom, and 4 oxygen atoms (and 7 total atoms). Subscripts indicate the number of atoms in the formula ( ...
... number and type of atoms in a molecule. For example, H2SO4 (sulfuric acid) is the formula for a molecule because it consists of only nonmetals. The molecule is made up of 2 hydrogen atoms, 1 sulfur atom, and 4 oxygen atoms (and 7 total atoms). Subscripts indicate the number of atoms in the formula ( ...
chemical bonds - geraldinescience
... • An equation is balanced when the number of atoms of each element on the right side of the equation is equal to the number of atoms of the same element on the left side. • To balance an equation, you must put numbers called coefficients in front of chemical formulas. • A coefficient multiplies the ...
... • An equation is balanced when the number of atoms of each element on the right side of the equation is equal to the number of atoms of the same element on the left side. • To balance an equation, you must put numbers called coefficients in front of chemical formulas. • A coefficient multiplies the ...
Chemical reactions
... • Ionic - lacking discrete unit, or molecule • Composed of both metallic and nonmetallic elements • Electronegativity difference > 1.7 ...
... • Ionic - lacking discrete unit, or molecule • Composed of both metallic and nonmetallic elements • Electronegativity difference > 1.7 ...
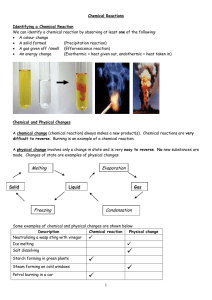
Chemical Reactions
... A word equation is a simple way of showing a chemical reaction. The reactants are shown on the left hand side and the products are shown on the right hand side. The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be a ...
... A word equation is a simple way of showing a chemical reaction. The reactants are shown on the left hand side and the products are shown on the right hand side. The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be a ...
File
... First letter is always capitalized Second letter, if present is always lowercase can be derived from common names or Latin names ...
... First letter is always capitalized Second letter, if present is always lowercase can be derived from common names or Latin names ...
Physical and Chemical Changes Worksheet
... An ice cube is placed in the sun. Later there is a puddle of water. Later still the puddle is gone. ...
... An ice cube is placed in the sun. Later there is a puddle of water. Later still the puddle is gone. ...
Unit 1 Matter Day 32 2016 Counting Atoms
... Is it balanced? If so, you have supported the Law of Conservation of Matter (Mass). ...
... Is it balanced? If so, you have supported the Law of Conservation of Matter (Mass). ...
High School Chemistry Essential Questions
... 2. What observations about chemical systems and chemical interactions lead us to form the physical, graphical, and mathematical models that we use to represent, analyze, and communicate structure and relationships in chemical systems and chemical interactions? 3. How do we use the physical models, s ...
... 2. What observations about chemical systems and chemical interactions lead us to form the physical, graphical, and mathematical models that we use to represent, analyze, and communicate structure and relationships in chemical systems and chemical interactions? 3. How do we use the physical models, s ...
Balancing Equations
... compound. Subscripts are determined by the valence electrons (charges for ionic or sharing for covalent) n Think ...
... compound. Subscripts are determined by the valence electrons (charges for ionic or sharing for covalent) n Think ...
Process Safety Management
... chemical to break down? • Does the process cause the chemical to boil or freeze? ...
... chemical to break down? • Does the process cause the chemical to boil or freeze? ...
chemical reaction?
... • Because of the law of conservation of mass, chemical equations must show the same numbers and kinds of atoms on both sides of an arrow. • To balance an equation, ________________ are placed in front of a chemical formula. Only ____________, not subscripts, can be changed when balancing a chemical ...
... • Because of the law of conservation of mass, chemical equations must show the same numbers and kinds of atoms on both sides of an arrow. • To balance an equation, ________________ are placed in front of a chemical formula. Only ____________, not subscripts, can be changed when balancing a chemical ...
CLASSROOM CONNECTORS
... 1. Dissolve a small amount of salt in water. Is this a chemical change or a physical change? (Physical) Allow the solution to evaporate. Is this a chemical or physical change? (physical) 2. Add a small piece of zinc to 5 ml of hydrochloric acid. Is the reaction a chemical or physical change? (Chemic ...
... 1. Dissolve a small amount of salt in water. Is this a chemical change or a physical change? (Physical) Allow the solution to evaporate. Is this a chemical or physical change? (physical) 2. Add a small piece of zinc to 5 ml of hydrochloric acid. Is the reaction a chemical or physical change? (Chemic ...