How to Balance Chemical Equations
... 2. How to balance a chemical equation In balancing chemical equations you need to remember to three important points. a) You do not change the chemical _____________ of a substance to balance an equation. b) An equation is balanced by writing __________numbers before a chemical symbol or formula. c) ...
... 2. How to balance a chemical equation In balancing chemical equations you need to remember to three important points. a) You do not change the chemical _____________ of a substance to balance an equation. b) An equation is balanced by writing __________numbers before a chemical symbol or formula. c) ...
matter and its reactivity. Objects in the universe are composed of
... color, odor, phase, density, solubility, heat and electrical conductivity, and boiling and freezing points. 3.1b Solubility can be affected by the nature of the solute and solvent, temperature, and pressure. The rate of solution can be affected by the size of the particles, stirring, temperature, an ...
... color, odor, phase, density, solubility, heat and electrical conductivity, and boiling and freezing points. 3.1b Solubility can be affected by the nature of the solute and solvent, temperature, and pressure. The rate of solution can be affected by the size of the particles, stirring, temperature, an ...
Name
... Ⓡ 8.5 (E) Chemical Reactions: Students will be able to investigate how evidences of chemical reactions indicate that new substances are formed. Ⓡ 8.5 (F) Balancing Equations: Students will be able to recognize whether or not a chemical equation is balanced and how that applies to the Law of Conserva ...
... Ⓡ 8.5 (E) Chemical Reactions: Students will be able to investigate how evidences of chemical reactions indicate that new substances are formed. Ⓡ 8.5 (F) Balancing Equations: Students will be able to recognize whether or not a chemical equation is balanced and how that applies to the Law of Conserva ...
Notes for Matter Packet- Balancing equations (PDF
... products – New pure substance/s that are made from reactants in a Chemical Reaction ...
... products – New pure substance/s that are made from reactants in a Chemical Reaction ...
Targets of Opportunity
... endangering refinery communities...The environmental hazards of HF as used at refineries have to do with the high volumes utilized, the potential for high temperatures and pressures to be involved in a release, and the tendency of HF, once released to the environment, to form deadly gas clouds that ...
... endangering refinery communities...The environmental hazards of HF as used at refineries have to do with the high volumes utilized, the potential for high temperatures and pressures to be involved in a release, and the tendency of HF, once released to the environment, to form deadly gas clouds that ...
A Study of Matter
... • Boiling point- liquid turns to a gas (water to water vapor) • Condensation- where a gas turns to a liquid (the sweating on a glass that is colder than it’s environment) • Sublimation point- temperature at which a solid changes directly to a gas without first changing into a liquid. (dry ice) ...
... • Boiling point- liquid turns to a gas (water to water vapor) • Condensation- where a gas turns to a liquid (the sweating on a glass that is colder than it’s environment) • Sublimation point- temperature at which a solid changes directly to a gas without first changing into a liquid. (dry ice) ...
Candle Mass Lab and the Law of Conservation of Matter Notes.
... Describing Chemical Reactions Balancing Chemical Equations • Is the following sentence true or false? • Because the equation ...
... Describing Chemical Reactions Balancing Chemical Equations • Is the following sentence true or false? • Because the equation ...
Chemistry Standard Outline
... SC2 Students will relate how the Law of Conservation of Matter is used to determine chemical composition in compounds and chemical reactions. SC2a. Identify and balance the following types of chemical equations: • Synthesis • Decomposition • Single Replacement • Double Replacement • Combustion SC2b. ...
... SC2 Students will relate how the Law of Conservation of Matter is used to determine chemical composition in compounds and chemical reactions. SC2a. Identify and balance the following types of chemical equations: • Synthesis • Decomposition • Single Replacement • Double Replacement • Combustion SC2b. ...
Chemical Formulas
... HC 2 H 3 O 2 (vinegar), NaHCO 3 (baking soda), NH 4 (ammonia), and C 6 H 12 O 6 (glucose). ...
... HC 2 H 3 O 2 (vinegar), NaHCO 3 (baking soda), NH 4 (ammonia), and C 6 H 12 O 6 (glucose). ...
TEK 8.5D: Chemical Formulas
... HC 2 H 3 O 2 (vinegar), NaHCO 3 (baking soda), NH 4 (ammonia), and C 6 H 12 O 6 (glucose). ...
... HC 2 H 3 O 2 (vinegar), NaHCO 3 (baking soda), NH 4 (ammonia), and C 6 H 12 O 6 (glucose). ...
C6-Chemical Reactions
... Do these 4 indications of chemical change ALWAYS mean chemical changes?? Transfer of energy Fire always means chemical change Heat, cooling, and light can indicate either type of change. To be ...
... Do these 4 indications of chemical change ALWAYS mean chemical changes?? Transfer of energy Fire always means chemical change Heat, cooling, and light can indicate either type of change. To be ...
1 - mvhs-fuhsd.org
... the changes it undergoes. 4. What branch of chemistry is most concerned with the study of carbon compounds? Organic Chemistry 5. What does the word chemical, as used by scientists ,mean? 6. Briefly describe the difference between basic research, applied research, and technological development. Provi ...
... the changes it undergoes. 4. What branch of chemistry is most concerned with the study of carbon compounds? Organic Chemistry 5. What does the word chemical, as used by scientists ,mean? 6. Briefly describe the difference between basic research, applied research, and technological development. Provi ...
ap chemistry – 2013-2014
... This AP Chemistry course is designed to be the equivalent of the general chemistry course usually taken during the first year of college. This course is structured around six big ideas that include: Structure of matter, properties of matter-characteristics, states, and forces of attraction, chemical ...
... This AP Chemistry course is designed to be the equivalent of the general chemistry course usually taken during the first year of college. This course is structured around six big ideas that include: Structure of matter, properties of matter-characteristics, states, and forces of attraction, chemical ...
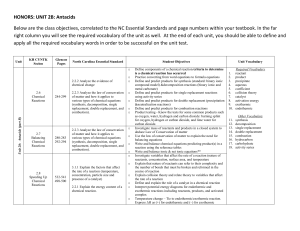
HONORS: UNIT 2B: Antacids Below are the class objectives
... Below are the class objectives, correlated to the NC Essential Standards and page numbers within your textbook. In the far right column you will see the required vocabulary of the unit as well. At the end of each unit, you should be able to define and apply all the required vocabulary words in order ...
... Below are the class objectives, correlated to the NC Essential Standards and page numbers within your textbook. In the far right column you will see the required vocabulary of the unit as well. At the end of each unit, you should be able to define and apply all the required vocabulary words in order ...
File
... with one another, that is, when they undergo a change in composition.. Chemical properties include: acidity, basicity, combustibility, and ___________________. Chemical and Physical properties can be used to ____________________ a substance by comparing the properties of the substance to unknown sub ...
... with one another, that is, when they undergo a change in composition.. Chemical properties include: acidity, basicity, combustibility, and ___________________. Chemical and Physical properties can be used to ____________________ a substance by comparing the properties of the substance to unknown sub ...
Chemical properties Chemical properties can be recognized only
... Chemical properties Chemical properties can be recognized only when substances react or do not react chemically with one another, that is, when they undergo a change in composition. The following chemical properties can be used to help identify a substance: Ability to burn The ability to burn involv ...
... Chemical properties Chemical properties can be recognized only when substances react or do not react chemically with one another, that is, when they undergo a change in composition. The following chemical properties can be used to help identify a substance: Ability to burn The ability to burn involv ...