13.2 Chemical Formulas
... What is a chemical formula? Chemical formulas have two important parts: chemical symbols for the elements in the compound and subscripts that tell how many atoms of each element are needed to form the compound. The chemical formula for water, H2O, tells us that a water molecule is made of the elemen ...
... What is a chemical formula? Chemical formulas have two important parts: chemical symbols for the elements in the compound and subscripts that tell how many atoms of each element are needed to form the compound. The chemical formula for water, H2O, tells us that a water molecule is made of the elemen ...
Ch. 1-- Matter and Change
... 2Hhydrogen 2H2O 2 + O2 molecules plus one oxygen which means “____________________________________ molecule yields two water molecules ________________________________________________.” ...
... 2Hhydrogen 2H2O 2 + O2 molecules plus one oxygen which means “____________________________________ molecule yields two water molecules ________________________________________________.” ...
Chemistry Study Guide
... Conservation of Mass- Mass cannot be created or destroyed in a chemical reaction. ...
... Conservation of Mass- Mass cannot be created or destroyed in a chemical reaction. ...
Chemistry Study Guide
... Conservation of Mass- Mass cannot be created or destroyed in a chemical reaction. ...
... Conservation of Mass- Mass cannot be created or destroyed in a chemical reaction. ...
CHAPTER 1 Practice Exercises 1.1 12.3 g Cd 1.3 26.9814 u 1.5
... Silver and gold are in the same periodic table group as copper, so they might well be expected to occur together in nature, because of their similar properties and tendencies to form similar compounds. ...
... Silver and gold are in the same periodic table group as copper, so they might well be expected to occur together in nature, because of their similar properties and tendencies to form similar compounds. ...
File
... Number of each element: ________________________________________________ Number of atoms: ____________________________________________________ ...
... Number of each element: ________________________________________________ Number of atoms: ____________________________________________________ ...
Name__________________________________ Block______
... Physical and Chemical Changes in Matter Put a “P” next to descriptions or indications of physical change or property. Put a “C” next to descriptions or indications of a chemical change or property. 1._____ the physical properties of a substance may change, but the kind of substance does not change 2 ...
... Physical and Chemical Changes in Matter Put a “P” next to descriptions or indications of physical change or property. Put a “C” next to descriptions or indications of a chemical change or property. 1._____ the physical properties of a substance may change, but the kind of substance does not change 2 ...
Safety Research and Competitiveness and First
... safe use of its products and the safety of its production facilities. It does, and will do, whatever is needed to protect people and the environment from harm. ...
... safe use of its products and the safety of its production facilities. It does, and will do, whatever is needed to protect people and the environment from harm. ...
Matter in Chemistry
... Chemical Change A chemical change occurs when matter changes chemically into an entirely different substance with different properties. • When vinegar (liquid) and baking soda (solid) combine, they form carbon dioxide (gas). ...
... Chemical Change A chemical change occurs when matter changes chemically into an entirely different substance with different properties. • When vinegar (liquid) and baking soda (solid) combine, they form carbon dioxide (gas). ...
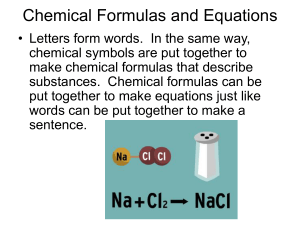
Chemical Formulas and Equations
... • Letters form words. In the same way, chemical symbols are put together to make chemical formulas that describe substances. Chemical formulas can be put together to make equations just like words can be put together to make a ...
... • Letters form words. In the same way, chemical symbols are put together to make chemical formulas that describe substances. Chemical formulas can be put together to make equations just like words can be put together to make a ...
Chemical Principles – by Steven Zumdahl (5 ) Chapter 1
... Fundamental laws of Nature (for chemical reactions) Law of conservation of matter: There is no detectible increase or decrease in the quantity of matter during an ordinary chemical change. Law of conservation of energy: Energy cannot be created or destroyed in an ordinary chemical reaction. It may o ...
... Fundamental laws of Nature (for chemical reactions) Law of conservation of matter: There is no detectible increase or decrease in the quantity of matter during an ordinary chemical change. Law of conservation of energy: Energy cannot be created or destroyed in an ordinary chemical reaction. It may o ...
Introduction to Chemical Reactions
... Chemical Equations are balanced to show the same number of atoms of each element on each side. The Law of Conservation of Mass says that atoms won’t be created or destroyed in a chemical reaction. That is why you have to balance chemical equations! ...
... Chemical Equations are balanced to show the same number of atoms of each element on each side. The Law of Conservation of Mass says that atoms won’t be created or destroyed in a chemical reaction. That is why you have to balance chemical equations! ...
SNC2D – Science 10 Tuesday April 26th, 2010 Mr. Sourlis and Mr
... b) Solid zinc metal reacts with aqueous hydrogen chloride to produce hydrogen gas (H2) and aqueous zinc chloride ...
... b) Solid zinc metal reacts with aqueous hydrogen chloride to produce hydrogen gas (H2) and aqueous zinc chloride ...
Discover Chemical Changes - gk-12
... This lesson should be an open inquiry-based lesson where you have many stations set up with before and after chemical changes so that students can make the chemical changes happen themselves or at least make observations of chemical changes that have happened at each station. I have listed above 9 p ...
... This lesson should be an open inquiry-based lesson where you have many stations set up with before and after chemical changes so that students can make the chemical changes happen themselves or at least make observations of chemical changes that have happened at each station. I have listed above 9 p ...