GHW - Louisiana Tech University
... Of course if we used some other mass unit for the mole such as "pound mole", the "number" would be different than 6.022 x 1023. 21) Given 5 moles of Sulfuric Acid having a formula of H2SO4 answer the following questions: ...
... Of course if we used some other mass unit for the mole such as "pound mole", the "number" would be different than 6.022 x 1023. 21) Given 5 moles of Sulfuric Acid having a formula of H2SO4 answer the following questions: ...
2.4 The Periodic Table
... and the percentage of each must be known. • The atomic weight is calculated as the sum of the masses of the individual isotopes for that element. Atomic weight = [(isotope abundance) × (isotope mass)] • The Greek symbol indicates summing of terms. ...
... and the percentage of each must be known. • The atomic weight is calculated as the sum of the masses of the individual isotopes for that element. Atomic weight = [(isotope abundance) × (isotope mass)] • The Greek symbol indicates summing of terms. ...
EPSc 446 STABLE ISOTOPE GEOCHEMISTRY Instructor: Bob Criss
... Problem: Disparity between Atomic No. and Atomic Weight ...
... Problem: Disparity between Atomic No. and Atomic Weight ...
Unit 3 Notes, Practice, and Review
... Unit 3 Review 19. The atomic number is unique for every element. It also tells the number of protons in that element. Every element on the periodic table has a unique number of protons. It’s like an element’s Social Security Number. 20. Atomic number is the number of protons and electrons in an ato ...
... Unit 3 Review 19. The atomic number is unique for every element. It also tells the number of protons in that element. Every element on the periodic table has a unique number of protons. It’s like an element’s Social Security Number. 20. Atomic number is the number of protons and electrons in an ato ...
11129_evl_ch1_ste_eleve (3)
... 7. Six different elements are represented below according to the Rutherford-Bohr atomic model. ...
... 7. Six different elements are represented below according to the Rutherford-Bohr atomic model. ...
11129_evl_ch1_ste_corr
... electron shells. Some of them (boron, nitrogen, fluorine and neon) have two electron shells; others (sodium and magnesium) have three. ...
... electron shells. Some of them (boron, nitrogen, fluorine and neon) have two electron shells; others (sodium and magnesium) have three. ...
10_Chemistry homework
... http://www.800mainstreet.com/33/0003-001-atoms-particles.html An atom of an element is the simplest particle that displays the properties of the element. We cannot see individual atoms with the naked eye. Visit the following site to see scanning tunneling microscope images of atoms. http://www.almad ...
... http://www.800mainstreet.com/33/0003-001-atoms-particles.html An atom of an element is the simplest particle that displays the properties of the element. We cannot see individual atoms with the naked eye. Visit the following site to see scanning tunneling microscope images of atoms. http://www.almad ...
What`s Inside an Element
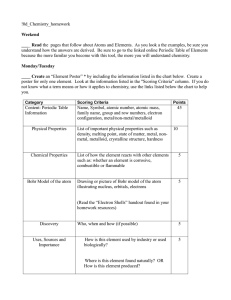
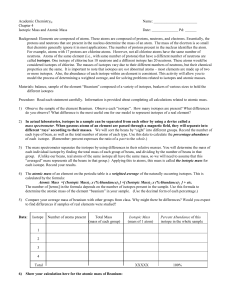
... When selecting elements for the models, consider the time you have available and the students you teach. Some of the elements have high atomic mass and will take time to add to their models, because of the high number of protons and electrons. Students will have an easier time if you make a sample m ...
... When selecting elements for the models, consider the time you have available and the students you teach. Some of the elements have high atomic mass and will take time to add to their models, because of the high number of protons and electrons. Students will have an easier time if you make a sample m ...
Atomic Structure 3: ISOTOPES
... element have the same number of protons, as given by the atomic number. You also know that the number of electrons in an uncharged atom equals the number of protons. The number of neutrons in the nucleus of an atom is not so predictable. The periodic table helps us out, though, by giving the atomic ...
... element have the same number of protons, as given by the atomic number. You also know that the number of electrons in an uncharged atom equals the number of protons. The number of neutrons in the nucleus of an atom is not so predictable. The periodic table helps us out, though, by giving the atomic ...
Notes 4.3 filled in
... each having the mass of 1 amu. Q: an atom has 5 protons and 7 neutrons, calculate the MN. A: 5 amu + 7 amu = 12 amu Q: Which element is that? (Look in the PT) A: 5 protons, it’s Boron D. Isotopes Every element exists as multiple versions, called isotopes Isotopes of the same element are identica ...
... each having the mass of 1 amu. Q: an atom has 5 protons and 7 neutrons, calculate the MN. A: 5 amu + 7 amu = 12 amu Q: Which element is that? (Look in the PT) A: 5 protons, it’s Boron D. Isotopes Every element exists as multiple versions, called isotopes Isotopes of the same element are identica ...
In actual laboratories, isotopes in a sample can be
... that chemists generally ignore it in most applications. The number of protons present in the nucleus identifies the atom. For example, atoms with 17 protons are chlorine atoms. However, not all chlorine atoms have the same number of neutrons. Atoms of the same element (i.e., with same number of prot ...
... that chemists generally ignore it in most applications. The number of protons present in the nucleus identifies the atom. For example, atoms with 17 protons are chlorine atoms. However, not all chlorine atoms have the same number of neutrons. Atoms of the same element (i.e., with same number of prot ...
Chapter 4 powerpoint
... radioactive decay. • Unstable radioactive elements undergo radioactive decay thus forming stable nonradioactive elements. ...
... radioactive decay. • Unstable radioactive elements undergo radioactive decay thus forming stable nonradioactive elements. ...
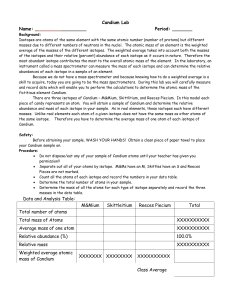
Candium Lab - OCPS TeacherPress
... 1.) Write a formula for each of the steps listed in calculations section. Example: Avg. mass of one atom = total mass of all atoms of an isotope total number of atoms of that isotope Relative Abundance = ...
... 1.) Write a formula for each of the steps listed in calculations section. Example: Avg. mass of one atom = total mass of all atoms of an isotope total number of atoms of that isotope Relative Abundance = ...
PPT_Topic2
... We will do this by Testing different metals in flame tests to discover which colour they produce. We will have succeeded if We can work out which elements might be best for our red distress flare. ...
... We will do this by Testing different metals in flame tests to discover which colour they produce. We will have succeeded if We can work out which elements might be best for our red distress flare. ...
1. Structure and Properties of the Atom
... Atoms of different elements can combine with one another in simple (whole number) ratios to form compounds. ...
... Atoms of different elements can combine with one another in simple (whole number) ratios to form compounds. ...
Make a large atom with p:95, n:146, e:95 - TSDCurriculum
... 5. READ: This simulation only lets you to build atoms that exist in nature or have been made by scientists. If you can’t build it, it can't be made in the real world. Scientists use the word isotope to distinguish between atoms that have the same number of protons but different numbers of neutrons. ...
... 5. READ: This simulation only lets you to build atoms that exist in nature or have been made by scientists. If you can’t build it, it can't be made in the real world. Scientists use the word isotope to distinguish between atoms that have the same number of protons but different numbers of neutrons. ...
The History of the Periodic Table
... simpler substances called elements, and for centuries, scientists have looked for patterns that would explain how and why certain elements react. Our definition of the word “element” has changed as our understanding of the structure of matter has improved. Almost 25 centuries ago, the Greek philosop ...
... simpler substances called elements, and for centuries, scientists have looked for patterns that would explain how and why certain elements react. Our definition of the word “element” has changed as our understanding of the structure of matter has improved. Almost 25 centuries ago, the Greek philosop ...
IPS Unit 8 – Periodic Table Review Worksheet
... 9. Atoms of the same element that have different numbers of neutrons are called (isotopes/electron clouds). 10. In the periodic table, elements are arranged by increasing atomic (power/number). 11. An electron dot diagram uses the symbol of an element and dots to represent the (quarks/electrons) in ...
... 9. Atoms of the same element that have different numbers of neutrons are called (isotopes/electron clouds). 10. In the periodic table, elements are arranged by increasing atomic (power/number). 11. An electron dot diagram uses the symbol of an element and dots to represent the (quarks/electrons) in ...
Notes powerpoint
... • Periodic means “repeated in a pattern.” • In the late 1800s, Dmitri Mendeleev, a Russian chemist, searched for a way to organize the elements. • When he arranged all the elements known at that time in order of increasing atomic masses, he discovered a pattern. ...
... • Periodic means “repeated in a pattern.” • In the late 1800s, Dmitri Mendeleev, a Russian chemist, searched for a way to organize the elements. • When he arranged all the elements known at that time in order of increasing atomic masses, he discovered a pattern. ...
SECTION REVIEW
... ________ 11. The atomic number of an element is the sum of the protons and electrons in an atom of that element. ________ 12. The atomic number of an atom is the total number of protons in an atom of that element. ________ 13. An atom of nitrogen has 7 protons and 7 neutrons. ________ 14. Relative a ...
... ________ 11. The atomic number of an element is the sum of the protons and electrons in an atom of that element. ________ 12. The atomic number of an atom is the total number of protons in an atom of that element. ________ 13. An atom of nitrogen has 7 protons and 7 neutrons. ________ 14. Relative a ...
Atom Internet Scavenger Hunt
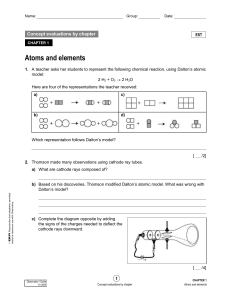
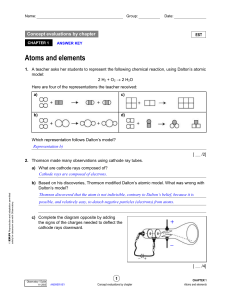
... that matter was made up of smaller particles and was the first person to write it down. But he had no experimental proof. A number of scientists after this attempted to prove Democritus’ theory. It wasn’t until the late 1700’s/ early 1800’s that John Dalton proposed that matter was composed of atoms ...
... that matter was made up of smaller particles and was the first person to write it down. But he had no experimental proof. A number of scientists after this attempted to prove Democritus’ theory. It wasn’t until the late 1700’s/ early 1800’s that John Dalton proposed that matter was composed of atoms ...
Chapter 4 Atomic Structure
... a) Cathode rays have identical properties regardless of the element used to produce them. All elements must contain identically charged electrons. b) Atoms are neutral, so there must be positive particles in the atom to balance the negative charge of the electrons c) Electrons have so little mass th ...
... a) Cathode rays have identical properties regardless of the element used to produce them. All elements must contain identically charged electrons. b) Atoms are neutral, so there must be positive particles in the atom to balance the negative charge of the electrons c) Electrons have so little mass th ...
EXPERIMENT 4 – The Periodic Table
... In this experiment, you will be looking at some elements in the laboratory display. Some look different from each other, while others look similar. Elements can be categorized in several ways. In this experiment, you are going to group elements by similarities in their physical properties. Elements ...
... In this experiment, you will be looking at some elements in the laboratory display. Some look different from each other, while others look similar. Elements can be categorized in several ways. In this experiment, you are going to group elements by similarities in their physical properties. Elements ...
atomic number on the periodic table
... History of the Periodic Table • Dmitri Mendeleev, a Russian scientist born in Siberia in 1834, is known as the father of the periodic table of the elements • The periodic table is designed to help you predict chemical and physical properties of elements ...
... History of the Periodic Table • Dmitri Mendeleev, a Russian scientist born in Siberia in 1834, is known as the father of the periodic table of the elements • The periodic table is designed to help you predict chemical and physical properties of elements ...