Chapter_04_Structure_of_the_atom
... • Understand how Dalton's theory explains the conservation of mass. ...
... • Understand how Dalton's theory explains the conservation of mass. ...
Chapter 17 notes
... • Scientists found five quarks and hypothesized that a sixth quark existed. However, it took a team of nearly 450 scientists from around the world several years to find the sixth quark. • The tracks of the sixth quark were hard to detect because only about one billionth of a percent of the proton co ...
... • Scientists found five quarks and hypothesized that a sixth quark existed. However, it took a team of nearly 450 scientists from around the world several years to find the sixth quark. • The tracks of the sixth quark were hard to detect because only about one billionth of a percent of the proton co ...
periodic table - Cloudfront.net
... • Scientists found five quarks and hypothesized that a sixth quark existed. However, it took a team of nearly 450 scientists from around the world several years to find the sixth quark. • The tracks of the sixth quark were hard to detect because only about one billionth of a percent of the proton co ...
... • Scientists found five quarks and hypothesized that a sixth quark existed. However, it took a team of nearly 450 scientists from around the world several years to find the sixth quark. • The tracks of the sixth quark were hard to detect because only about one billionth of a percent of the proton co ...
Masses of Atoms - Pelham City Schools
... • Scientists found five quarks and hypothesized that a sixth quark existed. However, it took a team of nearly 450 scientists from around the world several years to find the sixth quark. • The tracks of the sixth quark were hard to detect because only about one billionth of a percent of the proton co ...
... • Scientists found five quarks and hypothesized that a sixth quark existed. However, it took a team of nearly 450 scientists from around the world several years to find the sixth quark. • The tracks of the sixth quark were hard to detect because only about one billionth of a percent of the proton co ...
Synthesis of elements by helium and oxygen building blocks Bohr
... gives the hydrogen and helium isotopes enough kinetic energy to overcome the long-range repulsive Coulomb force and come within the short-range of the attractive strong nuclear force. The processes described above produce only nuclei of the elements: In the infernal heat of about 107 K atoms cannot ...
... gives the hydrogen and helium isotopes enough kinetic energy to overcome the long-range repulsive Coulomb force and come within the short-range of the attractive strong nuclear force. The processes described above produce only nuclei of the elements: In the infernal heat of about 107 K atoms cannot ...
Masses of Atoms
... 80% of Boron in nature have 5 protons, 6 neutrons ~ 11 amu 20% of Boron in nature have 5 protons, 5 neutrons ~ 10 amu (.8 • 11 amu) + (.2 • 10 amu) = 8.8 amu + 2 amu = 10.8 amu There are a few extra isotopes out there that we did not include. ...
... 80% of Boron in nature have 5 protons, 6 neutrons ~ 11 amu 20% of Boron in nature have 5 protons, 5 neutrons ~ 10 amu (.8 • 11 amu) + (.2 • 10 amu) = 8.8 amu + 2 amu = 10.8 amu There are a few extra isotopes out there that we did not include. ...
atom - Social Circle City Schools
... • In nature, most elements are found as mixtures of isotopes. Usually, the relative abundance of each isotope is constant. –Ex. In a banana, 93.26% is potassium-39, 6.73% is potassium-41 and 0.01% is potassium40. In another banana or in a different source of potassium, the percentage composition of ...
... • In nature, most elements are found as mixtures of isotopes. Usually, the relative abundance of each isotope is constant. –Ex. In a banana, 93.26% is potassium-39, 6.73% is potassium-41 and 0.01% is potassium40. In another banana or in a different source of potassium, the percentage composition of ...
Chemistry: Matter and Change
... • Understand how Dalton's theory explains the conservation of mass. ...
... • Understand how Dalton's theory explains the conservation of mass. ...
Atoms, Isotopes and Relative Atomic Masses
... Antimony, Sb, is a metal used in alloys to make lead harder. Bullets contain about 1% of antimony for this reason. Antimony has two main isotopes. (i) ...
... Antimony, Sb, is a metal used in alloys to make lead harder. Bullets contain about 1% of antimony for this reason. Antimony has two main isotopes. (i) ...
Atoms and Elements: Are they Related?
... 1) All matter is made of atoms. Atoms are indivisible and indestructible. 2) All atoms of a given element are identical in mass and properties 3) Compounds are formed by a combination of two or more different kinds of atoms. 4) A chemical reaction is a rearrangement of atoms. ...
... 1) All matter is made of atoms. Atoms are indivisible and indestructible. 2) All atoms of a given element are identical in mass and properties 3) Compounds are formed by a combination of two or more different kinds of atoms. 4) A chemical reaction is a rearrangement of atoms. ...
Chapter 2 Atoms and Radioactivity Outline 2.1 Atoms and Their
... – The mass number is the number of protons and neutrons for a given isotope. – For example, nitrogen-14 has seven protons and seven neutrons. – The atomic mass is the average atomic mass for all the isotopes of an element found in nature. – This number is found on the periodic table often below the ...
... – The mass number is the number of protons and neutrons for a given isotope. – For example, nitrogen-14 has seven protons and seven neutrons. – The atomic mass is the average atomic mass for all the isotopes of an element found in nature. – This number is found on the periodic table often below the ...
Atomic Number
... Chapter Two Summary (continued) • 2.4 Radioactivity and Radioisotopes – Some atomic isotopes emit radiation (a form of energy) spontaneously from their nucleus in a process called radioactive decay. – Isotopes that undergo radioactive decay are called radioisotopes, and the high-energy particles gi ...
... Chapter Two Summary (continued) • 2.4 Radioactivity and Radioisotopes – Some atomic isotopes emit radiation (a form of energy) spontaneously from their nucleus in a process called radioactive decay. – Isotopes that undergo radioactive decay are called radioisotopes, and the high-energy particles gi ...
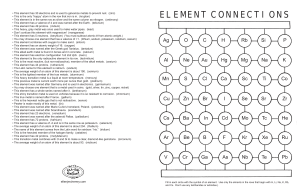
element connections
... • This element has 30 electrons and is used to galvanize metals to prevent rust. (zinc) • This is the only “happy” atom in the row that iron is in. (krypton) • This element is in the same row as silver and the same column as nitrogen. (antimony) • This element has a valence of 4 and was named after ...
... • This element has 30 electrons and is used to galvanize metals to prevent rust. (zinc) • This is the only “happy” atom in the row that iron is in. (krypton) • This element is in the same row as silver and the same column as nitrogen. (antimony) • This element has a valence of 4 and was named after ...
Chapter #4 Section Assessment #1 - 33
... *Elements with high electronegativies and ionization energies tend to be clustered in the upper right hand corner of the periodic table. [Don’t burden your brain too much with these examples just yet. We’ll be looking at them more closely in chapter 6.] ...
... *Elements with high electronegativies and ionization energies tend to be clustered in the upper right hand corner of the periodic table. [Don’t burden your brain too much with these examples just yet. We’ll be looking at them more closely in chapter 6.] ...
Periodic Law
... The groups in the periodic table have "A" and "B" designations. The elements in the A groups, which appear in two parts—two at the beginning and six at the end of the table—are known as the main group elements. Those in the B groups, which are in between the two A group divisions, are called transit ...
... The groups in the periodic table have "A" and "B" designations. The elements in the A groups, which appear in two parts—two at the beginning and six at the end of the table—are known as the main group elements. Those in the B groups, which are in between the two A group divisions, are called transit ...
Help us improve Wikipedia by supporting it financially
... including light and caloric. By 1818, Jöns Jakob Berzelius had determined atomic weights for forty-five of the forty-nine accepted elements. Dmitri Mendeleev had sixtysix elements in his periodic table of 1869. From Boyle until the early 20th century, an element was defined as a pure substance that ...
... including light and caloric. By 1818, Jöns Jakob Berzelius had determined atomic weights for forty-five of the forty-nine accepted elements. Dmitri Mendeleev had sixtysix elements in his periodic table of 1869. From Boyle until the early 20th century, an element was defined as a pure substance that ...
по темі “Atoms and Molecules. The Periodic Table”
... 2. The invention of the periodic table is generally credited to Henry Moseley. 3. Dmitri Mendeleev knew only 56 elements at that time. 4. Mendeleev found he could arrange the elements in a grid so that each element had a lower atomic weight than the one on its left. 5. Adams and Le Verrier could be ...
... 2. The invention of the periodic table is generally credited to Henry Moseley. 3. Dmitri Mendeleev knew only 56 elements at that time. 4. Mendeleev found he could arrange the elements in a grid so that each element had a lower atomic weight than the one on its left. 5. Adams and Le Verrier could be ...
1 TEST DATE:
... The electron has very little mass compared to the ____________________________ or ___________________________ . The mass of the atom depends on the nucleus and how many _________________________ and _________________________ it has. The sum of the protons and neutrons is the mass ___________________ ...
... The electron has very little mass compared to the ____________________________ or ___________________________ . The mass of the atom depends on the nucleus and how many _________________________ and _________________________ it has. The sum of the protons and neutrons is the mass ___________________ ...
14.1 Structure of the Atom
... graduate research under the guidance of the man who discovered the electron, J. J. Thomson. Through his research with Thomson, Rutherford became interested in studying radioactivity. In 1898 he described two kinds of particles emitted from radioactive atoms, calling them alpha and beta particles. He ...
... graduate research under the guidance of the man who discovered the electron, J. J. Thomson. Through his research with Thomson, Rutherford became interested in studying radioactivity. In 1898 he described two kinds of particles emitted from radioactive atoms, calling them alpha and beta particles. He ...
DO NOW
... Read “The Bohr Model and Valence Electrons” from page 141. Take Cornell Notes, defining the following terms: - Bohr Model - Valence Electrons ...
... Read “The Bohr Model and Valence Electrons” from page 141. Take Cornell Notes, defining the following terms: - Bohr Model - Valence Electrons ...
sample
... Around the same time as Brand’s discovery of phosphorus, John Mayow was experimenting with air. He published his results in 1668. In one experiment, Mayow put a lighted candle in a dish of water and covered it with an upturned jar. The flame eventually went out, and water rose a little inside the ja ...
... Around the same time as Brand’s discovery of phosphorus, John Mayow was experimenting with air. He published his results in 1668. In one experiment, Mayow put a lighted candle in a dish of water and covered it with an upturned jar. The flame eventually went out, and water rose a little inside the ja ...
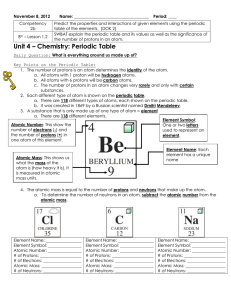
Intro to the Periodic Table
... c. How many protons are in one atom of this element? __________________ d. How many electrons are in one atom of this element? __________________ e. What is the atomic mass of this element? ______________________ f. How many neutrons are in one atom of this element? __________________ 8. Find the el ...
... c. How many protons are in one atom of this element? __________________ d. How many electrons are in one atom of this element? __________________ e. What is the atomic mass of this element? ______________________ f. How many neutrons are in one atom of this element? __________________ 8. Find the el ...
Word List
... 1.1 I can write the names and symbols of the elements in columns 1A – 4A on the periodic table. 1.5 I can write the names and symbols of the elements in columns 5A- 8A on the periodic table. 1.12 I can write the names and symbols of selected transition metals, lanthanides and actinides (1B12B) on th ...
... 1.1 I can write the names and symbols of the elements in columns 1A – 4A on the periodic table. 1.5 I can write the names and symbols of the elements in columns 5A- 8A on the periodic table. 1.12 I can write the names and symbols of selected transition metals, lanthanides and actinides (1B12B) on th ...