Chapter-18
... point (the initial state) and the on or by the gas. final point (the final state) and © 2014 John Wiley & Sons, Inc. All rights reserved. ...
... point (the initial state) and the on or by the gas. final point (the final state) and © 2014 John Wiley & Sons, Inc. All rights reserved. ...
Epoxies and Glass Transition Temperature
... however; they depend upon the duration and extent to which the Tg range is exceeded. Brief excursions above the Tg will not irrevocably “damage” the material. As an epoxy returns to ambient temperatures, its strength profile is typically restored. It is important that design engineers understand the ...
... however; they depend upon the duration and extent to which the Tg range is exceeded. Brief excursions above the Tg will not irrevocably “damage” the material. As an epoxy returns to ambient temperatures, its strength profile is typically restored. It is important that design engineers understand the ...
Instrumental Methods of Analysis
... Generation of phase diagram and study of phase transitions In determination of heat of reaction. In specific heat determination. In determination of thermal diffusitivity. 2)Analytical Chemistry Identification of substance Identification of products Melting point and boiling determination and study ...
... Generation of phase diagram and study of phase transitions In determination of heat of reaction. In specific heat determination. In determination of thermal diffusitivity. 2)Analytical Chemistry Identification of substance Identification of products Melting point and boiling determination and study ...
Measuring Temperature
... state of water 273.16 K. The unit is 1 Kelvin = 1 K. T is used for absolute temperature measured on Kelvin scale. The triple state of water is the temperature where water can exist in all its three states. Apart from the Kelvin scale, the Celsius scale is accepted in Europe. The unit of the Celsius ...
... state of water 273.16 K. The unit is 1 Kelvin = 1 K. T is used for absolute temperature measured on Kelvin scale. The triple state of water is the temperature where water can exist in all its three states. Apart from the Kelvin scale, the Celsius scale is accepted in Europe. The unit of the Celsius ...
MME 2006 Metallurgical Thermodynamics
... Isotherms lower than the critical temperature consist of three distinct regions: vapor, liquid-vapor, liquid • At the horizontal sections which represent the phase change between vapor and liquid, the constant pressure is the vapor pressure • Ratio of liquid and vapor phases can be obtained by the l ...
... Isotherms lower than the critical temperature consist of three distinct regions: vapor, liquid-vapor, liquid • At the horizontal sections which represent the phase change between vapor and liquid, the constant pressure is the vapor pressure • Ratio of liquid and vapor phases can be obtained by the l ...
Course 3: Pressure – Volume – Temperature Relationship of Pure
... A useful idealization known as incompressible fluid is employed in fluid mechanics for a sufficiently realistic model of liquid behavior The volume expansion and isothermal compression coefficient of the incompressible fluid are zero so it cannot be described by an equation of state relating V to T ...
... A useful idealization known as incompressible fluid is employed in fluid mechanics for a sufficiently realistic model of liquid behavior The volume expansion and isothermal compression coefficient of the incompressible fluid are zero so it cannot be described by an equation of state relating V to T ...
p250c13
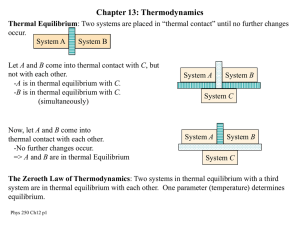
... The Zeroeth Law of Thermodynamics: Two systems in thermal equilibrium with a third system are in thermal equilibrium with each other. One parameter (temperature) determines equilibrium. Phys 250 Ch12 p1 ...
... The Zeroeth Law of Thermodynamics: Two systems in thermal equilibrium with a third system are in thermal equilibrium with each other. One parameter (temperature) determines equilibrium. Phys 250 Ch12 p1 ...
Examination Heat Transfer
... calculate then Nu L and express Nu L in Nu L (Nusselt at position x = L). c) Water at the rate of 68 kg/min is heated from 35 to 75 0C by an oil having a specific heat of 1.9 kJ/kg.0C. The oil enters the exchanger at 110 0C and leaves at 75 0C. The overall heat transfer coefficient is 320 W/m2.K. Th ...
... calculate then Nu L and express Nu L in Nu L (Nusselt at position x = L). c) Water at the rate of 68 kg/min is heated from 35 to 75 0C by an oil having a specific heat of 1.9 kJ/kg.0C. The oil enters the exchanger at 110 0C and leaves at 75 0C. The overall heat transfer coefficient is 320 W/m2.K. Th ...
Thermal Stability and Oxidation Processes
... Studying the resistance to oxidation in copper alloys is very important. For example, oxidative degradation can occur in these alloys as copper oxides form in atmospheres that are rich in oxygen. TGA can be used to study the static oxidation of materials such as these for practical use. ...
... Studying the resistance to oxidation in copper alloys is very important. For example, oxidative degradation can occur in these alloys as copper oxides form in atmospheres that are rich in oxygen. TGA can be used to study the static oxidation of materials such as these for practical use. ...
Topic 3
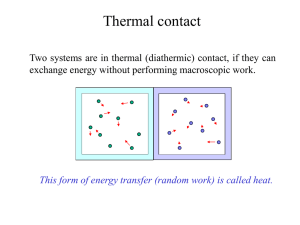
... Students should be familiar with the terms system and surroundings. They should also gas appreciate that if a system and its surroundings are at different temperatures and the system undergoes a process, the energy transferred by nonmechanical means to or from the system is referred to as thermal en ...
... Students should be familiar with the terms system and surroundings. They should also gas appreciate that if a system and its surroundings are at different temperatures and the system undergoes a process, the energy transferred by nonmechanical means to or from the system is referred to as thermal en ...
Changes of State
... WHY IS SALT SO STABLE? When salt is made, it forms a lattice, or a strong cube structure. Positive Sodium is attracted to negative Chlorine ...
... WHY IS SALT SO STABLE? When salt is made, it forms a lattice, or a strong cube structure. Positive Sodium is attracted to negative Chlorine ...
Thermal concepts - Uplift North Hills Prep
... • Students should be aware of the assumptions that underpin the molecular kinetic theory of ideal gases • Gas laws are limited to constant volume, constant temperature, constant pressure and the ideal gas law • Students should understand that a real gas approximates to an ideal gas at conditions of ...
... • Students should be aware of the assumptions that underpin the molecular kinetic theory of ideal gases • Gas laws are limited to constant volume, constant temperature, constant pressure and the ideal gas law • Students should understand that a real gas approximates to an ideal gas at conditions of ...
241 Lecture 11
... Calculate ΔU and ΔH for 1 kg of water when it is vaporized at the constant temperature of 100 °C and the constant pressure of 101.33 kPa. The specific volumes of liquid and vapor water at these conditions are 0.00104 and 1.673 m3/kg. For this change, heat in the amount of 2256.9 kJ is added to the ...
... Calculate ΔU and ΔH for 1 kg of water when it is vaporized at the constant temperature of 100 °C and the constant pressure of 101.33 kPa. The specific volumes of liquid and vapor water at these conditions are 0.00104 and 1.673 m3/kg. For this change, heat in the amount of 2256.9 kJ is added to the ...
Modulated Thermomechanical Analysis
... Reading introduced modulated temperature thermal analysis in 1993 (1) with what has become known as modulated differential scanning calorimetry (Modulated DSC®, MDSC®). This was followed a few years later by Blaine who reported on modulated thermogravimetry (MTGA™) (2) and by Price with modulated th ...
... Reading introduced modulated temperature thermal analysis in 1993 (1) with what has become known as modulated differential scanning calorimetry (Modulated DSC®, MDSC®). This was followed a few years later by Blaine who reported on modulated thermogravimetry (MTGA™) (2) and by Price with modulated th ...
Improved Temperature Determination from
... On macroscopic length scales temperature measurements are made using thermocouples, using optical pyrometers, and by detecting infrared radiation. These methods are not scalable to microelectronic device scales, such as the 22 nm gate width in modern transistors. Temperature measurements at these le ...
... On macroscopic length scales temperature measurements are made using thermocouples, using optical pyrometers, and by detecting infrared radiation. These methods are not scalable to microelectronic device scales, such as the 22 nm gate width in modern transistors. Temperature measurements at these le ...
slides - Biology Courses Server
... BUT, we can define the value of w (or q) for a specific process linking two states to be a change in a state function. We define the work for the reversible (infinitely slow) conversion of one state to the another, wrev , to be the change in state function ∆F . • ∆F is called the change in “free ene ...
... BUT, we can define the value of w (or q) for a specific process linking two states to be a change in a state function. We define the work for the reversible (infinitely slow) conversion of one state to the another, wrev , to be the change in state function ∆F . • ∆F is called the change in “free ene ...