BCJ0205-15 Thermal phenomena (3-1-4)
... Week 4: Heat and work in thermodynamic processes. Path between thermodynamic states and the first law of thermodynamics. Week 5: Ideal gases: microscopic interpretation; work performed by an ideal gas. Week 6: Idea gases: molar heat capacity, processes involving an ideal gas, adiabatic transformatio ...
... Week 4: Heat and work in thermodynamic processes. Path between thermodynamic states and the first law of thermodynamics. Week 5: Ideal gases: microscopic interpretation; work performed by an ideal gas. Week 6: Idea gases: molar heat capacity, processes involving an ideal gas, adiabatic transformatio ...
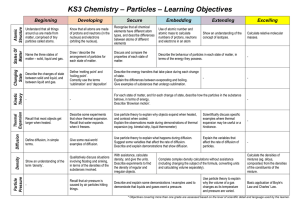
Particles - Townley Grammar School
... Explain the differences between evaporating and boiling. Give examples of substances that undergo sublimation. ...
... Explain the differences between evaporating and boiling. Give examples of substances that undergo sublimation. ...
Lecture Notes
... where k is the thermal conductivity of the material. It has units as W/m.K. Metals have k values in the range 20-400, ceramics 2-50, while polymers have in order of 0.3. Heat energy in solids in transported by two mechanisms: lattice vibrations (phonons) and free electrons. However, usually only one ...
... where k is the thermal conductivity of the material. It has units as W/m.K. Metals have k values in the range 20-400, ceramics 2-50, while polymers have in order of 0.3. Heat energy in solids in transported by two mechanisms: lattice vibrations (phonons) and free electrons. However, usually only one ...
Measurement Of Thermal Conductivity Using Thermal Comparator
... * Now a days, the thermal conductivity value for materials like Iron, Aluminium, Copper, etc., can be taken from the data book. * But for composite materials there is no ...
... * Now a days, the thermal conductivity value for materials like Iron, Aluminium, Copper, etc., can be taken from the data book. * But for composite materials there is no ...
Pyroelectric Effect. Primary Pyroelectricity. Secondary Pyroelectricity
... Independent of geometry, manufacture etc. Only a function of materials and temperature. ...
... Independent of geometry, manufacture etc. Only a function of materials and temperature. ...
Heat Chapter 12: Thermodynamics
... • As U goes, so goes T. isobaric – constant pressure • W = - pV On Gold Sheet isometric – constant volume • W = 0; Q = U adiabatic – no heat is exchanged • Q = 0; U = W • The area under the curve on a P-V graph is equal to work. • Internal energy is linked to temperature. Recall from Chapter 10, ...
... • As U goes, so goes T. isobaric – constant pressure • W = - pV On Gold Sheet isometric – constant volume • W = 0; Q = U adiabatic – no heat is exchanged • Q = 0; U = W • The area under the curve on a P-V graph is equal to work. • Internal energy is linked to temperature. Recall from Chapter 10, ...
met163_lecture_4
... In both cases the difference between expansion coefficients of two materials is used. Linear expansion is given by: ΔL=αL0ΔT Where α is the coefficient of linear expansion, L0 is the length of the ...
... In both cases the difference between expansion coefficients of two materials is used. Linear expansion is given by: ΔL=αL0ΔT Where α is the coefficient of linear expansion, L0 is the length of the ...
Homework 3
... Latent heat of fusion (to change water to ice at 0 C): Lice = 330 kJ kg To calculate how long the ice block will last, first determine the rate of heat transferred into the fridge ...
... Latent heat of fusion (to change water to ice at 0 C): Lice = 330 kJ kg To calculate how long the ice block will last, first determine the rate of heat transferred into the fridge ...
CHE 301 Problem set #3
... c. If the initial temperature of formalin solution is at 0oC, determine the time required for the reactor to reach 60oC for adiabatic operation. 4. We have a number of small jets that will be used in attitude control in space. These jets will be powered by low-pressure nitrogen gas heated by an arc ...
... c. If the initial temperature of formalin solution is at 0oC, determine the time required for the reactor to reach 60oC for adiabatic operation. 4. We have a number of small jets that will be used in attitude control in space. These jets will be powered by low-pressure nitrogen gas heated by an arc ...
Topic 2 The first law of thermodynamics
... The system is always infinitesimally close to equilibrium, and an infinitesimal change in conditions can reverse the process to restore both system and surrounding to their initial state. A reversible process is obviously an idealization Reversible isothermal process in a perfect gas ...
... The system is always infinitesimally close to equilibrium, and an infinitesimal change in conditions can reverse the process to restore both system and surrounding to their initial state. A reversible process is obviously an idealization Reversible isothermal process in a perfect gas ...
Tutorial II (thermodynamics)
... or false giving your thermodynamic reasoning. If the statement is false you may either state which law or laws of thermodynamics it violates or provide a physical counter example or any other plausible physical reason. Finally, correct the false statement with a clarifying phrase that makes the stat ...
... or false giving your thermodynamic reasoning. If the statement is false you may either state which law or laws of thermodynamics it violates or provide a physical counter example or any other plausible physical reason. Finally, correct the false statement with a clarifying phrase that makes the stat ...
Homework Assignment # 1
... 3. Two square plates are made out of the same homogeneous material, and both are initially heated to 100◦ . All four sides of the first plate are held at 0◦ , while for the second plate one of its sides is insulated and the other 3 held at 0◦ . Which plate cools down the fastest? How much faster? As ...
... 3. Two square plates are made out of the same homogeneous material, and both are initially heated to 100◦ . All four sides of the first plate are held at 0◦ , while for the second plate one of its sides is insulated and the other 3 held at 0◦ . Which plate cools down the fastest? How much faster? As ...
Plant development is strongly influenced by temperature. Other
... temperature surrounding plant. Temperature and time are integrated into thermal time which is defined as sum of temperatures above temperature at which development ceases which is called base temperature. Cardinal temperature at which is the rate of plant development highest is called optimal temper ...
... temperature surrounding plant. Temperature and time are integrated into thermal time which is defined as sum of temperatures above temperature at which development ceases which is called base temperature. Cardinal temperature at which is the rate of plant development highest is called optimal temper ...
1 Lecture: 2 Thermodynamic equilibrium 1
... This situation is called “Chemical Equilibrium” We will give more detailed description of the chemical equilibrium later on in this curse. A system that is in mechanical, thermal and chemical equilibrium at the same time is said to be in thermodynamic equilibrium. Functions of state When a system is ...
... This situation is called “Chemical Equilibrium” We will give more detailed description of the chemical equilibrium later on in this curse. A system that is in mechanical, thermal and chemical equilibrium at the same time is said to be in thermodynamic equilibrium. Functions of state When a system is ...
Some Physicochemical Properties of Yb MnSb and Its Solid Solutions with Gadolinium Yb
... where Ā- mean-square atomic weight; V- molecular volume; α- coefficient of thermal expansion. Both thermal expansion coefficients, melting temperatures and Debye temperatures evidences of changes after composition of gadolinium≈ ...
... where Ā- mean-square atomic weight; V- molecular volume; α- coefficient of thermal expansion. Both thermal expansion coefficients, melting temperatures and Debye temperatures evidences of changes after composition of gadolinium≈ ...