Classifying Chemical Reactions 9-3
... of element appearing on both sides Balance the atoms of an element one at a time by adding coefficients (the numbers in front) - save H and O until LAST! Check to make sure it is balanced. ...
... of element appearing on both sides Balance the atoms of an element one at a time by adding coefficients (the numbers in front) - save H and O until LAST! Check to make sure it is balanced. ...
Topic2890 Thermodynamics and Kinetics A given system at
... A given system at temperature T and pressure p is prepared using n1 moles of water (l) , the solvent, together with n 0X and n 0Y moles of chemical substances X and Y respectively at time ‘t = 0’. The molalities of these solutes are m 0X (= n 0X / n 1 ⋅ M 1 = n 0X / w 1 ) and m 0Y (= n 0Y / n 1 ⋅ M ...
... A given system at temperature T and pressure p is prepared using n1 moles of water (l) , the solvent, together with n 0X and n 0Y moles of chemical substances X and Y respectively at time ‘t = 0’. The molalities of these solutes are m 0X (= n 0X / n 1 ⋅ M 1 = n 0X / w 1 ) and m 0Y (= n 0Y / n 1 ⋅ M ...
The retrospect of the science and the thermodynamics
... Study the phase equilibriua behavior. Calculate the composition of a system at equilibrium. ...
... Study the phase equilibriua behavior. Calculate the composition of a system at equilibrium. ...
Reactive Materials - NC State University
... Do not combine nitric acid in unfamiliar combinations with organic compounds. ...
... Do not combine nitric acid in unfamiliar combinations with organic compounds. ...
Household Items That May Contain Mercury
... consequences can result. Consult a chemist who is knowledgeable in the manufacture of controlled substances for technical advice about shutting the operation down. Complex operations must usually be shut down in a specific sequence to avoid initiating an uncontrolled and ultimately violent reaction. ...
... consequences can result. Consult a chemist who is knowledgeable in the manufacture of controlled substances for technical advice about shutting the operation down. Complex operations must usually be shut down in a specific sequence to avoid initiating an uncontrolled and ultimately violent reaction. ...
Classification of Matter
... Properties of a solution Particles are not large enough to be seen ...
... Properties of a solution Particles are not large enough to be seen ...
STUDY GUIDE for DIGESTION and NUTRITION
... Understand what a chemical formula tells us about a compound Give the charge of an ion when an element gains or loses electrons Determine how many electrons an element will gain or lose Draw electron transfer diagrams of ionic compounds Use combining capacities to write formulas of ionic c ...
... Understand what a chemical formula tells us about a compound Give the charge of an ion when an element gains or loses electrons Determine how many electrons an element will gain or lose Draw electron transfer diagrams of ionic compounds Use combining capacities to write formulas of ionic c ...
File - Flipped Out Science with Mrs. Thomas!
... Chemical property – the ability of a substance to combine with or change into one or more other substances. Chemical symbol – a shorthand method of representing an element. Instead of writing out the name of an element, we represent an element name with one or two letters. Coefficient - the numbers ...
... Chemical property – the ability of a substance to combine with or change into one or more other substances. Chemical symbol – a shorthand method of representing an element. Instead of writing out the name of an element, we represent an element name with one or two letters. Coefficient - the numbers ...
File - Flipped Out Science with Mrs. Thomas!
... Chemical property – the ability of a substance to combine with or change into one or more other substances. Chemical symbol – a shorthand method of representing an element. Instead of writing out the name of an element, we represent an element name with one or two letters. Coefficient - the numbers ...
... Chemical property – the ability of a substance to combine with or change into one or more other substances. Chemical symbol – a shorthand method of representing an element. Instead of writing out the name of an element, we represent an element name with one or two letters. Coefficient - the numbers ...
Science 9
... What are the properties of ionic compounds? (list 2 properties, and what ionic compounds are made of) ...
... What are the properties of ionic compounds? (list 2 properties, and what ionic compounds are made of) ...
Notes for Types of Reactions:
... chemical reaction = the _________ by which one or more substances are __________ into one or more _________ substances. in any chemical reaction, the _________ substances are known as the reactants and the __________ substances are known as the products. total mass of reactants = according to ...
... chemical reaction = the _________ by which one or more substances are __________ into one or more _________ substances. in any chemical reaction, the _________ substances are known as the reactants and the __________ substances are known as the products. total mass of reactants = according to ...
Reading-Chem v Phys
... … Several occurrences are common indicators of a chemical change. Energy is usually absorbed or evolved in chemical reactions. Burning coal evolves heat; cooking food absorbs heat. Energy is also absorbed or evolved, however, in physical changes of state. A color change, as in leaves turning in the ...
... … Several occurrences are common indicators of a chemical change. Energy is usually absorbed or evolved in chemical reactions. Burning coal evolves heat; cooking food absorbs heat. Energy is also absorbed or evolved, however, in physical changes of state. A color change, as in leaves turning in the ...
CV and Seminar
... opportunity, and a major modeling and informatics challenge, for material design and discovery. Such a data deluge is coming from smart sensors in process plants, ab initio quantum calculations, molecular dynamics simulations, and so on. We are moving from an era of limited data obtained through tim ...
... opportunity, and a major modeling and informatics challenge, for material design and discovery. Such a data deluge is coming from smart sensors in process plants, ab initio quantum calculations, molecular dynamics simulations, and so on. We are moving from an era of limited data obtained through tim ...
Review Sheet
... b. Whenever you add a coefficient in front of a formula, remember that it affects the number of each atom in that formula. c. Sometimes an element is found in more than one compound on the same side of an equation, which can make it even more challenging to balance. d. There are times when you may h ...
... b. Whenever you add a coefficient in front of a formula, remember that it affects the number of each atom in that formula. c. Sometimes an element is found in more than one compound on the same side of an equation, which can make it even more challenging to balance. d. There are times when you may h ...
Document
... A vapor is the gaseous state of a substance that is generally a liquid or solid at room temperature (i.e.water vapor). ...
... A vapor is the gaseous state of a substance that is generally a liquid or solid at room temperature (i.e.water vapor). ...
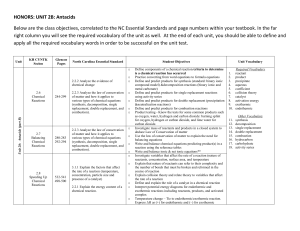
HONORS: UNIT 2B: Antacids Below are the class objectives
... Below are the class objectives, correlated to the NC Essential Standards and page numbers within your textbook. In the far right column you will see the required vocabulary of the unit as well. At the end of each unit, you should be able to define and apply all the required vocabulary words in order ...
... Below are the class objectives, correlated to the NC Essential Standards and page numbers within your textbook. In the far right column you will see the required vocabulary of the unit as well. At the end of each unit, you should be able to define and apply all the required vocabulary words in order ...
Chemical Reactions
... • Product – what is made during the chemical reaction • Law of conservation of mass (or matter)- mass is neither created or destroyed in ordinary chemical or physical changes • Subscript - number (representing atoms) written below and to the right of a chemical symbol ...
... • Product – what is made during the chemical reaction • Law of conservation of mass (or matter)- mass is neither created or destroyed in ordinary chemical or physical changes • Subscript - number (representing atoms) written below and to the right of a chemical symbol ...
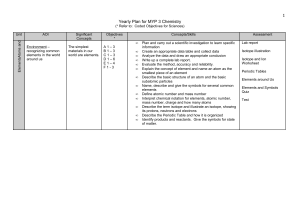
Yearly Plan for MYP 1 Science
... environment and that we cannot survive without them - recognizing common chemical reactions in our world - understanding what happens in a chemical change - noticing and identifying common chemicals we use in our everyday lives ...
... environment and that we cannot survive without them - recognizing common chemical reactions in our world - understanding what happens in a chemical change - noticing and identifying common chemicals we use in our everyday lives ...
Describing Chemical Reactions
... Of what? – chemical formulas In what state? – physical state Letters in parentheses indicate the physical state of each substance involved in the reaction (g) gas ; (l) liquid ; (s) solid ; (aq) aqueous solution ...
... Of what? – chemical formulas In what state? – physical state Letters in parentheses indicate the physical state of each substance involved in the reaction (g) gas ; (l) liquid ; (s) solid ; (aq) aqueous solution ...
CHEMISTRY EXAM 2 REVIEW
... 5. ductility property of a metal in which it can be stretched without breaking. 6. elasticity the ability of a substance to be stretched and then to return to its original shape. 7. endothermic reaction heat energy is absorbed in a chemical reaction; feels cold/cool 8. exothermic reaction heat energ ...
... 5. ductility property of a metal in which it can be stretched without breaking. 6. elasticity the ability of a substance to be stretched and then to return to its original shape. 7. endothermic reaction heat energy is absorbed in a chemical reaction; feels cold/cool 8. exothermic reaction heat energ ...
Chemical plant
A chemical plant is an industrial process plant that manufactures (or otherwise processes) chemicals, usually on a large scale. The general objective of a chemical plant is to create new material wealth via the chemical or biological transformation and or separation of materials. Chemical plants use specialized equipment, units, and technology in the manufacturing process. Other kinds of plants, such as polymer, pharmaceutical, food, and some beverage production facilities, power plants, oil refineries or other refineries, natural gas processing and biochemical plants, water and wastewater treatment, and pollution control equipment use many technologies that have similarities to chemical plant technology such as fluid systems and chemical reactor systems. Some would consider an oil refinery or a pharmaceutical or polymer manufacturer to be effectively a chemical plant.Petrochemical plants (plants using chemicals from petroleum as a raw material or feedstock ) are usually located adjacent to an oil refinery to minimize transportation costs for the feedstocks produced by the refinery. Speciality chemical and fine chemical plants are usually much smaller and not as sensitive to location. Tools have been developed for converting a base project cost from one geographic location to another.