Give reasons for the following: (i) Bond enthalpy of F2
... Helium mixed with oxygen under pressure is given to sea-divers for respiration. Air is not given to sea-divers because nitrogen present in air being soluble in blood will give a painful sensation called bends by bubbling out blood on moving from high pressure(in deep sea) to the atmospheric pressure ...
... Helium mixed with oxygen under pressure is given to sea-divers for respiration. Air is not given to sea-divers because nitrogen present in air being soluble in blood will give a painful sensation called bends by bubbling out blood on moving from high pressure(in deep sea) to the atmospheric pressure ...
MOLES, MASS, and VOLUME OF A GAS
... a) the mass of Oxygen needed; b) the volume of carbon Dioxide produced. ...
... a) the mass of Oxygen needed; b) the volume of carbon Dioxide produced. ...
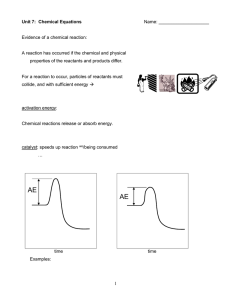
Chemical Equations
... that two molecules of hydrogen need to react for every 1 molecule of oxygen ...
... that two molecules of hydrogen need to react for every 1 molecule of oxygen ...
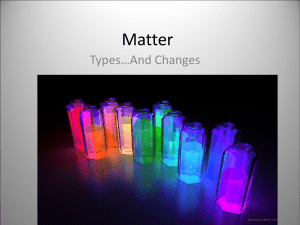
Matter- Types and Changes
... Chemical Formulas • CO2 contains 1 atom of carbon and two atoms of oxygen all chemically linked. • H2SO4 contains 2 hydrogen, 1 sulfur, and 4 oxygen atoms. • (NH4)2C2O4 - A subscript outside parentheses applies to everything within the parentheses; 2 N, 8 H, 2 C, 4 O ...
... Chemical Formulas • CO2 contains 1 atom of carbon and two atoms of oxygen all chemically linked. • H2SO4 contains 2 hydrogen, 1 sulfur, and 4 oxygen atoms. • (NH4)2C2O4 - A subscript outside parentheses applies to everything within the parentheses; 2 N, 8 H, 2 C, 4 O ...
Ms - cloudfront.net
... 18. Describe how a cation and an anion is formed. 19. What do metals typically do when they become ions? What about nonmetals? 20. What type of elements bond together in ionic bonds? covalent bonds? metallic bonds? 21. How do electrons in ionic bonding interact? Covalent bonding? 22. How does the re ...
... 18. Describe how a cation and an anion is formed. 19. What do metals typically do when they become ions? What about nonmetals? 20. What type of elements bond together in ionic bonds? covalent bonds? metallic bonds? 21. How do electrons in ionic bonding interact? Covalent bonding? 22. How does the re ...
Elements
... Generic form – referring to the atoms of element in various forms and combinations (e.g. the human body contains a lot of the element Oxygen) ...
... Generic form – referring to the atoms of element in various forms and combinations (e.g. the human body contains a lot of the element Oxygen) ...
AP Chemistry - Jackson County School System
... 82. Calculate the molarity of a solution that contains 0.0345 mol NH4Cl in exactly 400 ml of solution? 83. Calculate the molarity of a solution that contains 20.0grams of sodium hydroxide in 200ml? 84. How many grams of solute are present in 50.0 ml of 0.360 M sodium chloride? 85. The compound adren ...
... 82. Calculate the molarity of a solution that contains 0.0345 mol NH4Cl in exactly 400 ml of solution? 83. Calculate the molarity of a solution that contains 20.0grams of sodium hydroxide in 200ml? 84. How many grams of solute are present in 50.0 ml of 0.360 M sodium chloride? 85. The compound adren ...
South Pasadena · AP Chemistry
... stock 280 tables, 1750 chairs, 550 bookshelves, 300 china cabinets, and 325 sideboards. He asked his assistant to figure out how many dining room sets they could sell, how much money they would make if they sold all the sets possible, and what they would have left that could not be sold as part of t ...
... stock 280 tables, 1750 chairs, 550 bookshelves, 300 china cabinets, and 325 sideboards. He asked his assistant to figure out how many dining room sets they could sell, how much money they would make if they sold all the sets possible, and what they would have left that could not be sold as part of t ...
Science 9
... The cup should contain only salt. The water evaporated. Since a new substance was not formed, a physical change has taken place. ...
... The cup should contain only salt. The water evaporated. Since a new substance was not formed, a physical change has taken place. ...
chemical reaction - Peoria Public Schools
... A word equation is an equation in which the reactants and products in a chemical reaction are represented with words methane + oxygen → carbon dioxide + water ...
... A word equation is an equation in which the reactants and products in a chemical reaction are represented with words methane + oxygen → carbon dioxide + water ...
students - Teach-n-Learn-Chem
... Hint: Start with most complicated substances first and leave simplest substances for last. solid sodium reacts w/oxygen to form solid sodium oxide ...
... Hint: Start with most complicated substances first and leave simplest substances for last. solid sodium reacts w/oxygen to form solid sodium oxide ...
Exam Review
... 5. Classify each of the following reactions as synthesis, single displacement, double displacement, combustion or decomposition. a) iron + copper(I) nitrate iron(II) nitrate + copper (SD) b) phosphorus + oxygen diphosphorus pentoxide (S) c) calcium carbonate calcium oxide + carbon dioxide (D) ...
... 5. Classify each of the following reactions as synthesis, single displacement, double displacement, combustion or decomposition. a) iron + copper(I) nitrate iron(II) nitrate + copper (SD) b) phosphorus + oxygen diphosphorus pentoxide (S) c) calcium carbonate calcium oxide + carbon dioxide (D) ...
File
... It’s time to practice what you have already learned about moles, chemical reactions and dimensional analysis. We will learn one new conversion factor and then combine it with other concepts. Molar Volume is the volume of one mole of gas. Since the space between molecules in a gas is very great compa ...
... It’s time to practice what you have already learned about moles, chemical reactions and dimensional analysis. We will learn one new conversion factor and then combine it with other concepts. Molar Volume is the volume of one mole of gas. Since the space between molecules in a gas is very great compa ...
inorganic chemistry
... electronegative due to their high effective nuclear charge. They can gain an electron by reacting with atoms of other elements. Fluorine is one of the most reactive elements in existence, attacking otherwise inert materials such as glass, and forming compounds with the heavier noble gases. It is a c ...
... electronegative due to their high effective nuclear charge. They can gain an electron by reacting with atoms of other elements. Fluorine is one of the most reactive elements in existence, attacking otherwise inert materials such as glass, and forming compounds with the heavier noble gases. It is a c ...
Class Notes
... Slime is a Unique Material Slime is a unique POLYMER because it has qualities of both a solid and a liquid. It can take the shape of its containers like a liquid does, yet you can hold it in your hand and pick it up like a solid. As you might know, solid molecules are tight together, liquid molecul ...
... Slime is a Unique Material Slime is a unique POLYMER because it has qualities of both a solid and a liquid. It can take the shape of its containers like a liquid does, yet you can hold it in your hand and pick it up like a solid. As you might know, solid molecules are tight together, liquid molecul ...
educator exam series
... Mathematical tables and electronic calculations may be used All working MUST be clearly shown where necessary For examiner’s use only: Questions Max. score Candidates score ...
... Mathematical tables and electronic calculations may be used All working MUST be clearly shown where necessary For examiner’s use only: Questions Max. score Candidates score ...
AP Chemistry Summer Assignment
... Answer each of the following questions and give an example that helps with your explanation. 1. If a compound ends in –ide, what does it tell you about the compound? 2. If a compound ends in –ate what does it tell you about the compound? 3. If a compound ends in –ite what does it tell you about the ...
... Answer each of the following questions and give an example that helps with your explanation. 1. If a compound ends in –ide, what does it tell you about the compound? 2. If a compound ends in –ate what does it tell you about the compound? 3. If a compound ends in –ite what does it tell you about the ...
CfE Higher Chemistry Homework 3.5
... To avoid these contaminants, hydrogen sulfide can be made by reacting aluminium sulfide with water. Hydrogen sulfide and aluminium hydroxide are produced. Write a balanced chemical equation for the production of hydrogen sulfide from aluminium sulfide and water. ...
... To avoid these contaminants, hydrogen sulfide can be made by reacting aluminium sulfide with water. Hydrogen sulfide and aluminium hydroxide are produced. Write a balanced chemical equation for the production of hydrogen sulfide from aluminium sulfide and water. ...
AP Chemistry Summer Assignment
... 63. A 2.0g sample of SX6 (g) has a volume of 329.5 cm3 at 1.00 atm and 20oC. Identify the element ‘X’. Name the compound. ...
... 63. A 2.0g sample of SX6 (g) has a volume of 329.5 cm3 at 1.00 atm and 20oC. Identify the element ‘X’. Name the compound. ...
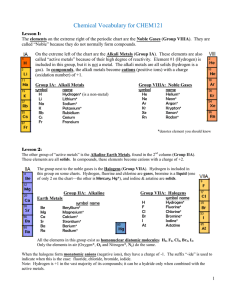
Vocabulary CHEM121
... Metals can only form cations. Non-metals form anions when combined with metals. Elements that touch the line are called metalloids (except Al, which is a metal). Oxygen combines with all elements except Noble Gases. Oxygen usually forms the oxide (O2-) ion in compounds, but also may form a hom ...
... Metals can only form cations. Non-metals form anions when combined with metals. Elements that touch the line are called metalloids (except Al, which is a metal). Oxygen combines with all elements except Noble Gases. Oxygen usually forms the oxide (O2-) ion in compounds, but also may form a hom ...
balancing chemical equations worksheet
... The following questions relate to these four steps. a. What symbols should we use to describe the physical states? b. Chemists and other scientists always balance chemical equations. Please explain why this is so important. (Hint, refer to the law of conservation of mass) PART B, read the following ...
... The following questions relate to these four steps. a. What symbols should we use to describe the physical states? b. Chemists and other scientists always balance chemical equations. Please explain why this is so important. (Hint, refer to the law of conservation of mass) PART B, read the following ...
Industrial Chemicals Technology Hand Book
... Growth in demand for chemicals in developing countries is high leading to substantial cross border investment in the chemical sector. The chemical industry comprises the companies that produce industrial chemicals. Chemicals are used to make a wide variety of consumer goods, as well as thousands inp ...
... Growth in demand for chemicals in developing countries is high leading to substantial cross border investment in the chemical sector. The chemical industry comprises the companies that produce industrial chemicals. Chemicals are used to make a wide variety of consumer goods, as well as thousands inp ...
activity series
... 3. All binary compounds of the halogens (other than F) with metals are soluble, except those of Ag, Hg (I), and Pb. (Pb halides are soluble in hot water.) ...
... 3. All binary compounds of the halogens (other than F) with metals are soluble, except those of Ag, Hg (I), and Pb. (Pb halides are soluble in hot water.) ...
Name……………………………………............................. Index number
... Write your name, index number class and admission number in the spaces provided Sign and write the date of examination in the spaces provided. Answer all the questions in the spaces provided. Mathematical tables and silent electronic calculators may be used. All working must be clearly shown where n ...
... Write your name, index number class and admission number in the spaces provided Sign and write the date of examination in the spaces provided. Answer all the questions in the spaces provided. Mathematical tables and silent electronic calculators may be used. All working must be clearly shown where n ...
Fluorochemical industry
The global market for chemicals from fluorine was about US$16 billion per year as of 2006. The industry was predicted to reach 2.6 million metric tons per year by 2015. The largest market is the United States. Western Europe is the second largest. Asia Pacific is the fastest growing region of production. China in particular has experienced significant growth as a fluorochemical market and is becoming a producer of them as well. Fluorite mining (the main source of fluorine) was estimated in 2003 to be a $550 million industry, extracting 4.5 million tons per year.Mined fluorite is separated into two main grades, with about equal production of each. Acidspar is at least 97% CaF2; metspar is much lower purity, 60–85%. (A small amount of the intermediate, ceramic, grade is also made.) Metspar is used almost exclusively for iron smelting. Acidspar is primarily converted to hydrofluoric acid (by reaction with sulfuric acid). The resultant HF is mostly used to produce organofluorides and synthetic cryolite.