What is Matter PowerPoint
... • Physical combinations of two or more substances (no chemical reactions occur) • Mixtures can be either homogeneous (same throughout) or heterogeneous (different) ...
... • Physical combinations of two or more substances (no chemical reactions occur) • Mixtures can be either homogeneous (same throughout) or heterogeneous (different) ...
Sample Questions
... The above equation is properly balanced when: 23. Give (in order) the correct coefficients to balance the following reaction: H2SnCl6 + H2S SnS2 + HCl 24. A reaction occurs between sodium carbonate and hydrochloric acid producing sodium chloride, carbon dioxide, and water. The correct set of coeff ...
... The above equation is properly balanced when: 23. Give (in order) the correct coefficients to balance the following reaction: H2SnCl6 + H2S SnS2 + HCl 24. A reaction occurs between sodium carbonate and hydrochloric acid producing sodium chloride, carbon dioxide, and water. The correct set of coeff ...
Study Guide – Unit Test (9-27-13)
... grams of vinegar is combined, the amount of liquid will decrease because the chemical reaction between the two substances creates CO2 (Carbon Dioxide gas), however the mass inside the container is still 16 grams. You cannot see the gas, but the matter was not destroyed. ...
... grams of vinegar is combined, the amount of liquid will decrease because the chemical reaction between the two substances creates CO2 (Carbon Dioxide gas), however the mass inside the container is still 16 grams. You cannot see the gas, but the matter was not destroyed. ...
+ O2 (g)
... Combustion of Methane, Balanced To show the reaction obeys the Law of Conservation of Mass, the equation must be balanced. CH4(g) + O2(g) ➜ CO2(g) + H2O(g) ! ...
... Combustion of Methane, Balanced To show the reaction obeys the Law of Conservation of Mass, the equation must be balanced. CH4(g) + O2(g) ➜ CO2(g) + H2O(g) ! ...
CHEMISTRY EXAM 2 REVIEW
... My child completed this review and studied for at least 30 minutes. Define the following chemistry terms: [Chemistry Dictionary] 1. alloy a mixture of metals 2. brittleness the property of matter that is how easily the substance breaks or shatters when force is applied to it. 3. compound a substance ...
... My child completed this review and studied for at least 30 minutes. Define the following chemistry terms: [Chemistry Dictionary] 1. alloy a mixture of metals 2. brittleness the property of matter that is how easily the substance breaks or shatters when force is applied to it. 3. compound a substance ...
2009-10 Chemistry 1st Semester Final Exam Topics and Review
... e. One of the problems with space travel is the building up of carbon dioxide produced by the astronauts. The typical procedure is to react the carbon dioxide with lithium hydroxide to form lithium carbonate and liquid water. 36. Write and balance a chemical equation for this reaction. a. Aluminum m ...
... e. One of the problems with space travel is the building up of carbon dioxide produced by the astronauts. The typical procedure is to react the carbon dioxide with lithium hydroxide to form lithium carbonate and liquid water. 36. Write and balance a chemical equation for this reaction. a. Aluminum m ...
1 - Montville.net
... THIS ASSIGNMENT IS DUE ON THE FIRST DAY OF SCHOOL IN SEPTEMBER. For mathematical problems, you must show how the problem is set up. CIRCLE ALL MATHEMATICAL ANSWERS! ...
... THIS ASSIGNMENT IS DUE ON THE FIRST DAY OF SCHOOL IN SEPTEMBER. For mathematical problems, you must show how the problem is set up. CIRCLE ALL MATHEMATICAL ANSWERS! ...
AP Chemistry Summer Assignment THIS
... THIS ASSIGNMENT IS DUE ON THE FIRST DAY OF SCHOOL IN SEPTEMBER. For mathematical problems, you must show how the problem is set up. CIRCLE ALL MATHEMATICAL ANSWERS! ...
... THIS ASSIGNMENT IS DUE ON THE FIRST DAY OF SCHOOL IN SEPTEMBER. For mathematical problems, you must show how the problem is set up. CIRCLE ALL MATHEMATICAL ANSWERS! ...
Chemicals: What`s in? What`s out?
... the first thing students want to know is, “Will we be blowing anything up?” Chemistry should be fun and exciting, but much preparation and skill are needed by the teacher and students in working with chemicals. Unfortunately, accidents do happen and things can blow up, but you can help prevent these ...
... the first thing students want to know is, “Will we be blowing anything up?” Chemistry should be fun and exciting, but much preparation and skill are needed by the teacher and students in working with chemicals. Unfortunately, accidents do happen and things can blow up, but you can help prevent these ...
Honors Chemistry Review Packet KEY
... 6. Liquids and gases both have an indefinite shape; while the shape of a solid is definite, the shape of a liquid is indefinite. 7. It is reversible because solid mercury can be melted back into a liquid again. 8. Platinum and copper can have the same mass and volume (extensive properties). They can ...
... 6. Liquids and gases both have an indefinite shape; while the shape of a solid is definite, the shape of a liquid is indefinite. 7. It is reversible because solid mercury can be melted back into a liquid again. 8. Platinum and copper can have the same mass and volume (extensive properties). They can ...
SNC2D – Science 10 Tuesday April 26th, 2010 Mr. Sourlis and Mr
... 6. What is the correct chemical formula for Calcium Nitride? a. CaN b. Ca2N c. Ca2N3 d. Ca3N2 e. Ca(NO3)2 7. Which of the following metals does NOT have more than one possible ion charge? a. Mn b. Ag c. Fe d. Cu e. Sn 8. What is the name of the compound PbSe? a. Lead (II) Selenide b. Lead (IV) Selen ...
... 6. What is the correct chemical formula for Calcium Nitride? a. CaN b. Ca2N c. Ca2N3 d. Ca3N2 e. Ca(NO3)2 7. Which of the following metals does NOT have more than one possible ion charge? a. Mn b. Ag c. Fe d. Cu e. Sn 8. What is the name of the compound PbSe? a. Lead (II) Selenide b. Lead (IV) Selen ...
1st Semester Exam in High School Chemistry
... Boyle was the first to use phosphorus to ignite sulfur tipped wooden splints, forerunners of our modern matches, in 1680. One of the ways in which it can be made involves the fusion of calcium phosphate with carbon and sand in an electric furnace. 2Ca3(PO4)2 + 6SiO2 + 10C → 6CaSiO3 + P4 + 10CO Start ...
... Boyle was the first to use phosphorus to ignite sulfur tipped wooden splints, forerunners of our modern matches, in 1680. One of the ways in which it can be made involves the fusion of calcium phosphate with carbon and sand in an electric furnace. 2Ca3(PO4)2 + 6SiO2 + 10C → 6CaSiO3 + P4 + 10CO Start ...
Welcome to AP Chemistry!
... a. Write a balanced chemical equation for this reaction. b. How many grams of calcium oxide will be produced after 12.25 g of calcium carbonate is completely decomposed? c. What volume of carbon dioxide gas is produced from this amount of calcium carbonate, at STP? 4) Hydrogen gas and bromine gas re ...
... a. Write a balanced chemical equation for this reaction. b. How many grams of calcium oxide will be produced after 12.25 g of calcium carbonate is completely decomposed? c. What volume of carbon dioxide gas is produced from this amount of calcium carbonate, at STP? 4) Hydrogen gas and bromine gas re ...
groups (families) vs rows
... Quantitative analysis shows that a compound contains 32.38% sodium, 22.65% sulfur and 44.99% oxygen. Find the empirical formula of this compound. ...
... Quantitative analysis shows that a compound contains 32.38% sodium, 22.65% sulfur and 44.99% oxygen. Find the empirical formula of this compound. ...
Document
... is shown above. What are the coefficients when this equation is balanced? F 2,2,1 G 2,3,1 H 4,3,2 J 4,2,2 ...
... is shown above. What are the coefficients when this equation is balanced? F 2,2,1 G 2,3,1 H 4,3,2 J 4,2,2 ...
Introductory Chemistry Test Review
... 25. In the laboratory, potassium chlorate will decompose when heated to form potassium chloride and oxygen gas according to the following equation. Calculate how much oxygen in grams is produced when 35.0 grams of potassium chlorate decomposes. 2 KClO3(s) ...
... 25. In the laboratory, potassium chlorate will decompose when heated to form potassium chloride and oxygen gas according to the following equation. Calculate how much oxygen in grams is produced when 35.0 grams of potassium chlorate decomposes. 2 KClO3(s) ...
Chapter 6-student notes
... chemical reaction occurs in which a gas is produced. If the mass of the final mixture is 85g, what mass of gas was produced? ...
... chemical reaction occurs in which a gas is produced. If the mass of the final mixture is 85g, what mass of gas was produced? ...
NAME
... i) lithium hydroxide pellets are added to a solution of sulfuric acid, lithium sulfate and water are formed. ...
... i) lithium hydroxide pellets are added to a solution of sulfuric acid, lithium sulfate and water are formed. ...
Unit 3 Practice Test
... 25. CH4, the first member of Group IVA hydrogen compounds, does not show the reversal in trend because CH4 is A. nonpolar and has only van der Waals forces C. nonpolar and has only London dispersion force ...
... 25. CH4, the first member of Group IVA hydrogen compounds, does not show the reversal in trend because CH4 is A. nonpolar and has only van der Waals forces C. nonpolar and has only London dispersion force ...
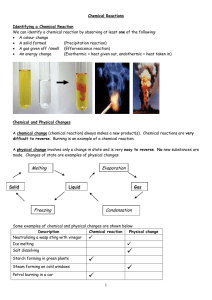
Chemical Reactions
... The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be an arrow in the middle. Examples: When sodium is mixed with water, a purple alkaline solution of sodium hydroxide is produced and hydrogen gas is evolved. Sodium ...
... The reactants are separated from each other by a plus sign and the products are separated from each other by a plus sign. There should be an arrow in the middle. Examples: When sodium is mixed with water, a purple alkaline solution of sodium hydroxide is produced and hydrogen gas is evolved. Sodium ...
Oxidation Number Rules
... a. Fluorine, the most electronegative element, is -1 in all fluorine containing compounds. b. In most oxygen containing compounds oxygen is -2. In peroxides (i.e. H2O2) each oxygen has an oxidation number of -1. In the compound OF2, the oxygen atom has an oxidation number of +2. c. Hydrogen usually ...
... a. Fluorine, the most electronegative element, is -1 in all fluorine containing compounds. b. In most oxygen containing compounds oxygen is -2. In peroxides (i.e. H2O2) each oxygen has an oxidation number of -1. In the compound OF2, the oxygen atom has an oxidation number of +2. c. Hydrogen usually ...
practice-exam-2
... 15. [7 points] Which of the following equations is the balanced chemical equation representing the combustion of propanol (C3H7OH)? (a) C3H7OH (l) + 9O(g) � 3CO2(g) + 4H2O(l) (b) C3H7OH (l) � 3CO2(g) + 4H2O(l) (c) C3H7OH (l) � CO(g) + 4H2(g) + 2C(s) (d) 2C3H7OH (l) + 9O2(g) � 6CO2(g) + 8H2O(l) (e) C ...
... 15. [7 points] Which of the following equations is the balanced chemical equation representing the combustion of propanol (C3H7OH)? (a) C3H7OH (l) + 9O(g) � 3CO2(g) + 4H2O(l) (b) C3H7OH (l) � 3CO2(g) + 4H2O(l) (c) C3H7OH (l) � CO(g) + 4H2(g) + 2C(s) (d) 2C3H7OH (l) + 9O2(g) � 6CO2(g) + 8H2O(l) (e) C ...
NAME…………… - Kcse Online
... Describe how the following reagents can be used to prepare calcium sulphate. Solid potassium sulphate, solid calcium carbonate, dilute nitric (V) acid and distilled water. (2mks) ___________________________________________________________________________ _____________________________________________ ...
... Describe how the following reagents can be used to prepare calcium sulphate. Solid potassium sulphate, solid calcium carbonate, dilute nitric (V) acid and distilled water. (2mks) ___________________________________________________________________________ _____________________________________________ ...
Fluorochemical industry
The global market for chemicals from fluorine was about US$16 billion per year as of 2006. The industry was predicted to reach 2.6 million metric tons per year by 2015. The largest market is the United States. Western Europe is the second largest. Asia Pacific is the fastest growing region of production. China in particular has experienced significant growth as a fluorochemical market and is becoming a producer of them as well. Fluorite mining (the main source of fluorine) was estimated in 2003 to be a $550 million industry, extracting 4.5 million tons per year.Mined fluorite is separated into two main grades, with about equal production of each. Acidspar is at least 97% CaF2; metspar is much lower purity, 60–85%. (A small amount of the intermediate, ceramic, grade is also made.) Metspar is used almost exclusively for iron smelting. Acidspar is primarily converted to hydrofluoric acid (by reaction with sulfuric acid). The resultant HF is mostly used to produce organofluorides and synthetic cryolite.