end of year review
... _____7. Two compounds that contain the elements carbon and chlorine are carbon tetrachloride (CCl4) and chloroform (CHCl3). Which of the following statements describes the geometry around carbon in these two compounds? A. CCl4 and CHCl3 have bent geometries. B. CCl4 and CHCl3 have tetrahedral geomet ...
... _____7. Two compounds that contain the elements carbon and chlorine are carbon tetrachloride (CCl4) and chloroform (CHCl3). Which of the following statements describes the geometry around carbon in these two compounds? A. CCl4 and CHCl3 have bent geometries. B. CCl4 and CHCl3 have tetrahedral geomet ...
Chemistry Final Exam Review 2006-2007
... nature as a diatomic molecule? b. 3 a. Nitrogen c. 4 b. Helium d. 2 c. Hydrogen 11. In the correct Lewis structure for the methane d. oxygen molecule, how many unshared electron pairs 2. Ionic compounds generally form: surround the carbon? a. Liquids a. 2 b. Gases b. 0 c. Crystals c. 8 d. molecules ...
... nature as a diatomic molecule? b. 3 a. Nitrogen c. 4 b. Helium d. 2 c. Hydrogen 11. In the correct Lewis structure for the methane d. oxygen molecule, how many unshared electron pairs 2. Ionic compounds generally form: surround the carbon? a. Liquids a. 2 b. Gases b. 0 c. Crystals c. 8 d. molecules ...
2007 - SolPass
... monoxide has one more atom of nitrogen. dioxide has one fewer atom of oxygen. monoxide has one fewer atom of oxygen. dioxide has one more atom of nitrogen. ...
... monoxide has one more atom of nitrogen. dioxide has one fewer atom of oxygen. monoxide has one fewer atom of oxygen. dioxide has one more atom of nitrogen. ...
Lecture 2 - Columbia University
... He established the Law of Conservation of Mass by burning phosphorus and sulfur in air, and proving that the products weighed more than the original. The weight gained was lost from the air. Repeating the experiments of Priestley, he demonstrated that air is composed of two parts, one of which combi ...
... He established the Law of Conservation of Mass by burning phosphorus and sulfur in air, and proving that the products weighed more than the original. The weight gained was lost from the air. Repeating the experiments of Priestley, he demonstrated that air is composed of two parts, one of which combi ...
Intro to Chem
... A chemical reaction (rxn) has two parts the reactants and the products. Reactant – substance present at the start of the rxn Product – substance produced in the rxn. H2 + O2 → 2H2O ...
... A chemical reaction (rxn) has two parts the reactants and the products. Reactant – substance present at the start of the rxn Product – substance produced in the rxn. H2 + O2 → 2H2O ...
Chapter 3 Part 2 Review
... Mass to mass stoichiometric relationships: Ex 1. The food we eat is degraded in our bodies to provide energy for growth and function. A general equation for this very complex process is: C6H12O6 + 6O2 6CO2 + 6H2O If 856 g of C6H12O6 is consumed by a person over a certain period, what is the mass ...
... Mass to mass stoichiometric relationships: Ex 1. The food we eat is degraded in our bodies to provide energy for growth and function. A general equation for this very complex process is: C6H12O6 + 6O2 6CO2 + 6H2O If 856 g of C6H12O6 is consumed by a person over a certain period, what is the mass ...
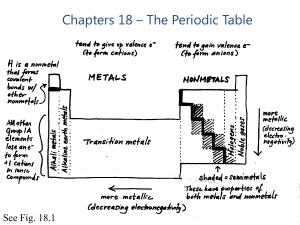
Chapters 18 – The Periodic Table
... agent such as potassium chlorate together with tetraphosphorus trisulfide (P4S3), glass and binder. The phosphorus sulfide is easily ignited, the potassium chlorate decomposes to give oxygen, which in turn causes the phosphorus sulfide to burn more vigorously. The head of safety matches are made of ...
... agent such as potassium chlorate together with tetraphosphorus trisulfide (P4S3), glass and binder. The phosphorus sulfide is easily ignited, the potassium chlorate decomposes to give oxygen, which in turn causes the phosphorus sulfide to burn more vigorously. The head of safety matches are made of ...
Chemistry I Exams and Answer Keys 2015 Season
... Magnesium metal reacts with hydrochloric acid forming a solution of magnesium chloride and hydrogen gas. When a small single piece of magnesium ribbon is dropped into a test tube filled with dilute hydrochloric acid, the metal soon floats to the surface of the liquid. The density of magnesium is 1.7 ...
... Magnesium metal reacts with hydrochloric acid forming a solution of magnesium chloride and hydrogen gas. When a small single piece of magnesium ribbon is dropped into a test tube filled with dilute hydrochloric acid, the metal soon floats to the surface of the liquid. The density of magnesium is 1.7 ...
end of year review
... _____19. The atomic theories of Dalton, Thomson, Rutherford, and Bohr all support which of the following statements? A. Atoms are mostly composed of empty space. B. All matter is composed of tiny, discrete particles called atoms. C. Electrons orbit the nucleus of an atom at distinct energy levels. D ...
... _____19. The atomic theories of Dalton, Thomson, Rutherford, and Bohr all support which of the following statements? A. Atoms are mostly composed of empty space. B. All matter is composed of tiny, discrete particles called atoms. C. Electrons orbit the nucleus of an atom at distinct energy levels. D ...
HIGHER TIER CHEMISTRY MINI-MOCK UNIT 2
... Calcium carbonate reacts with dilute hydrochloric acid as shown in the equation below. CaCO3(s) + 2HCl(aq) → CaCl2(aq) + H2O(l) + CO2(g) The rate at which this reaction takes place can be studied by measuring the amount of carbon dioxide gas produced. The graph below shows the results of four experi ...
... Calcium carbonate reacts with dilute hydrochloric acid as shown in the equation below. CaCO3(s) + 2HCl(aq) → CaCl2(aq) + H2O(l) + CO2(g) The rate at which this reaction takes place can be studied by measuring the amount of carbon dioxide gas produced. The graph below shows the results of four experi ...
A Low-Fluorine Solution with the F/Ba Mole Ratio of 2 for the
... Cu and Y salts [8, 9]; (3) Cu and Ba salts [10]. Relative to the conventional TFA-MOD solution (100% fluorine content), the fluorine contents in these precursor solutions could be estimated to be about 53.8%, 30.8% (or 23.1% if the poorBa stoichiometry is used [11]) and 23.1%, respectively. Using a ...
... Cu and Y salts [8, 9]; (3) Cu and Ba salts [10]. Relative to the conventional TFA-MOD solution (100% fluorine content), the fluorine contents in these precursor solutions could be estimated to be about 53.8%, 30.8% (or 23.1% if the poorBa stoichiometry is used [11]) and 23.1%, respectively. Using a ...
Physical and Chemical Changes
... C6H8O7 + 3NaHCO3 → 3H2O + 3CO2 + Na3C6H5O7 citric acid + baking soda → water + carbon dioxide + sodium citrate ...
... C6H8O7 + 3NaHCO3 → 3H2O + 3CO2 + Na3C6H5O7 citric acid + baking soda → water + carbon dioxide + sodium citrate ...
Unit 2
... Welcome to Advanced Placement Chemistry. AP Chem is a fast paced course, with higher orders of thinking. You will be expected to pull on previous knowledge constantly to solve problems. Along the way, we will see some fun demonstrations and perform some intense chemical experiments. The attached sum ...
... Welcome to Advanced Placement Chemistry. AP Chem is a fast paced course, with higher orders of thinking. You will be expected to pull on previous knowledge constantly to solve problems. Along the way, we will see some fun demonstrations and perform some intense chemical experiments. The attached sum ...
Unit 2
... Welcome to Advanced Placement Chemistry. AP Chem is a fast paced course, with higher orders of thinking. You will be expected to pull on previous knowledge constantly to solve problems. Along the way, we will see some fun demonstrations and perform some intense chemical experiments. The attached sum ...
... Welcome to Advanced Placement Chemistry. AP Chem is a fast paced course, with higher orders of thinking. You will be expected to pull on previous knowledge constantly to solve problems. Along the way, we will see some fun demonstrations and perform some intense chemical experiments. The attached sum ...
Practice Toxins Mid-Unit Test 08-09
... ______1. What type of reaction is this? Ag (s) + CuI2 (aq) AgI (s) + Cu(s) (A) single displacement (B) double displacement (C) combination reaction (D) decomposition reaction ______2.Calcium Chloride is abbreviated (A) CaCl (C) Ca2Cl (B) CaCl2 (D) Cl2Ca ______3. What is the molarity of 3.5 moles o ...
... ______1. What type of reaction is this? Ag (s) + CuI2 (aq) AgI (s) + Cu(s) (A) single displacement (B) double displacement (C) combination reaction (D) decomposition reaction ______2.Calcium Chloride is abbreviated (A) CaCl (C) Ca2Cl (B) CaCl2 (D) Cl2Ca ______3. What is the molarity of 3.5 moles o ...
Unit 8 Homework Packet
... 26. Although they were formerly called the inert gases, at least the heavier elements of Group 8 do form relatively stable compounds. For example, xenon combines directly with elemental fluorine at elevated temperatures in the presence of a nickel catalyst. Xe(g) + 2F2(g) → XeF4(s) ...
... 26. Although they were formerly called the inert gases, at least the heavier elements of Group 8 do form relatively stable compounds. For example, xenon combines directly with elemental fluorine at elevated temperatures in the presence of a nickel catalyst. Xe(g) + 2F2(g) → XeF4(s) ...
double-replacement reaction
... Combination Reactions • A combination reaction is a reaction in which simpler substances are combined into a more complex compound. • Combination reactions are also called synthesis reactions. • We will look at two combination reactions: 1. The reaction of a metal with nonmetal 2. The reaction of a ...
... Combination Reactions • A combination reaction is a reaction in which simpler substances are combined into a more complex compound. • Combination reactions are also called synthesis reactions. • We will look at two combination reactions: 1. The reaction of a metal with nonmetal 2. The reaction of a ...
Group 2 - UC Davis Canvas
... 11. The bond energy of the noble gas fluorine is too small to offset the energy required to break the F—F bond. 13. Iodide ion is slowly oxidized to iodine, which is yellow-brown in aqueous solution, by oxygen in the air: 4 I − ( aq ) + O 2 ( g ) + 4 H + ( aq ) → 2 I 2 ( aq ) + 2 H 2 O(l) . 15. D ...
... 11. The bond energy of the noble gas fluorine is too small to offset the energy required to break the F—F bond. 13. Iodide ion is slowly oxidized to iodine, which is yellow-brown in aqueous solution, by oxygen in the air: 4 I − ( aq ) + O 2 ( g ) + 4 H + ( aq ) → 2 I 2 ( aq ) + 2 H 2 O(l) . 15. D ...
Chemical Changes and Structure Homework Booklet
... 12Mg are two different kinds of magnesium atom. a. What word is used to describe these types of atoms? b. Explain why they can be regarded as atoms of the same element? c. The relative atomic mass of magnesium is 24.3. What does this tell you about the relative amounts of each atom? An atom has atom ...
... 12Mg are two different kinds of magnesium atom. a. What word is used to describe these types of atoms? b. Explain why they can be regarded as atoms of the same element? c. The relative atomic mass of magnesium is 24.3. What does this tell you about the relative amounts of each atom? An atom has atom ...
AP Chem -‐ Unit 1 Part 1 AP Chemistry 2016
... d. Chemistry majors sometimes get holes in their cotton jeans they wear to lab because of acid spills. Complete the following table: ...
... d. Chemistry majors sometimes get holes in their cotton jeans they wear to lab because of acid spills. Complete the following table: ...
AP Chem
... Can have a +1 and a +2 oxidation state. Has the largest atomic radius Has naturally radioactive isotopes used in smoke detectors. ...
... Can have a +1 and a +2 oxidation state. Has the largest atomic radius Has naturally radioactive isotopes used in smoke detectors. ...
Lab Stuff:
... What types of substances are removed from mixtures using filtration? Adsorption? Distillation? Think about the foul water lab! ...
... What types of substances are removed from mixtures using filtration? Adsorption? Distillation? Think about the foul water lab! ...
Chapter #2-Newest CPub
... • Matter is composed of indivisible atoms. • All atoms of a given chemical element are identical in mass and in all other properties. • Different chemical elements are composed of different atoms of different masses. • Atoms are indestructible. They retain their identities in a chemical reaction. • ...
... • Matter is composed of indivisible atoms. • All atoms of a given chemical element are identical in mass and in all other properties. • Different chemical elements are composed of different atoms of different masses. • Atoms are indestructible. They retain their identities in a chemical reaction. • ...
CO 2(g) - cloudfront.net
... • In a chemical reaction, all the atoms present at the beginning are still present at the end. If all the atoms are still there, then the mass will not change. ...
... • In a chemical reaction, all the atoms present at the beginning are still present at the end. If all the atoms are still there, then the mass will not change. ...
File
... NaOH is needed to titrate the sample to the equivalence point. What is the molar mass of the acid? A) 50.0 g B) 62.5 g C) 125 g D) 200 g E) 250 g 63. The pH of a solution prepared by adding 10.0 mL of 0.020 molar KOH (aq) to 10.0 mL of distilled water is closest to A) 13 B) 12 C) 11 D) 3 E) 2 64. Fa ...
... NaOH is needed to titrate the sample to the equivalence point. What is the molar mass of the acid? A) 50.0 g B) 62.5 g C) 125 g D) 200 g E) 250 g 63. The pH of a solution prepared by adding 10.0 mL of 0.020 molar KOH (aq) to 10.0 mL of distilled water is closest to A) 13 B) 12 C) 11 D) 3 E) 2 64. Fa ...
Fluorochemical industry
The global market for chemicals from fluorine was about US$16 billion per year as of 2006. The industry was predicted to reach 2.6 million metric tons per year by 2015. The largest market is the United States. Western Europe is the second largest. Asia Pacific is the fastest growing region of production. China in particular has experienced significant growth as a fluorochemical market and is becoming a producer of them as well. Fluorite mining (the main source of fluorine) was estimated in 2003 to be a $550 million industry, extracting 4.5 million tons per year.Mined fluorite is separated into two main grades, with about equal production of each. Acidspar is at least 97% CaF2; metspar is much lower purity, 60–85%. (A small amount of the intermediate, ceramic, grade is also made.) Metspar is used almost exclusively for iron smelting. Acidspar is primarily converted to hydrofluoric acid (by reaction with sulfuric acid). The resultant HF is mostly used to produce organofluorides and synthetic cryolite.