1 - cloudfront.net
... How is the number of neutrons in the nucleus of an atom calculated? All atoms are neutral, with the number of protons equaling the ___. Isotopes of the same element have different _____. Using the periodic table, determine the number of neutrons in 16O. What does the number 84 represent in the name ...
... How is the number of neutrons in the nucleus of an atom calculated? All atoms are neutral, with the number of protons equaling the ___. Isotopes of the same element have different _____. Using the periodic table, determine the number of neutrons in 16O. What does the number 84 represent in the name ...
Chapter 5
... Unlike charges attract and like charges repel Charges may be transferred by contact or induction The closer two object are, the greater the force of attraction ...
... Unlike charges attract and like charges repel Charges may be transferred by contact or induction The closer two object are, the greater the force of attraction ...
Beryllium isotopes in geochronology Cosmogenic Be and Be
... fission – the spontaneous (or induced by particle collision) splitting of a heavy nucleus into a pair (only rarely more) of nearly equal fission fragments (fission products) generally with some neutrons. Fission is accompanied by the release of a large quantity of energy. [return] gamma rays (gamma ...
... fission – the spontaneous (or induced by particle collision) splitting of a heavy nucleus into a pair (only rarely more) of nearly equal fission fragments (fission products) generally with some neutrons. Fission is accompanied by the release of a large quantity of energy. [return] gamma rays (gamma ...
Atomic Structure - hrsbstaff.ednet.ns.ca
... So, what’s up with all these isotopes anyway? In nature elements are not made up of atoms that are all exactly the same! Some will be heavier than others, even though they are still the same type of atom. C-12 and C-14 are both Carbon, with all the usual Carbon properties, but the C-14 has two more ...
... So, what’s up with all these isotopes anyway? In nature elements are not made up of atoms that are all exactly the same! Some will be heavier than others, even though they are still the same type of atom. C-12 and C-14 are both Carbon, with all the usual Carbon properties, but the C-14 has two more ...
Chemistry Review: Antoine Lavoisier (1743
... there are 3 isotopes of hydrogen, with mass numbers of 1, 2 and 3. These atoms have different masses because they have different numbers of neutrons. However, since they have the same number of protons, they are the same element. Most elements have more than 1 naturally occurring isotope. However, t ...
... there are 3 isotopes of hydrogen, with mass numbers of 1, 2 and 3. These atoms have different masses because they have different numbers of neutrons. However, since they have the same number of protons, they are the same element. Most elements have more than 1 naturally occurring isotope. However, t ...
Practice Test #2 - smhs
... 107.868 = 106.9041 (X) + 108.9047 (1.000 - X) -1.0367 = -2.0006 X X = 0.51819 ...
... 107.868 = 106.9041 (X) + 108.9047 (1.000 - X) -1.0367 = -2.0006 X X = 0.51819 ...
Chemistry lecture notes
... Isotopes have the same atomic number (same number of protons), but a different atomic mass number (a different number of neutrons). Isotopes behave the same chemically, because they are the same element. The only difference is that one is heavier than the other, because of the additional ...
... Isotopes have the same atomic number (same number of protons), but a different atomic mass number (a different number of neutrons). Isotopes behave the same chemically, because they are the same element. The only difference is that one is heavier than the other, because of the additional ...
Chapter 6 Review“The Periodic Table”
... 1. How is the number of neutrons in the nucleus of an atom calculated? 2. All atoms are neutral, with the number of protons equaling the ___. 3. Isotopes of the same element have different _____. 4. Using the periodic table, determine the number of neutrons in 16O. 5. What does the number 84 represe ...
... 1. How is the number of neutrons in the nucleus of an atom calculated? 2. All atoms are neutral, with the number of protons equaling the ___. 3. Isotopes of the same element have different _____. 4. Using the periodic table, determine the number of neutrons in 16O. 5. What does the number 84 represe ...
Practice Test #2 - smhs
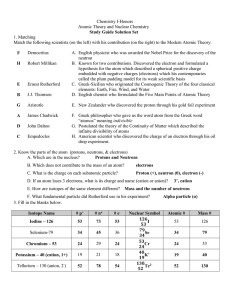
... 15._____ His oil-drop experiment enabled scientists to measure the charge on the electron. 16._____ He concluded that the atom had a small, compact, positively-charged nucleus surrounded by electrons based on his gold-foil experiment. 17._____ He invented the mass spectrograph, an instrument that is ...
... 15._____ His oil-drop experiment enabled scientists to measure the charge on the electron. 16._____ He concluded that the atom had a small, compact, positively-charged nucleus surrounded by electrons based on his gold-foil experiment. 17._____ He invented the mass spectrograph, an instrument that is ...
Intro to Atoms Clicker Questions 1. "atomos" means? 2. Atoms of one
... are located where? 6. Rutherford's proof of the proton's location in the atom came from an experiment with _______ 7. In the Bohr model of the atom, electrons are arranged how? 8. A neutron has (a) _____ charge 9. (T/F) The number of neutrons in an atom has to equal the number of protons. 10. (T/F) ...
... are located where? 6. Rutherford's proof of the proton's location in the atom came from an experiment with _______ 7. In the Bohr model of the atom, electrons are arranged how? 8. A neutron has (a) _____ charge 9. (T/F) The number of neutrons in an atom has to equal the number of protons. 10. (T/F) ...
MrsB-Chemistry
... Lesson 14: Isotopia (You will need the isotope graph handed out during lesson 14) 1. Would an atom with 50 protons and a mass of 155 amu be stable? ...
... Lesson 14: Isotopia (You will need the isotope graph handed out during lesson 14) 1. Would an atom with 50 protons and a mass of 155 amu be stable? ...
PS 2.2
... the weighted average of the masses of the naturally occurring isotopes of an element. The atomic mass of an element can be found on the periodic table. Since it is an average, it is usually not a whole number. ...
... the weighted average of the masses of the naturally occurring isotopes of an element. The atomic mass of an element can be found on the periodic table. Since it is an average, it is usually not a whole number. ...
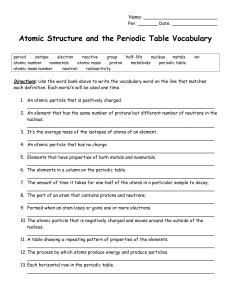
Atomic Structure and the Periodic Table Vocabulary
... 2. An element that has the same number of protons but different number of neutrons in the nucleus. __________________________________________________________________ 3. It’s the average mass of the isotopes of atoms of an element. __________________________________________________________________ 4. ...
... 2. An element that has the same number of protons but different number of neutrons in the nucleus. __________________________________________________________________ 3. It’s the average mass of the isotopes of atoms of an element. __________________________________________________________________ 4. ...
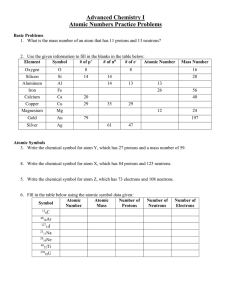
Atomic Numbers Practice Problems
... 3. Write the chemical symbol for atom Y, which has 27 protons and a mass number of 59. ...
... 3. Write the chemical symbol for atom Y, which has 27 protons and a mass number of 59. ...
The study of biology can help you better understand human
... Atoms of isotopes of an element have different number of protons The nucleus of an atom has positive charge. Atoms are mostly empty space. ...
... Atoms of isotopes of an element have different number of protons The nucleus of an atom has positive charge. Atoms are mostly empty space. ...
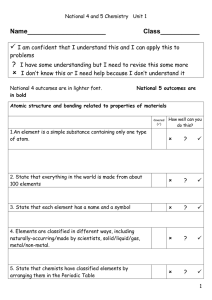
L.O.
... I have some understanding but I need to revise this some more I don’t know this or I need help because I don’t understand it ...
... I have some understanding but I need to revise this some more I don’t know this or I need help because I don’t understand it ...
Notes
... -the number of protons in an atom of an element •all atoms of an element have the same atomic # •written as a subscript next to the element’s symbol •in a neutral atom, the number of protons is equal to the number of electrons (balanced charges). ...
... -the number of protons in an atom of an element •all atoms of an element have the same atomic # •written as a subscript next to the element’s symbol •in a neutral atom, the number of protons is equal to the number of electrons (balanced charges). ...
Study Guide - Honors Chemistry
... multiple nuclei form a single product one nucleus is broken into multiple (2 in this case) nuclei by force (an alpha particle is used to break it up) one nucleus is broken into multiple (2 in this case) nuclei on its own. No force is needed. one nucleus is transformed into another nucleus by bombard ...
... multiple nuclei form a single product one nucleus is broken into multiple (2 in this case) nuclei by force (an alpha particle is used to break it up) one nucleus is broken into multiple (2 in this case) nuclei on its own. No force is needed. one nucleus is transformed into another nucleus by bombard ...
Review for Periodic - Mr-Durands
... 1. Find Scandium (Sc) on the table what is the atomic number? 2. What is the atomic mass of Chromium (Cr)? 3. What is the number of neutrons for Cesium (Cs)? 4. What is the difference between atomic mass and number? 5. How do isotopes affect the atomic mass of an element? 6. What is a group on the p ...
... 1. Find Scandium (Sc) on the table what is the atomic number? 2. What is the atomic mass of Chromium (Cr)? 3. What is the number of neutrons for Cesium (Cs)? 4. What is the difference between atomic mass and number? 5. How do isotopes affect the atomic mass of an element? 6. What is a group on the p ...
Pre-Knowledge: Chemistry and Physics Vocabulary Atomic Number
... become more stable by emitting radiation. Radioactive decay is the process in which an unstable atomic nucleus loses energy by emitting radiation in the form of particles or electromagnetic waves. This radiation can be emitted in the form of a positively charged alpha particle, a negatively charged ...
... become more stable by emitting radiation. Radioactive decay is the process in which an unstable atomic nucleus loses energy by emitting radiation in the form of particles or electromagnetic waves. This radiation can be emitted in the form of a positively charged alpha particle, a negatively charged ...
Einsteinium

Einsteinium is a synthetic element with symbol Es and atomic number 99. It is the seventh transuranic element, and an actinide.Einsteinium was discovered as a component of the debris of the first hydrogen bomb explosion in 1952, and named after Albert Einstein. Its most common isotope einsteinium-253 (half life 20.47 days) is produced artificially from decay of californium-253 in a few dedicated high-power nuclear reactors with a total yield on the order of one milligram per year. The reactor synthesis is followed by a complex process of separating einsteinium-253 from other actinides and products of their decay. Other isotopes are synthesized in various laboratories, but at much smaller amounts, by bombarding heavy actinide elements with light ions. Owing to the small amounts of produced einsteinium and the short half-life of its most easily produced isotope, there are currently almost no practical applications for it outside of basic scientific research. In particular, einsteinium was used to synthesize, for the first time, 17 atoms of the new element mendelevium in 1955.Einsteinium is a soft, silvery, paramagnetic metal. Its chemistry is typical of the late actinides, with a preponderance of the +3 oxidation state; the +2 oxidation state is also accessible, especially in solids. The high radioactivity of einsteinium-253 produces a visible glow and rapidly damages its crystalline metal lattice, with released heat of about 1000 watts per gram. Difficulty in studying its properties is due to einsteinium-253's conversion to berkelium and then californium at a rate of about 3% per day. The isotope of einsteinium with the longest half life, einsteinium-252 (half life 471.7 days) would be more suitable for investigation of physical properties, but it has proven far more difficult to produce and is available only in minute quantities, and not in bulk. Einsteinium is the element with the highest atomic number which has been observed in macroscopic quantities in its pure form, and this was the common short-lived isotope einsteinium-253.Like all synthetic transuranic elements, isotopes of einsteinium are very radioactive and are considered highly dangerous to health on ingestion.