Protons, electrons and neutrons worksheet
... Keep a periodic table handy to solve this puzzle. Atomic symbol is the symbol you find for each element shown in the periodic table. Magnesium symbol is Mg Gold symbol is Au Potassium symbol is K Phosphorous symbol is P Note: First letter of the element is not always the symbol. Atomic number is the ...
... Keep a periodic table handy to solve this puzzle. Atomic symbol is the symbol you find for each element shown in the periodic table. Magnesium symbol is Mg Gold symbol is Au Potassium symbol is K Phosphorous symbol is P Note: First letter of the element is not always the symbol. Atomic number is the ...
Atomic Structure_Tre..
... 5) Importance: The number of electrons located in the last energy level determine the chemical activity of the element. ...
... 5) Importance: The number of electrons located in the last energy level determine the chemical activity of the element. ...
ISOSTOPE NOTES - Mr. Collier`s 9th Grade Physical Science
... • Carbon will always have 6 protons How many neutrons does Carbon-14 have? there are 14 total protons and neutrons. 6 of them are protons. So 14 (total) – 6 (protons) = 8 Neutrons How many neutrons does Carbon-13 have? ...
... • Carbon will always have 6 protons How many neutrons does Carbon-14 have? there are 14 total protons and neutrons. 6 of them are protons. So 14 (total) – 6 (protons) = 8 Neutrons How many neutrons does Carbon-13 have? ...
ps-5-1-and-5-2-ws
... Moving across the periodic table from left to right, one finds, in order, metals, then gases, then nonmetals. ...
... Moving across the periodic table from left to right, one finds, in order, metals, then gases, then nonmetals. ...
800 - Paint Valley Local Schools
... considered radioactive because of its large, unstable nucleus. It was one of the fuels used to construct the early atomic bombs in the WWII era. ...
... considered radioactive because of its large, unstable nucleus. It was one of the fuels used to construct the early atomic bombs in the WWII era. ...
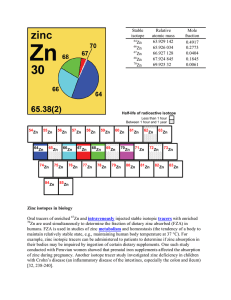
Zinc isotopes in biology Oral tracers of enriched Zn and
... anthropogenic – resulting from human activity. [return] atomic number (Z) – The number of protons in the nucleus of an atom. atomic weight (relative mean atomic mass) – the sum of the products of the relative atomic mass and the mole fraction of each stable and long-lived radioactive isotope of that ...
... anthropogenic – resulting from human activity. [return] atomic number (Z) – The number of protons in the nucleus of an atom. atomic weight (relative mean atomic mass) – the sum of the products of the relative atomic mass and the mole fraction of each stable and long-lived radioactive isotope of that ...
Atomic Structure
... • 1. All elements are composed of tiny indivisible particles called atoms. • 2. Atoms of the same element are identical. The atoms of one element are different from the atoms of another element. • 3. Atoms of different elements can physically mix together or can chemically combine in simplewhole num ...
... • 1. All elements are composed of tiny indivisible particles called atoms. • 2. Atoms of the same element are identical. The atoms of one element are different from the atoms of another element. • 3. Atoms of different elements can physically mix together or can chemically combine in simplewhole num ...
Chapter 3 notes
... • Atomic number- the number of protons in the nucleus of an atom. • Atomic mass- the total number of protons and neutrons in the nucleus of an atom. • Isotopes- any atoms having the same numbers of protons but a different number of neutrons. ...
... • Atomic number- the number of protons in the nucleus of an atom. • Atomic mass- the total number of protons and neutrons in the nucleus of an atom. • Isotopes- any atoms having the same numbers of protons but a different number of neutrons. ...
Atomic Structure - Learn District 196
... • Tells us the number of protons in the nucleus of an atom ...
... • Tells us the number of protons in the nucleus of an atom ...
Fall Semester Review Packet
... variable and a control. Describe how these variables relate to one another during an experiment. 12. Explain the difference between accuracy and precision when describing scientific measurements. 13. Explain how atoms of the same element may differ. Include all of the following terms in your explana ...
... variable and a control. Describe how these variables relate to one another during an experiment. 12. Explain the difference between accuracy and precision when describing scientific measurements. 13. Explain how atoms of the same element may differ. Include all of the following terms in your explana ...
Atoms, Molecules, and Ions Chapter 2 Handout 1 The Atom Dalton`s
... 1. Each element is composed of extremely small particles called atoms. 2. All atoms of a given element are identical to one another in mass and other properties, but the atoms of one element are different from the atoms of all the other elements. 3. Atoms are neither created nor destroyed in chem ...
... 1. Each element is composed of extremely small particles called atoms. 2. All atoms of a given element are identical to one another in mass and other properties, but the atoms of one element are different from the atoms of all the other elements. 3. Atoms are neither created nor destroyed in chem ...
chapter_3_study_guide
... model of the atom to include negatively charged particles called ______________________. note: (Later, a different Thompson named ___________________ Thompson reasoned that the atom might be a uniform positively charged "___________________" like form with electrons scattered around like the way rai ...
... model of the atom to include negatively charged particles called ______________________. note: (Later, a different Thompson named ___________________ Thompson reasoned that the atom might be a uniform positively charged "___________________" like form with electrons scattered around like the way rai ...
Name Date Class Period ______
... Name ______________________________________ Date __________________ Class Period _________ Atoms, Elements, and Compound Test Study Guide I. ...
... Name ______________________________________ Date __________________ Class Period _________ Atoms, Elements, and Compound Test Study Guide I. ...
Chapter 5 “Atomic Structure and the Periodic table”
... 2)Atoms of the same element are identical. Atoms of any one element are different from those of any other element. 3)Atoms of different elements combine in simple whole-number ratios to form chemical compounds 4)In chemical reactions, atoms are combined, separated, or rearranged – but never changed ...
... 2)Atoms of the same element are identical. Atoms of any one element are different from those of any other element. 3)Atoms of different elements combine in simple whole-number ratios to form chemical compounds 4)In chemical reactions, atoms are combined, separated, or rearranged – but never changed ...
Problem Set 4 - Morrisville.org
... 5) True False Questions – determine if the following statements are true or false according to Democritus’s ideas about atomos. a. Atomos are divisible. b. Atomos are hard dense spheres c. Atomos have varying density – they are heterogeneous. d. Changes in matter are due to the changes in atomos. 6) ...
... 5) True False Questions – determine if the following statements are true or false according to Democritus’s ideas about atomos. a. Atomos are divisible. b. Atomos are hard dense spheres c. Atomos have varying density – they are heterogeneous. d. Changes in matter are due to the changes in atomos. 6) ...
Notes - Science 2015-2016
... ▫ Protons and neutrons are in the _________ ? ▫ Electrons are located __________ ? ...
... ▫ Protons and neutrons are in the _________ ? ▫ Electrons are located __________ ? ...
Chapter 3
... • Law of Definite Proportions : • All compounds have the same proportion by mass for example: NaCl is always 66.66% chlorine and 39.34% sodium • Law of Multiple Proportions: when two elements can form two compounds, the masses that combine are in simple whole number ratios, CO and CO2 ...
... • Law of Definite Proportions : • All compounds have the same proportion by mass for example: NaCl is always 66.66% chlorine and 39.34% sodium • Law of Multiple Proportions: when two elements can form two compounds, the masses that combine are in simple whole number ratios, CO and CO2 ...
Chapter 3
... • Law of Definite Proportions : • All compounds have the same proportion by mass for example: NaCl is always 60.66% chlorine and 39.34% sodium • Law of Multiple Proportions: when two elements can form two compounds, the masses that combine are in simple whole number ratios, CO and CO2 ...
... • Law of Definite Proportions : • All compounds have the same proportion by mass for example: NaCl is always 60.66% chlorine and 39.34% sodium • Law of Multiple Proportions: when two elements can form two compounds, the masses that combine are in simple whole number ratios, CO and CO2 ...
Present - Images
... and the smallest thing there is) Atoms of agiven element have identical physical and chemical properties. (all atoms of X will These two are behave the same anywhere) usually combined Different atoms have different properties. (X behaves differently than Y) Atoms combine in whole-number ratios to fo ...
... and the smallest thing there is) Atoms of agiven element have identical physical and chemical properties. (all atoms of X will These two are behave the same anywhere) usually combined Different atoms have different properties. (X behaves differently than Y) Atoms combine in whole-number ratios to fo ...
Chemistry Worksheet: Atomic Structure and Isotopes
... 13. Write the name and symbol for the elements that are described below: a. 78 protons in the nucleus of each atom. b. 42 electrons in each uncharged atom. c. 18 electrons in each +2 cation. d. 10 electrons in each -1 anion. Identify the isotope in each of the following. Give the appropriate symbol ...
... 13. Write the name and symbol for the elements that are described below: a. 78 protons in the nucleus of each atom. b. 42 electrons in each uncharged atom. c. 18 electrons in each +2 cation. d. 10 electrons in each -1 anion. Identify the isotope in each of the following. Give the appropriate symbol ...
Goal 4.01
... The atomic mass of an element is the average of all the naturally existing isotopes of an element. It can be found on the periodic table under the element’s symbol. ...
... The atomic mass of an element is the average of all the naturally existing isotopes of an element. It can be found on the periodic table under the element’s symbol. ...
Name Period Nuclear Study Packet Set 1 1. What subatomic
... 1. What is the half-life of a 100.0 g sample of nitrogen-16 that decays to 12.5 g of nitrogen-16 in 21.6 s? 2. All isotopes of technetium are radioactive, but they have widely varying half-lives. If an 800.0 g sample of technetium-99 decays to 50.0 g of technetium-99 in 639 000 y, what is its ha ...
... 1. What is the half-life of a 100.0 g sample of nitrogen-16 that decays to 12.5 g of nitrogen-16 in 21.6 s? 2. All isotopes of technetium are radioactive, but they have widely varying half-lives. If an 800.0 g sample of technetium-99 decays to 50.0 g of technetium-99 in 639 000 y, what is its ha ...
Isotopes and Average Atomic Mass
... Their abundances in nature are 75%, 20%, and 5% respectively. What is the average atomic mass of cesium? Steps #1, #2 and #3 can be performed together: ...
... Their abundances in nature are 75%, 20%, and 5% respectively. What is the average atomic mass of cesium? Steps #1, #2 and #3 can be performed together: ...
Getting to Know: Periodic Table
... element do not have the same number of neutrons. For example, most atoms of the element carbon, C, have 6 neutrons in their nucleus. There are some atoms of carbon that have 7 or even 8 neutrons in their nucleus. Atoms of the same element that have a different number of neutrons are called isotopes. ...
... element do not have the same number of neutrons. For example, most atoms of the element carbon, C, have 6 neutrons in their nucleus. There are some atoms of carbon that have 7 or even 8 neutrons in their nucleus. Atoms of the same element that have a different number of neutrons are called isotopes. ...
Lawrencium

Lawrencium is a synthetic chemical element with chemical symbol Lr (formerly Lw) and atomic number 103. It is named in honor of Ernest Lawrence, inventor of the cyclotron, a device that was used to discover many artificial radioactive elements. A radioactive metal, lawrencium is the eleventh transuranic element and is also the final member of the actinide series. Like all elements with atomic number over 100, lawrencium can only be produced in particle accelerators by bombarding lighter elements with charged particles. Twelve isotopes of lawrencium are currently known; the most stable is 266Lr with a half-life of 11 hours, but the shorter-lived 260Lr (half-life 2.7 minutes) is most commonly used in chemistry because it can be produced on a larger scale.Chemistry experiments have confirmed that lawrencium indeed behaves as a heavier homolog to lutetium in the periodic table, and is a trivalent element. It thus could also be classified as the first of the 7th-period transition metals: however, its electron configuration is anomalous for its position in the periodic table, having an s2p configuration instead of the s2d configuration of its homolog lutetium. This means that lawrencium may be less volatile than expected for its position in the periodic table and have a volatility comparable to that of lead.In the 1950s, 1960s, and 1970s, many claims of the synthesis of lawrencium of varying quality were made from laboratories in the Soviet Union and the United States. The priority of the discovery and therefore the naming of the element was disputed between Soviet and American scientists, and while the International Union of Pure and Applied Chemistry (IUPAC) established lawrencium as the official name for the element and gave the American team credit for the discovery, this was reevaluated in 1997, giving both teams shared credit for the discovery but not changing the element's name.