File
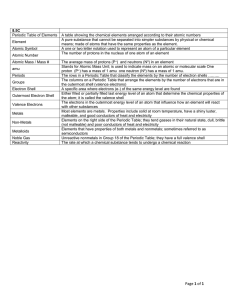
... Periodic table: A chart in which elements are arranged by increasing atomic number and by changes in physical and chemical properties. atom: The smallest particles that make up matter. proton: a subatomic particle that has a positive charge and that is located in the nucleus of an atom. (The number ...
... Periodic table: A chart in which elements are arranged by increasing atomic number and by changes in physical and chemical properties. atom: The smallest particles that make up matter. proton: a subatomic particle that has a positive charge and that is located in the nucleus of an atom. (The number ...
Chapter 9 - Fayetteville State University
... 23. The most important factor in determining the chemical behavior of an atom is its a. nuclear structure b. electron structure c. atomic mass d. solubility 24. An atom that loses its outer electron or electrons readily is a. an active metal b. an active nonmetal c. an inactive metal d. an inactive ...
... 23. The most important factor in determining the chemical behavior of an atom is its a. nuclear structure b. electron structure c. atomic mass d. solubility 24. An atom that loses its outer electron or electrons readily is a. an active metal b. an active nonmetal c. an inactive metal d. an inactive ...
Atomic terms - ATOMIC NUMBER: The number of protons in the
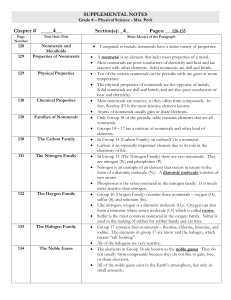
... - almost all solids at room temperature (exception: Mercury - Hg - is liquid) - appearance: shiny, mirrored surface - mostly grey - ductile (can be drawn into wires), malleable (can be hammered) - located on the left hand side of the periodic table NONMETALS - poor conductors of heat and electricity ...
... - almost all solids at room temperature (exception: Mercury - Hg - is liquid) - appearance: shiny, mirrored surface - mostly grey - ductile (can be drawn into wires), malleable (can be hammered) - located on the left hand side of the periodic table NONMETALS - poor conductors of heat and electricity ...
8.5C Vocabulary
... Either filled or partially filled last energy level of an atom that determine the chemical properties of the atom; it is called the valence shell The electrons in the outermost energy level of an atom that influence how an element will react with other substances Most elements are metals. Properties ...
... Either filled or partially filled last energy level of an atom that determine the chemical properties of the atom; it is called the valence shell The electrons in the outermost energy level of an atom that influence how an element will react with other substances Most elements are metals. Properties ...
chapters 1-4
... Atom – smallest building block; molecule – combination of two or more atoms. Can be an element or compound. ...
... Atom – smallest building block; molecule – combination of two or more atoms. Can be an element or compound. ...
Notes - Organization of Matter
... • Compounds are pure substances that are composed of two or more atoms that are chemically combined • Compounds can only be changed into simpler substances called elements by chemical changes ...
... • Compounds are pure substances that are composed of two or more atoms that are chemically combined • Compounds can only be changed into simpler substances called elements by chemical changes ...
Chapter 1_chemh
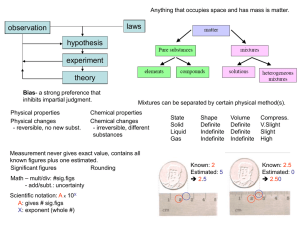
... ●Physical Property: characteristic that can be observed or measured without changing the identity of the substance. (melting, boiling, etc) oPhysical Change: change that does not involve a change in the identity of the substance. (cutting, ...
... ●Physical Property: characteristic that can be observed or measured without changing the identity of the substance. (melting, boiling, etc) oPhysical Change: change that does not involve a change in the identity of the substance. (cutting, ...
Chemistry Test Study Guide
... Atoms and The Periodic Table 4. Matter - anything that has mass and takes up space. 5. Metalloids - an element that has some characteristics of both metals and nonmetals. 6. Homogeneous Mixture – Two or more substances not chemically combined. CANNOT see individual parts of the mixture. 7. Heteroge ...
... Atoms and The Periodic Table 4. Matter - anything that has mass and takes up space. 5. Metalloids - an element that has some characteristics of both metals and nonmetals. 6. Homogeneous Mixture – Two or more substances not chemically combined. CANNOT see individual parts of the mixture. 7. Heteroge ...
Study Guide Answers
... 17. Does every atom of the same element have the same number of protons? Why or Why not? Yes, every atom of the same element has to have the same number of protons. The number of protons determines the type of atom. Example, all hydrogen atoms have 1 proton and all helium atoms have 2 protons. 18. ...
... 17. Does every atom of the same element have the same number of protons? Why or Why not? Yes, every atom of the same element has to have the same number of protons. The number of protons determines the type of atom. Example, all hydrogen atoms have 1 proton and all helium atoms have 2 protons. 18. ...
Final Exam Review Part 1
... 2. What group number are the Noble Gases found in? __________ 3. Choose a color then color the group which represents the most reactive metals on the above periodic table: Color used: ________ 4. What is the group name of the most reactive metals you colored in question 3? ___________ 5. Choose a co ...
... 2. What group number are the Noble Gases found in? __________ 3. Choose a color then color the group which represents the most reactive metals on the above periodic table: Color used: ________ 4. What is the group name of the most reactive metals you colored in question 3? ___________ 5. Choose a co ...
Periodic Table
... conductors of heat and electricity. They are mostly solids at room temp. What is one exception? ...
... conductors of heat and electricity. They are mostly solids at room temp. What is one exception? ...
Objective 3 Stations Student Sheet
... 1. How is the periodic table organized? 2. What family of elements has valence electrons at two energy levels? 3. What are the elements called that are between metals and nonmetals? 4. Which family of nonmetals has seven valence electrons? 5. What are some properties of noble gases? 6. What is anoth ...
... 1. How is the periodic table organized? 2. What family of elements has valence electrons at two energy levels? 3. What are the elements called that are between metals and nonmetals? 4. Which family of nonmetals has seven valence electrons? 5. What are some properties of noble gases? 6. What is anoth ...
Element Symbol
... chemically combined. Compounds can only be changed into simpler substances called elements by chemical changes 7. One way that two or more atoms can combine is to form a molecule. ...
... chemically combined. Compounds can only be changed into simpler substances called elements by chemical changes 7. One way that two or more atoms can combine is to form a molecule. ...
2.9 Use the helium-4 isotope to define atomic number and mass
... 2.34 Give two examples of each of the following: (a) a diatomic molecule containing atoms of the same element, (b) a diatomic molecule containing atoms of different elements, (c) polyatomic molecule containing atoms of the same element, (d) a polyatomic molecule containing atoms of different element ...
... 2.34 Give two examples of each of the following: (a) a diatomic molecule containing atoms of the same element, (b) a diatomic molecule containing atoms of different elements, (c) polyatomic molecule containing atoms of the same element, (d) a polyatomic molecule containing atoms of different element ...
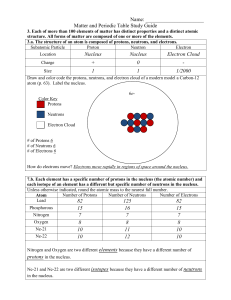
Matter and the Periodic Table Study Guide Answer Key
... How do electrons move? Electrons move rapidly in regions of space around the nucleus. 7.b. Each element has a specific number of protons in the nucleus (the atomic number) and each isotope of an element has a different but specific number of neutrons in the nucleus. Unless otherwise indicated, round ...
... How do electrons move? Electrons move rapidly in regions of space around the nucleus. 7.b. Each element has a specific number of protons in the nucleus (the atomic number) and each isotope of an element has a different but specific number of neutrons in the nucleus. Unless otherwise indicated, round ...
Metals, Nonmetals, and Metalloids (Vocabulary)
... Metals, Nonmetals, and Metalloids (Vocabulary) ...
... Metals, Nonmetals, and Metalloids (Vocabulary) ...
Periodic Trends
... – Malleable: hammered into thin sheets – Good conductors of heat/electricity – Luster: shine – Solid at room temperature (except for Hg) ...
... – Malleable: hammered into thin sheets – Good conductors of heat/electricity – Luster: shine – Solid at room temperature (except for Hg) ...
Metals, Nonmetals, and Metalloids (Vocabulary)
... are arranged by properties and are represented by one or two letter chemical symbols. ...
... are arranged by properties and are represented by one or two letter chemical symbols. ...
Name - TeacherWeb
... The elements in Group 18 are known as the noble gases. They do not usually form compounds because they do not like to gain, lose, or share electrons. All of the noble gases exist in the Earth’s atmosphere, but only in small amounts. ...
... The elements in Group 18 are known as the noble gases. They do not usually form compounds because they do not like to gain, lose, or share electrons. All of the noble gases exist in the Earth’s atmosphere, but only in small amounts. ...
Nonmetal
In chemistry, a nonmetal (or non-metal) is a chemical element that mostly lacks metallic attributes. Physically, nonmetals tend to be highly volatile (easily vaporised), have low elasticity, and are good insulators of heat and electricity; chemically, they tend to have high ionization energy and electronegativity values, and gain or share electrons when they react with other elements or compounds. Seventeen elements are generally classified as nonmetals; most are gases (hydrogen, helium, nitrogen, oxygen, fluorine, neon, chlorine, argon, krypton, xenon and radon); one is a liquid (bromine); and a few are solids (carbon, phosphorus, sulfur, selenium, and iodine).Moving rightward across the standard form of periodic table, nonmetals adopt structures that have progressively fewer nearest neighbours. Polyatomic nonmetals have structures with either three nearest neighbours, as is the case (for example) with carbon (in its standard state of graphite), or two nearest neighbours (for example) in the case of sulfur. Diatomic nonmetals, such as hydrogen, have one nearest neighbour, and the monatomic noble gases, such as helium, have none. This gradual fall in the number of nearest neighbours is associated with a reduction in metallic character and an increase in nonmetallic character. The distinction between the three categories of nonmetals, in terms of receding metallicity is not absolute. Boundary overlaps occur as outlying elements in each category show (or begin to show) less-distinct, hybrid-like or atypical properties.Although five times more elements are metals than nonmetals, two of the nonmetals—hydrogen and helium—make up over 99 per cent of the observable Universe, and one—oxygen—makes up close to half of the Earth's crust, oceans and atmosphere. Living organisms are also composed almost entirely of nonmetals, and nonmetals form many more compounds than metals.