Chapter 1.1 –Chemistry is a Physical Science Chemistry is one of
... Examples: Table salt (sodium chloride, NaCl), sugar (sucrose, C12H22O11), water (H2O), iron (Fe), copper (Cu), and oxygen (O2). ...
... Examples: Table salt (sodium chloride, NaCl), sugar (sucrose, C12H22O11), water (H2O), iron (Fe), copper (Cu), and oxygen (O2). ...
Document
... 41) An atom of fluorine has 10 neutrons. What is the total number of other subatomic particles in this atom? a) 9 protons and 9 electrons b) 9 protons and 19 electrons c) 10 protons and 10 electrons d) 19 protons and 19 electrons ...
... 41) An atom of fluorine has 10 neutrons. What is the total number of other subatomic particles in this atom? a) 9 protons and 9 electrons b) 9 protons and 19 electrons c) 10 protons and 10 electrons d) 19 protons and 19 electrons ...
Metals and non-metals III IMPORTANT POINTS Non-metals
... 1. a. Magnesium, chromium and sodium are all metals, hence, they react with oxygen to form basic oxides b. Chromium, as it is a transition metal. Metals have high density and coloured compounds are formed by transition metals. c. Bromine - the formula is Br2, that is, two atoms of bromine. d. Bromin ...
... 1. a. Magnesium, chromium and sodium are all metals, hence, they react with oxygen to form basic oxides b. Chromium, as it is a transition metal. Metals have high density and coloured compounds are formed by transition metals. c. Bromine - the formula is Br2, that is, two atoms of bromine. d. Bromin ...
Periodic Trends & the Periodic Table
... elements is called the lanthanides because they follow element number 57, lanthanum. • The second series of inner transition elements, the actinides, have atomic numbers ranging from 90 (thorium, Th) to 103 (lawrencium, Lr). ...
... elements is called the lanthanides because they follow element number 57, lanthanum. • The second series of inner transition elements, the actinides, have atomic numbers ranging from 90 (thorium, Th) to 103 (lawrencium, Lr). ...
Atoms, Molecules and Ions
... - Robert Boyle (1627-91): the first “chemist” - Performed quantitative experiments. - Dalton (1766-44), Darwin(1809-82) ...
... - Robert Boyle (1627-91): the first “chemist” - Performed quantitative experiments. - Dalton (1766-44), Darwin(1809-82) ...
Periodic Table Powerpoint
... Elements from different families Not similar properties Last element an inactive gas Reactive – unreactive 7 periods Valance electrons increase from left to right across a period. ...
... Elements from different families Not similar properties Last element an inactive gas Reactive – unreactive 7 periods Valance electrons increase from left to right across a period. ...
Regents questions
... Sample 7.1 Natural gas used in home heating and cooking is odorless. Because natural gas leaks pose the danger of explosion or suffocation, various smelly substances are added to the gas to allow detection of a leak. One such substance is methyl mercaptan, CH3SH. Use Figure 7.6 to predict the lengt ...
... Sample 7.1 Natural gas used in home heating and cooking is odorless. Because natural gas leaks pose the danger of explosion or suffocation, various smelly substances are added to the gas to allow detection of a leak. One such substance is methyl mercaptan, CH3SH. Use Figure 7.6 to predict the lengt ...
Periodic Table
... d. form negative ions (anions) e. poor conductors of heat and electricity f. generally gases or brittle solids g. low melting and boiling points 3. Metalloids (Semimetals) = have properties of both metals and non-metals III. Trends or Patterns in the Periodic Table A. Certain properties of elements ...
... d. form negative ions (anions) e. poor conductors of heat and electricity f. generally gases or brittle solids g. low melting and boiling points 3. Metalloids (Semimetals) = have properties of both metals and non-metals III. Trends or Patterns in the Periodic Table A. Certain properties of elements ...
CHE 1401 - Fall 2013 - Chapter 7 Homework 7 (Chapter 7: Periodic
... A) alkali metals have lower densities B) alkali metals have greater electron affinities C) alkali metals have lower ionization energies D) alkali metals have lower melting points E) alkali metals are not more reactive than alkaline earth metals ...
... A) alkali metals have lower densities B) alkali metals have greater electron affinities C) alkali metals have lower ionization energies D) alkali metals have lower melting points E) alkali metals are not more reactive than alkaline earth metals ...
Chemistry Notes with Blanks
... A _________ bond holds the two hydrogen atoms together. A molecule is a group of atoms held together by covalent bonds. It has no_________charge. _________ electrons in the 1st shell, _________in the 2nd, and _________ in the 3rd An atom (or group of atoms) that gains or loses electrons has ...
... A _________ bond holds the two hydrogen atoms together. A molecule is a group of atoms held together by covalent bonds. It has no_________charge. _________ electrons in the 1st shell, _________in the 2nd, and _________ in the 3rd An atom (or group of atoms) that gains or loses electrons has ...
1) - Kurt Niedenzu
... within a group is primarily due to an increase in the number of a) neutrons in the nucleus b) electrons in the outermost shell c) unpaired electrons d) occupied principal energy levels 33) Elements that have properties of both metals and nonmetals are called a) alkali metals c) metalloids b) transit ...
... within a group is primarily due to an increase in the number of a) neutrons in the nucleus b) electrons in the outermost shell c) unpaired electrons d) occupied principal energy levels 33) Elements that have properties of both metals and nonmetals are called a) alkali metals c) metalloids b) transit ...
Chapter 3 STUDY GUIDE True/False Indicate whether the statement
... 9. Which of the following CANNOT be classified as matter? a. an odorless, colorless gas c. alpha particles b. the heat given off by a light bulb d. an electron cloud 10. The millions of compounds that exist are made from approximately ____ elements. a. 50 c. 115 b. 75 d. 150 11. In general, nonmetal ...
... 9. Which of the following CANNOT be classified as matter? a. an odorless, colorless gas c. alpha particles b. the heat given off by a light bulb d. an electron cloud 10. The millions of compounds that exist are made from approximately ____ elements. a. 50 c. 115 b. 75 d. 150 11. In general, nonmetal ...
The Periodic Table - Harlan Independent Schools
... The lighter alkaline earth metals such as magnesium and calcium are very important in animal and plant physiology. You all know that calcium helps build your ...
... The lighter alkaline earth metals such as magnesium and calcium are very important in animal and plant physiology. You all know that calcium helps build your ...
Study Guide Matter: Building Blocks of the Universe
... Matter: Building Blocks of the Universe Test consists of Multiple Choice, filling in charts with missing data and short answer. You should be prepared to answer questions on these topics. * Know the key people in the history of the atom and their contribution to our understanding of the atom. These ...
... Matter: Building Blocks of the Universe Test consists of Multiple Choice, filling in charts with missing data and short answer. You should be prepared to answer questions on these topics. * Know the key people in the history of the atom and their contribution to our understanding of the atom. These ...
Notes matter energy
... On the Periodic Table of the Elements (See Week 1 handout), gaseous elements have symbols with an italic font, liquid elements have symbols with an outline font, and solids have symbols with a Times-Roman font. The Law of Definite Composition states that compounds always contain the same proportions ...
... On the Periodic Table of the Elements (See Week 1 handout), gaseous elements have symbols with an italic font, liquid elements have symbols with an outline font, and solids have symbols with a Times-Roman font. The Law of Definite Composition states that compounds always contain the same proportions ...
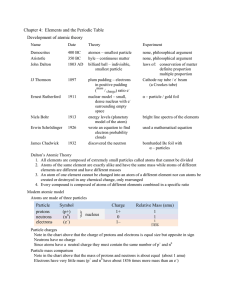
Chapter 4: Elements and the Periodic Table Development of atomic
... Luster – most metals are very shiny or have high metallic luster Magnetic – many metals (but not all) are attracted to magnets Chemical properties of metals Reactivity – metals react by losing electrons to form positive ions Some metals are useful because they are extremely reactive (Li in batteries ...
... Luster – most metals are very shiny or have high metallic luster Magnetic – many metals (but not all) are attracted to magnets Chemical properties of metals Reactivity – metals react by losing electrons to form positive ions Some metals are useful because they are extremely reactive (Li in batteries ...
Chapter 10 Handouts_1
... •Active elements liberate more heat when they react than do inactive elements. •Active elements usually form stable compounds. ...
... •Active elements liberate more heat when they react than do inactive elements. •Active elements usually form stable compounds. ...
Chapter 10 Handouts - Bakersfield College
... •Active elements liberate more heat when they react than do inactive elements. •Active elements usually form stable compounds. ...
... •Active elements liberate more heat when they react than do inactive elements. •Active elements usually form stable compounds. ...
Chapter 10_Handouts_6
... •All metals, except mercury, are •Nonmetals may be solid, solid at room temperature. liquid, or gaseous. •Metals have a characteristic metallic luster. ...
... •All metals, except mercury, are •Nonmetals may be solid, solid at room temperature. liquid, or gaseous. •Metals have a characteristic metallic luster. ...
Overall Score: _____ / 22 (each question is worth
... Metals have a higher melting point than nonmetals. The particles in a metal are more tightly packed together, so it takes a higher temperature to give the particles enough energy to separate them. The particles in a nonmetal are not as tightly packed together, so it does not take as much heat to giv ...
... Metals have a higher melting point than nonmetals. The particles in a metal are more tightly packed together, so it takes a higher temperature to give the particles enough energy to separate them. The particles in a nonmetal are not as tightly packed together, so it does not take as much heat to giv ...
Ch 2-1 Properties of Matter
... on the type of matter. Mass and volume are extensive properties. Color and hardness are intensive properties. 36) Possible answers: reddish-yellow color, conductor of heat and electricity, malleable, melting point of 1084oC, and boiling point of 2562oC. ...
... on the type of matter. Mass and volume are extensive properties. Color and hardness are intensive properties. 36) Possible answers: reddish-yellow color, conductor of heat and electricity, malleable, melting point of 1084oC, and boiling point of 2562oC. ...
Chapter 10 The Periodic Law
... •Nonmetals may be solid, liquid, or gaseous. •Nonmetals do not have a luster. •Most nonmetals are transparent in thin sheets. •Solid nonmetals are brittle. •Nonmetals are insulators. ...
... •Nonmetals may be solid, liquid, or gaseous. •Nonmetals do not have a luster. •Most nonmetals are transparent in thin sheets. •Solid nonmetals are brittle. •Nonmetals are insulators. ...
Nonmetal
In chemistry, a nonmetal (or non-metal) is a chemical element that mostly lacks metallic attributes. Physically, nonmetals tend to be highly volatile (easily vaporised), have low elasticity, and are good insulators of heat and electricity; chemically, they tend to have high ionization energy and electronegativity values, and gain or share electrons when they react with other elements or compounds. Seventeen elements are generally classified as nonmetals; most are gases (hydrogen, helium, nitrogen, oxygen, fluorine, neon, chlorine, argon, krypton, xenon and radon); one is a liquid (bromine); and a few are solids (carbon, phosphorus, sulfur, selenium, and iodine).Moving rightward across the standard form of periodic table, nonmetals adopt structures that have progressively fewer nearest neighbours. Polyatomic nonmetals have structures with either three nearest neighbours, as is the case (for example) with carbon (in its standard state of graphite), or two nearest neighbours (for example) in the case of sulfur. Diatomic nonmetals, such as hydrogen, have one nearest neighbour, and the monatomic noble gases, such as helium, have none. This gradual fall in the number of nearest neighbours is associated with a reduction in metallic character and an increase in nonmetallic character. The distinction between the three categories of nonmetals, in terms of receding metallicity is not absolute. Boundary overlaps occur as outlying elements in each category show (or begin to show) less-distinct, hybrid-like or atypical properties.Although five times more elements are metals than nonmetals, two of the nonmetals—hydrogen and helium—make up over 99 per cent of the observable Universe, and one—oxygen—makes up close to half of the Earth's crust, oceans and atmosphere. Living organisms are also composed almost entirely of nonmetals, and nonmetals form many more compounds than metals.