Matter - HCC Learning Web
... • Nonmetals typically have low melting points and low densities and have a dull appearance. • Nonmetals are poor conductors of heat and electricity. • Nonmetals are not malleable or ductile and crush into a powder when hammered. • 11 nonmetals occur naturally in the gaseous state. ...
... • Nonmetals typically have low melting points and low densities and have a dull appearance. • Nonmetals are poor conductors of heat and electricity. • Nonmetals are not malleable or ductile and crush into a powder when hammered. • 11 nonmetals occur naturally in the gaseous state. ...
AP CHEMISTRY SUMMER ASSIGNMENT AP Chemistry is a
... conductors, shiny, malleable and ductile. They are solids except for liquid mercury. Nonmetals – elements located to the right of the staircase, plus Hydrogen; they share or gain electrons. Nonmetals can be solids, liquid (Br2) or gases. They are brittle and are poor conductors of electricity. Metal ...
... conductors, shiny, malleable and ductile. They are solids except for liquid mercury. Nonmetals – elements located to the right of the staircase, plus Hydrogen; they share or gain electrons. Nonmetals can be solids, liquid (Br2) or gases. They are brittle and are poor conductors of electricity. Metal ...
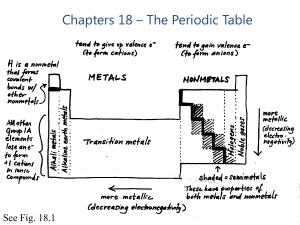
Chapters 18 – The Periodic Table
... agent such as potassium chlorate together with tetraphosphorus trisulfide (P4S3), glass and binder. The phosphorus sulfide is easily ignited, the potassium chlorate decomposes to give oxygen, which in turn causes the phosphorus sulfide to burn more vigorously. The head of safety matches are made of ...
... agent such as potassium chlorate together with tetraphosphorus trisulfide (P4S3), glass and binder. The phosphorus sulfide is easily ignited, the potassium chlorate decomposes to give oxygen, which in turn causes the phosphorus sulfide to burn more vigorously. The head of safety matches are made of ...
specific vocabulary of the unit
... are sometimes called semi-metals and have characteristics that resemble both metals and nonmetals. Alkalis ['ælkəlaɪ] First group of the periodic table of elements. ...
... are sometimes called semi-metals and have characteristics that resemble both metals and nonmetals. Alkalis ['ælkəlaɪ] First group of the periodic table of elements. ...
Chemistry- CST Review
... a) sodium, lithium b) strontium, magnesium c) carbon, oxygen d) selenium, bromine ...
... a) sodium, lithium b) strontium, magnesium c) carbon, oxygen d) selenium, bromine ...
ch22 lecture 7e
... – The inorganic cycle involves slow weathering of phosphatecontaining rocks, which causes PO43– to leach into the rivers and seas. – The land-based biological cycle involves incorporation of PO43– into organisms and its release through excretion and ...
... – The inorganic cycle involves slow weathering of phosphatecontaining rocks, which causes PO43– to leach into the rivers and seas. – The land-based biological cycle involves incorporation of PO43– into organisms and its release through excretion and ...
Honors Mid-Term Review Sheet
... 66. What charges do the following elements have when they form ions? a. S b. Ne c. Ca d. P 67. Draw the periodic trends for atomic size, ionization energy, and electronegativity. Chapter 7 68. Write the formula for the compound formed between the following elements: a. lithium and sulfur b. aluminum ...
... 66. What charges do the following elements have when they form ions? a. S b. Ne c. Ca d. P 67. Draw the periodic trends for atomic size, ionization energy, and electronegativity. Chapter 7 68. Write the formula for the compound formed between the following elements: a. lithium and sulfur b. aluminum ...
PPTB&W - Gmu - George Mason University
... Other group members are metals – shiny, relatively soft with low melting points Aluminum is more ionic; its low density and 3 valence electrons make it a good electrical conductor Although Aluminum is a metal, its halides exist in the gaseous state as covalent dimers - AL2Cl6 (contrast salts of grou ...
... Other group members are metals – shiny, relatively soft with low melting points Aluminum is more ionic; its low density and 3 valence electrons make it a good electrical conductor Although Aluminum is a metal, its halides exist in the gaseous state as covalent dimers - AL2Cl6 (contrast salts of grou ...
g - Santa Rosa Junior College
... – The inorganic cycle involves slow weathering of phosphatecontaining rocks, which causes PO43- to leach into the rivers and seas. – The land-based biological cycle involves incorporation of PO43- into organisms and its release through excretion and ...
... – The inorganic cycle involves slow weathering of phosphatecontaining rocks, which causes PO43- to leach into the rivers and seas. – The land-based biological cycle involves incorporation of PO43- into organisms and its release through excretion and ...
PPT - George Mason University
... Other group members are metals – shiny, relatively soft with low melting points Aluminum is more ionic; its low density and 3 valence electrons make it a good electrical conductor Although Aluminum is a metal, its halides exist in the gaseous state as covalent dimers - AL2Cl6 (contrast salts of grou ...
... Other group members are metals – shiny, relatively soft with low melting points Aluminum is more ionic; its low density and 3 valence electrons make it a good electrical conductor Although Aluminum is a metal, its halides exist in the gaseous state as covalent dimers - AL2Cl6 (contrast salts of grou ...
3 chemical foundations: elements, atoms and ions
... The electrons move at random outside of the nucleus, although they do occupy preferred regions in space. Although the electrons move at random, they each have a very definite fixed energy. The physical and chemical properties of an atom really depend on the energy, number and arrangement of the elec ...
... The electrons move at random outside of the nucleus, although they do occupy preferred regions in space. Although the electrons move at random, they each have a very definite fixed energy. The physical and chemical properties of an atom really depend on the energy, number and arrangement of the elec ...
ch22_lecture_6e_final
... – The inorganic cycle involves slow weathering of phosphatecontaining rocks, which causes PO43- to leach into the rivers and seas. – The land-based biological cycle involves incorporation of PO43- into organisms and its release through excretion and ...
... – The inorganic cycle involves slow weathering of phosphatecontaining rocks, which causes PO43- to leach into the rivers and seas. – The land-based biological cycle involves incorporation of PO43- into organisms and its release through excretion and ...
The Representative Elements: Group 5A Through 8A
... cyclo-S8. The most stable allotrope is orthorhombic -S8, which is made up entirely of the S8 molecules. All other allotropes of sulfur eventually revert to this form. The Group 6A elements exhibit the widest range of atomic properties. At the top, oxygen has the highest electronegativity of 3.5 and ...
... cyclo-S8. The most stable allotrope is orthorhombic -S8, which is made up entirely of the S8 molecules. All other allotropes of sulfur eventually revert to this form. The Group 6A elements exhibit the widest range of atomic properties. At the top, oxygen has the highest electronegativity of 3.5 and ...
MYP 10 PeriodicityWS
... 3.1.1 Describe the arrangement of elements in the periodic table in order of increasing atomic number. 3.1.2 Distinguish between the terms group and period. 3.1.3 Apply the relationship between the electron arrangement of elements and their position in the periodic table up to Z = 20. 3.1.4 Apply th ...
... 3.1.1 Describe the arrangement of elements in the periodic table in order of increasing atomic number. 3.1.2 Distinguish between the terms group and period. 3.1.3 Apply the relationship between the electron arrangement of elements and their position in the periodic table up to Z = 20. 3.1.4 Apply th ...
Chapter 20 – The Representative Elements
... Important Trends in The Chemical Behaviors of the Group 5A Elements Elements of Group 5A overwhelmingly form covalent compounds. Whereas nitrogen can form a maximum of four covalent bonds, other elements in the group can form more than four covalent bonds by utilizing one or more of the nd orbitals. ...
... Important Trends in The Chemical Behaviors of the Group 5A Elements Elements of Group 5A overwhelmingly form covalent compounds. Whereas nitrogen can form a maximum of four covalent bonds, other elements in the group can form more than four covalent bonds by utilizing one or more of the nd orbitals. ...
3 molecules
... Valence Electrons in Ionic Compounds • The A-group (representative) elements follow the OCTET RULE; they obtain an inert gas valence (outer) shell that contains 8 electrons • Metals - lose # electrons = group number e.g. Ca Ca2+ + 2e- (Ar outer shell) • Nonmetals - gain electrons = 8 - group # e. ...
... Valence Electrons in Ionic Compounds • The A-group (representative) elements follow the OCTET RULE; they obtain an inert gas valence (outer) shell that contains 8 electrons • Metals - lose # electrons = group number e.g. Ca Ca2+ + 2e- (Ar outer shell) • Nonmetals - gain electrons = 8 - group # e. ...
Atoms, Elements and Compounds Home
... All students are expected to complete the bronze level tasks. These are designed to consolidate students’ knowledge of the key concepts met in the unit. Students who complete the all the bronze level tasks to a suitable standard will be rewarded with one house point. We would expect most students to ...
... All students are expected to complete the bronze level tasks. These are designed to consolidate students’ knowledge of the key concepts met in the unit. Students who complete the all the bronze level tasks to a suitable standard will be rewarded with one house point. We would expect most students to ...
Sub Unit Plan 1 Chem Periodic Table
... II.3 Elements can be classified by their properties and located on the Periodic Table as metals, nonmetals, metalloids (B, Si, Ge, As, Sb, Te), and noble gases. (3.1v) II.4 Elements can be differentiated by their physical properties. Physical properties of substances, such as density, conductivity, ...
... II.3 Elements can be classified by their properties and located on the Periodic Table as metals, nonmetals, metalloids (B, Si, Ge, As, Sb, Te), and noble gases. (3.1v) II.4 Elements can be differentiated by their physical properties. Physical properties of substances, such as density, conductivity, ...
Ionic Bonding - petersonORHS
... dot notation for the first two elements in each group. The other elements in that group will be the same. >Use these example to help you draw dot notations. ...
... dot notation for the first two elements in each group. The other elements in that group will be the same. >Use these example to help you draw dot notations. ...
Chemistry Mid-Term Review Sheet
... 59. What charges do the following elements have when they form ions? a. S b. Ne c. Ca d. P 60. Draw the periodic trends for atomic size, ionization energy, and electronegativity. Chapter 7 61. Write the formula for the compound formed between the following elements: a. lithium and sulfur b. aluminum ...
... 59. What charges do the following elements have when they form ions? a. S b. Ne c. Ca d. P 60. Draw the periodic trends for atomic size, ionization energy, and electronegativity. Chapter 7 61. Write the formula for the compound formed between the following elements: a. lithium and sulfur b. aluminum ...
Chapter 1 Chemistry: The Study of Matter
... Properties of nonmetals Nonmetals have certain properties: poor conductor of heat and electricity, dull luster and brittle if solids, many nonmetals are gases. Bromine is a dark red liquid. ...
... Properties of nonmetals Nonmetals have certain properties: poor conductor of heat and electricity, dull luster and brittle if solids, many nonmetals are gases. Bromine is a dark red liquid. ...
Chemistry Midterm Review 2006
... 13. Define the words “reactant” and “product”. In a chemical equation, where are the reactants located? Where are the products located? What separates them from each other? 14. Classify each as a physical or chemical change: food spoiling, water boils, nail rusting, baking bread, sugar dissolving in ...
... 13. Define the words “reactant” and “product”. In a chemical equation, where are the reactants located? Where are the products located? What separates them from each other? 14. Classify each as a physical or chemical change: food spoiling, water boils, nail rusting, baking bread, sugar dissolving in ...
Chapter One Powerpoint - Geneva Area City Schools
... Introduction to the Periodic Table • All known elements are organized into a chart known as the periodic table • The vertical columns of the periodic table are called groups, or families. • Each group contains elements with similar chemical properties. • The horizontal rows of elements in the perio ...
... Introduction to the Periodic Table • All known elements are organized into a chart known as the periodic table • The vertical columns of the periodic table are called groups, or families. • Each group contains elements with similar chemical properties. • The horizontal rows of elements in the perio ...
The representative Elements: Groups 1A – 4A
... Properties of Boron (1s2 2s2 2p1) • A metalloid; forms covalent network solid; highest melting point in the group, and the least reactive • All boron compounds are covalent molecules; • Boron molecules, such as BF3, have incomplete octet and acts as Lewis acid, example: BF3 + :NH3 F3B:NH3 • Boron ...
... Properties of Boron (1s2 2s2 2p1) • A metalloid; forms covalent network solid; highest melting point in the group, and the least reactive • All boron compounds are covalent molecules; • Boron molecules, such as BF3, have incomplete octet and acts as Lewis acid, example: BF3 + :NH3 F3B:NH3 • Boron ...
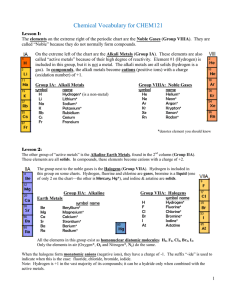
Vocabulary CHEM121
... compound is neutral (the positive and negative charges must add up to zero). The symbol for the metal is listed first and subscripts are used to indicate the number of ions (if more than 1). The name is that of the metal followed by that of the non-metal with the “-ide” ending to indicate the homonu ...
... compound is neutral (the positive and negative charges must add up to zero). The symbol for the metal is listed first and subscripts are used to indicate the number of ions (if more than 1). The name is that of the metal followed by that of the non-metal with the “-ide” ending to indicate the homonu ...
Nonmetal
In chemistry, a nonmetal (or non-metal) is a chemical element that mostly lacks metallic attributes. Physically, nonmetals tend to be highly volatile (easily vaporised), have low elasticity, and are good insulators of heat and electricity; chemically, they tend to have high ionization energy and electronegativity values, and gain or share electrons when they react with other elements or compounds. Seventeen elements are generally classified as nonmetals; most are gases (hydrogen, helium, nitrogen, oxygen, fluorine, neon, chlorine, argon, krypton, xenon and radon); one is a liquid (bromine); and a few are solids (carbon, phosphorus, sulfur, selenium, and iodine).Moving rightward across the standard form of periodic table, nonmetals adopt structures that have progressively fewer nearest neighbours. Polyatomic nonmetals have structures with either three nearest neighbours, as is the case (for example) with carbon (in its standard state of graphite), or two nearest neighbours (for example) in the case of sulfur. Diatomic nonmetals, such as hydrogen, have one nearest neighbour, and the monatomic noble gases, such as helium, have none. This gradual fall in the number of nearest neighbours is associated with a reduction in metallic character and an increase in nonmetallic character. The distinction between the three categories of nonmetals, in terms of receding metallicity is not absolute. Boundary overlaps occur as outlying elements in each category show (or begin to show) less-distinct, hybrid-like or atypical properties.Although five times more elements are metals than nonmetals, two of the nonmetals—hydrogen and helium—make up over 99 per cent of the observable Universe, and one—oxygen—makes up close to half of the Earth's crust, oceans and atmosphere. Living organisms are also composed almost entirely of nonmetals, and nonmetals form many more compounds than metals.