Day 13 Main Group Pt 1
... A. Introduction. Hydrogen is one of the most unique elements of the periodic table. Most people live their entire lives without ever encountering molecular hydrogen — yet we are constantly in contact with and dependent on compounds of hydrogen. The most familiar compound of hydrogen is water. Hydrog ...
... A. Introduction. Hydrogen is one of the most unique elements of the periodic table. Most people live their entire lives without ever encountering molecular hydrogen — yet we are constantly in contact with and dependent on compounds of hydrogen. The most familiar compound of hydrogen is water. Hydrog ...
Topic 4 Chemistry of the Elements of the Main Group
... groups: Metals, Metalloids and non-Metals Non-metals are not electrical conductors, they are characterised by a high electronegativity value (over 2). The valence electrons of non-metals are strongly attracted to their positively charged nucleus and are not available to conduct electricity. Metals ...
... groups: Metals, Metalloids and non-Metals Non-metals are not electrical conductors, they are characterised by a high electronegativity value (over 2). The valence electrons of non-metals are strongly attracted to their positively charged nucleus and are not available to conduct electricity. Metals ...
CHAPTER 1
... liquid water takes up a definite amount of space, but the water takes the shape of its container. Liquids have this characteristic because the particles in them are close together but can move past one another. The particles in a liquid move more rapidly than those in a solid. This causes them to ov ...
... liquid water takes up a definite amount of space, but the water takes the shape of its container. Liquids have this characteristic because the particles in them are close together but can move past one another. The particles in a liquid move more rapidly than those in a solid. This causes them to ov ...
Elements and the Periodic Table
... • Nuclear reaction - reaction involving the particles in the nucleus of an atom • Unlike a chemical reaction which only involves an atom’s electrons • Isotope - atoms with the same number of protons and different numbers of neutrons • Some are unstable if the nuclei do not hold together well • Radio ...
... • Nuclear reaction - reaction involving the particles in the nucleus of an atom • Unlike a chemical reaction which only involves an atom’s electrons • Isotope - atoms with the same number of protons and different numbers of neutrons • Some are unstable if the nuclei do not hold together well • Radio ...
Chapter 1: Matter and Change
... Properties and Changes in Matter Every substance, whether it is an element or a compound, has characteristic properties. Chemists use properties to distinguish between substances and to separate them. Most chemical investigations are related to or depend on the properties of substances. A property m ...
... Properties and Changes in Matter Every substance, whether it is an element or a compound, has characteristic properties. Chemists use properties to distinguish between substances and to separate them. Most chemical investigations are related to or depend on the properties of substances. A property m ...
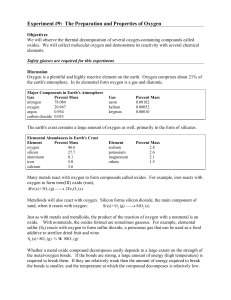
Experiment # 9 Properties of Oxygen
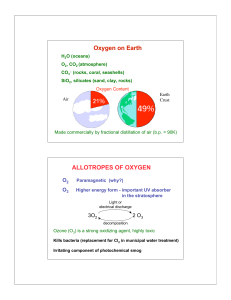
... Just as with metals and metalloids, the product of the reaction of oxygen with a nonmetal is an oxide. With nonmetals, the oxides formed are sometimes gaseous. For example, elemental sulfur (S8) reacts with oxygen to form sulfur dioxide, a poisonous gas that can be used as a food additive to sterili ...
... Just as with metals and metalloids, the product of the reaction of oxygen with a nonmetal is an oxide. With nonmetals, the oxides formed are sometimes gaseous. For example, elemental sulfur (S8) reacts with oxygen to form sulfur dioxide, a poisonous gas that can be used as a food additive to sterili ...
Atoms and Elements: Are they Related?
... Family: Elements in the same column have similar chemical property because of similar valence electrons Alkali, Alkaline, halogens, noble gases ...
... Family: Elements in the same column have similar chemical property because of similar valence electrons Alkali, Alkaline, halogens, noble gases ...
Elements and the Periodic Table
... • Nuclear reaction - reaction involving the particles in the nucleus of an atom • Unlike a chemical reaction which only involves an atom’s electrons • Isotope - atoms with the same number of protons and different numbers of neutrons • Some are unstable if the nuclei do not hold together well • Radio ...
... • Nuclear reaction - reaction involving the particles in the nucleus of an atom • Unlike a chemical reaction which only involves an atom’s electrons • Isotope - atoms with the same number of protons and different numbers of neutrons • Some are unstable if the nuclei do not hold together well • Radio ...
solid metal
... element in Earth’s crust, oceans, and atmosphere. The natural abundance of an element listed as rare is less than 1 mg per metric ton (1000 kg). An element listed as synthetic is made artificially and does not occur naturally. An element listed as unstable often disintegrates in a fraction of a seco ...
... element in Earth’s crust, oceans, and atmosphere. The natural abundance of an element listed as rare is less than 1 mg per metric ton (1000 kg). An element listed as synthetic is made artificially and does not occur naturally. An element listed as unstable often disintegrates in a fraction of a seco ...
p-Block Elements, Part 1
... e.g. Li2O = 2Li+ O2− Peroxide Ion ⇒ O22− = −O – O− e.g. Na2O2 = 2 Na+ −O – O − Also, H2O2 (hydrogen peroxide) Superoxide Ion ⇒ O2− e.g. KO2 = K+ O2− Can have positive oxidation states in combination with fluorine + 2 in OF2 ...
... e.g. Li2O = 2Li+ O2− Peroxide Ion ⇒ O22− = −O – O− e.g. Na2O2 = 2 Na+ −O – O − Also, H2O2 (hydrogen peroxide) Superoxide Ion ⇒ O2− e.g. KO2 = K+ O2− Can have positive oxidation states in combination with fluorine + 2 in OF2 ...
pblock - Chemistry Courses
... 2nd period: Only s and p orbitals are possible with n = 2 Therefore, the maximum number of bonds is 4 (single and/or double bonds) Examples: CH4, NF4+, BH43rd (and higher periods): can use d-orbitals to make bonds E.g. ...
... 2nd period: Only s and p orbitals are possible with n = 2 Therefore, the maximum number of bonds is 4 (single and/or double bonds) Examples: CH4, NF4+, BH43rd (and higher periods): can use d-orbitals to make bonds E.g. ...
Unit 3 Review Packet
... a. A chemical reaction produced a gas. b. Only a physical change occurred. c. The temperature of the substance changed. d. No chemical reaction took place. Which of the following observed properties is most reliable in classifying a substance as a metal? a. Brittle ...
... a. A chemical reaction produced a gas. b. Only a physical change occurred. c. The temperature of the substance changed. d. No chemical reaction took place. Which of the following observed properties is most reliable in classifying a substance as a metal? a. Brittle ...
Chemistry-5th-Edition-Brady-Solution-Manual
... is made, the same proportions by mass are used. This could be done by decomposing a compound and showing that the masses of the elements are always in the same ratio. To test the law of multiple proportions, two different compounds made up of the same elements would have to be decomposed. The amount ...
... is made, the same proportions by mass are used. This could be done by decomposing a compound and showing that the masses of the elements are always in the same ratio. To test the law of multiple proportions, two different compounds made up of the same elements would have to be decomposed. The amount ...
Memorization?
... and a nonmetal react. Covalent (molecular) compounds are formed when two nonmetals react. ...
... and a nonmetal react. Covalent (molecular) compounds are formed when two nonmetals react. ...
Introduction(s)
... and a nonmetal react. Covalent (molecular) compounds are formed when two nonmetals react. ...
... and a nonmetal react. Covalent (molecular) compounds are formed when two nonmetals react. ...
Nitrogen`s oxidation states
... electronegativities of the elements increase. Oxygen's electronegativity (3.5) is quite large and second only to fluorine's. All of the non-metals have electronegativities that are greater than 2.0 so the difference in electronegativities between a non-metal and oxygen is always less than 1.5. Elect ...
... electronegativities of the elements increase. Oxygen's electronegativity (3.5) is quite large and second only to fluorine's. All of the non-metals have electronegativities that are greater than 2.0 so the difference in electronegativities between a non-metal and oxygen is always less than 1.5. Elect ...
Name - Piscataway High School
... In the first week, she studied daily for 15 minutes and her end of the week test scores were 60%. During the second week, she studied daily for 30 minutes and her end of the week test scores were 70%. During the third week, she studied for 45 minutes and her end of the week test scores were 80%. Fin ...
... In the first week, she studied daily for 15 minutes and her end of the week test scores were 60%. During the second week, she studied daily for 30 minutes and her end of the week test scores were 70%. During the third week, she studied for 45 minutes and her end of the week test scores were 80%. Fin ...
Unit 1. Materials: Formulating Matter A. How do chemists describe
... A.3 Physical and Chemical Properties Classify each statement as a describing either a physical property (P) or a chemical property (C). [Hint: Decide whether the chemical identity of the material does or does not change when the property is observed.] 1. Pure silver has a high luster (is shiny and r ...
... A.3 Physical and Chemical Properties Classify each statement as a describing either a physical property (P) or a chemical property (C). [Hint: Decide whether the chemical identity of the material does or does not change when the property is observed.] 1. Pure silver has a high luster (is shiny and r ...
Chapter 1 Matter and Energy Classifying Matter – An Exercise
... Explain the characteristics you considered in making your decision. ...
... Explain the characteristics you considered in making your decision. ...
Ionic Bonding
... (b) Give a theoretical explanation for your answer to (a). 9. How are coordinate covalent bonds similar to covalent bonds? How are they different? 10. (a) Use an electron dot diagram to explain the formula for nitrogen, N2. (b) Draw the Lewis structure for nitrogen. (c) Nitrogen is a fairly inert (u ...
... (b) Give a theoretical explanation for your answer to (a). 9. How are coordinate covalent bonds similar to covalent bonds? How are they different? 10. (a) Use an electron dot diagram to explain the formula for nitrogen, N2. (b) Draw the Lewis structure for nitrogen. (c) Nitrogen is a fairly inert (u ...
CHEMISTRY The Central Science 9th Edition
... their outer shell , or the outer shell electron configuration of the nearest noble gas , either by losing one electron (or more) or by gaining one electron (or more) , or by sharing electrons. ...
... their outer shell , or the outer shell electron configuration of the nearest noble gas , either by losing one electron (or more) or by gaining one electron (or more) , or by sharing electrons. ...
Determining Density through graphing
... In the first week, she studied daily for 15 minutes and her end of the week test scores were 60%. During the second week, she studied daily for 30 minutes and her end of the week test scores were 70%. During the third week, she studied for 45 minutes and her end of the week test scores were 80%. Fin ...
... In the first week, she studied daily for 15 minutes and her end of the week test scores were 60%. During the second week, she studied daily for 30 minutes and her end of the week test scores were 70%. During the third week, she studied for 45 minutes and her end of the week test scores were 80%. Fin ...
Determining Density through graphing
... In the first week, she studied daily for 15 minutes and her end of the week test scores were 60%. During the second week, she studied daily for 30 minutes and her end of the week test scores were 70%. During the third week, she studied for 45 minutes and her end of the week test scores were 80%. Fin ...
... In the first week, she studied daily for 15 minutes and her end of the week test scores were 60%. During the second week, she studied daily for 30 minutes and her end of the week test scores were 70%. During the third week, she studied for 45 minutes and her end of the week test scores were 80%. Fin ...
Chemistry of the Non
... We divide the periodic table into metals, nonmetals and metalloids. Nonmetals occupy the upper right portion of the periodic table. • H is a special case. Electronegativity is important when determining whether an element is a metal. Nonmetals tend to have higher electronegativities than metals. • T ...
... We divide the periodic table into metals, nonmetals and metalloids. Nonmetals occupy the upper right portion of the periodic table. • H is a special case. Electronegativity is important when determining whether an element is a metal. Nonmetals tend to have higher electronegativities than metals. • T ...
Nonmetal
In chemistry, a nonmetal (or non-metal) is a chemical element that mostly lacks metallic attributes. Physically, nonmetals tend to be highly volatile (easily vaporised), have low elasticity, and are good insulators of heat and electricity; chemically, they tend to have high ionization energy and electronegativity values, and gain or share electrons when they react with other elements or compounds. Seventeen elements are generally classified as nonmetals; most are gases (hydrogen, helium, nitrogen, oxygen, fluorine, neon, chlorine, argon, krypton, xenon and radon); one is a liquid (bromine); and a few are solids (carbon, phosphorus, sulfur, selenium, and iodine).Moving rightward across the standard form of periodic table, nonmetals adopt structures that have progressively fewer nearest neighbours. Polyatomic nonmetals have structures with either three nearest neighbours, as is the case (for example) with carbon (in its standard state of graphite), or two nearest neighbours (for example) in the case of sulfur. Diatomic nonmetals, such as hydrogen, have one nearest neighbour, and the monatomic noble gases, such as helium, have none. This gradual fall in the number of nearest neighbours is associated with a reduction in metallic character and an increase in nonmetallic character. The distinction between the three categories of nonmetals, in terms of receding metallicity is not absolute. Boundary overlaps occur as outlying elements in each category show (or begin to show) less-distinct, hybrid-like or atypical properties.Although five times more elements are metals than nonmetals, two of the nonmetals—hydrogen and helium—make up over 99 per cent of the observable Universe, and one—oxygen—makes up close to half of the Earth's crust, oceans and atmosphere. Living organisms are also composed almost entirely of nonmetals, and nonmetals form many more compounds than metals.