Hemoglobin a hemoglobinpatie
... 2 copies are deleted: RBC are of decreased size (microcytic) and reduced Hb concentration (hypochromic), individual is usually not anemic 3 copies are deleted: moderately severe microcytic hypochromic anemia with ...
... 2 copies are deleted: RBC are of decreased size (microcytic) and reduced Hb concentration (hypochromic), individual is usually not anemic 3 copies are deleted: moderately severe microcytic hypochromic anemia with ...
Question paper - Unit F224/01 - Energy, reproduction and
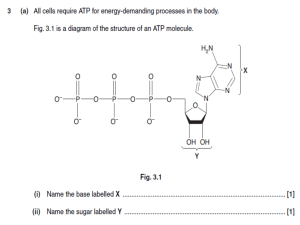
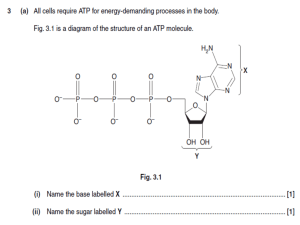
... (b) Plant cells can use the nitrate in the hydroponic mineral solution to make amino acids. These amino acids can then be used to synthesise the proteins a plant needs for its growth. (i) ...
... (b) Plant cells can use the nitrate in the hydroponic mineral solution to make amino acids. These amino acids can then be used to synthesise the proteins a plant needs for its growth. (i) ...
WHAT SHOULD I KNOW ABOUT RESPIRATION NAME ANSWERS
... How many CO2 molecules are produced from one molecule of glucose? 1 glucose (C6H12O6) produces 6 CO2 molecules Compare the amount of ATP made during fermentation (without O 2) and cellular respiration (with O2) Fermentation = 2 ATP (produced during glycolysis) Compare the production of ATP’s during ...
... How many CO2 molecules are produced from one molecule of glucose? 1 glucose (C6H12O6) produces 6 CO2 molecules Compare the amount of ATP made during fermentation (without O 2) and cellular respiration (with O2) Fermentation = 2 ATP (produced during glycolysis) Compare the production of ATP’s during ...
File
... We get our energy from the __________ plants made during _______________ PHOTOSYNTHESIS when we eat plants or eat animals that ate the plants. ...
... We get our energy from the __________ plants made during _______________ PHOTOSYNTHESIS when we eat plants or eat animals that ate the plants. ...
29
... With the procedure, usually between 1 to 4 units of a person's own blood (autologos) are withdrawn, the plasma is removed and immediately reinfused, and the packed red cells are placed in frozen storage. To prevent a dramatic reduction in blood cell concentration, each unit of blood is withdrawn ove ...
... With the procedure, usually between 1 to 4 units of a person's own blood (autologos) are withdrawn, the plasma is removed and immediately reinfused, and the packed red cells are placed in frozen storage. To prevent a dramatic reduction in blood cell concentration, each unit of blood is withdrawn ove ...
Cellular oxygen utilization in health and sepsis
... Cytopathic hypoxia or cellular metabolic derangement Fink7 first used the term cytopathic hypoxia to describe the dysregulation of oxygen metabolism during sepsis; cells fail to produce adequate amounts of ATP in the presence of sufficient molecular oxygen. These terms reflect the fact that it may n ...
... Cytopathic hypoxia or cellular metabolic derangement Fink7 first used the term cytopathic hypoxia to describe the dysregulation of oxygen metabolism during sepsis; cells fail to produce adequate amounts of ATP in the presence of sufficient molecular oxygen. These terms reflect the fact that it may n ...
Anaerobic Respiration
... From here, pyruvate is eventually converted back to glucose and returned to muscle cells or stored as glycogen. ...
... From here, pyruvate is eventually converted back to glucose and returned to muscle cells or stored as glycogen. ...
Anaerobic Respiration
... From here, pyruvate is eventually converted back to glucose and returned to muscle cells or stored as glycogen. ...
... From here, pyruvate is eventually converted back to glucose and returned to muscle cells or stored as glycogen. ...
CELLULAR RESPIRTION Powerpoint
... 5. Know that electrons are stripped from glucose as it is broken down and that these electrons are carried to the electron transport chain in the form of NADH to make most of the ATP 6. Know that the entire process converts 1 molecule of glucose to 36 molecules of ATP mainly through the enzyme ATP s ...
... 5. Know that electrons are stripped from glucose as it is broken down and that these electrons are carried to the electron transport chain in the form of NADH to make most of the ATP 6. Know that the entire process converts 1 molecule of glucose to 36 molecules of ATP mainly through the enzyme ATP s ...
Hemoglobin and Cytochrome c
... aggregation of fibrinogen. At the other end of the spectrum, a protein for which very few amino acid rates are acceptable would have a very slow rate of evolution. ...
... aggregation of fibrinogen. At the other end of the spectrum, a protein for which very few amino acid rates are acceptable would have a very slow rate of evolution. ...
Chapter 4
... • All Mass spectrometers separate atoms and molecules by first converting them into ions and then separating those ions based on the ratio of their masses to their electric charges. ...
... • All Mass spectrometers separate atoms and molecules by first converting them into ions and then separating those ions based on the ratio of their masses to their electric charges. ...
Ground state reactants Ground state products Ground state
... a triplet sensitizer to molecular oxygen • It cannot occur if the sensitizer energy is significantly below 22 kcal/mol. • It can only populate the 1∆g level of molecular oxygen if the sensitizer energy is between 22 and 37 kcal/mol, since population of the 1Σg level would be energetically unfavorabl ...
... a triplet sensitizer to molecular oxygen • It cannot occur if the sensitizer energy is significantly below 22 kcal/mol. • It can only populate the 1∆g level of molecular oxygen if the sensitizer energy is between 22 and 37 kcal/mol, since population of the 1Σg level would be energetically unfavorabl ...
Early Ideas of Atoms
... Let’s begin our discussion of the atomic theory by considering a simple but important experiment that suggested matter might be made up of atoms. In the late 1700s and early 1800s, scientists began noticing that when certain substances, like hydrogen and oxygen, were combined to produce a new substa ...
... Let’s begin our discussion of the atomic theory by considering a simple but important experiment that suggested matter might be made up of atoms. In the late 1700s and early 1800s, scientists began noticing that when certain substances, like hydrogen and oxygen, were combined to produce a new substa ...
Lipid Oxidation - anslab.iastate.edu
... Reactive Oxygen Species - Ground-state oxygen (3O2) • Oxygen is the most important factor on the development of lipid oxidation • Ground state oxygen is itself a radical, with two unpaired electrons each located in a * antibonding orbital • Ground state oxygen has its outermost pair of electrons p ...
... Reactive Oxygen Species - Ground-state oxygen (3O2) • Oxygen is the most important factor on the development of lipid oxidation • Ground state oxygen is itself a radical, with two unpaired electrons each located in a * antibonding orbital • Ground state oxygen has its outermost pair of electrons p ...
Free radicals
... blood and attacts a number of biological targets, acts as vasodilator, may have a role in intracellular signaling and ...
... blood and attacts a number of biological targets, acts as vasodilator, may have a role in intracellular signaling and ...
Fibrous proteins
... two “open” coordination bonds of iron can result in irreversible conversion of Fe2+ to Fe3+. • Iron in the Fe2+ state binds oxygen reversibly; in the Fe3+ state it does not bind oxygen. • One of these two open coordination bonds is occupied by a side-chain nitrogen of a His residue (proximal histidi ...
... two “open” coordination bonds of iron can result in irreversible conversion of Fe2+ to Fe3+. • Iron in the Fe2+ state binds oxygen reversibly; in the Fe3+ state it does not bind oxygen. • One of these two open coordination bonds is occupied by a side-chain nitrogen of a His residue (proximal histidi ...
Editorial Comment
... bioenergetic efficiency. Therefore, the failure of these patients to increase Vo2 appropriately, in response to the work load, may reflect inability of their circulation to transport oxygen fast enough to regenerate the ATP required, that is, oxygen supplydemand imbalance. An oxygen supply inadequat ...
... bioenergetic efficiency. Therefore, the failure of these patients to increase Vo2 appropriately, in response to the work load, may reflect inability of their circulation to transport oxygen fast enough to regenerate the ATP required, that is, oxygen supplydemand imbalance. An oxygen supply inadequat ...
Regulation of the Tricarboxylic Acid Cycle and Poly-p
... NADH oxidase, which is associated with the process of respiratory protection of the nitrogenase system, and the level of this enzyme reflects the environmental oxygen concentration. Thus, the imposition of an oxygen limitation (Fig. 2c) resulted in an immediate, marked decrease in the level of NADH ...
... NADH oxidase, which is associated with the process of respiratory protection of the nitrogenase system, and the level of this enzyme reflects the environmental oxygen concentration. Thus, the imposition of an oxygen limitation (Fig. 2c) resulted in an immediate, marked decrease in the level of NADH ...
Unit 9 Chemical Equations and Reactions Balancing Equations Notes
... Single Replacement- a metal will _________________ a less active metal in an ionic compound OR a nonmetal will replace a less active nonmetal. Double Replacement- the metals in ionic compounds _________________ places. Combustion- an ____________________ compound containing carbon, hydrogen and some ...
... Single Replacement- a metal will _________________ a less active metal in an ionic compound OR a nonmetal will replace a less active nonmetal. Double Replacement- the metals in ionic compounds _________________ places. Combustion- an ____________________ compound containing carbon, hydrogen and some ...
Complete breakdown of Glucose:
... A) Glucose Activation: Initiate the reaction (takes energy) B) Energy Harvesting: Complete the reaction (makes energy) ...
... A) Glucose Activation: Initiate the reaction (takes energy) B) Energy Harvesting: Complete the reaction (makes energy) ...
Growth final1 - TOP Recommended Websites
... catalase H2O2 => H20 + O2 peroxidase H2O2 + NADH + H+ => 2H20 + NAD ...
... catalase H2O2 => H20 + O2 peroxidase H2O2 + NADH + H+ => 2H20 + NAD ...
- TestbankU
... A) there is much more distance [at least 10 times more] between neighboring atoms in a liquid than between neighboring atoms in a solid. B) the liquid's atoms move throughout the liquid, while the solid's atoms remain near their original locations. C) in a liquid, the individual atoms are larger. D) ...
... A) there is much more distance [at least 10 times more] between neighboring atoms in a liquid than between neighboring atoms in a solid. B) the liquid's atoms move throughout the liquid, while the solid's atoms remain near their original locations. C) in a liquid, the individual atoms are larger. D) ...
Chapter 4 Exercise Metabolism
... -Blood lactate levels low -Resting 02 consumption is .25 L*min-1 or 3.5 ml*kg-1*min-1 -Rest 2/3 fuel contributed by fats (beta oxidation) and 1/3 fuel contributed by CHO (aerobic glycolysis) ...
... -Blood lactate levels low -Resting 02 consumption is .25 L*min-1 or 3.5 ml*kg-1*min-1 -Rest 2/3 fuel contributed by fats (beta oxidation) and 1/3 fuel contributed by CHO (aerobic glycolysis) ...
Document
... As long as the molecules are in the oxy state the cleft does not appear. The cleft appears in the deoxy state, when the vessels are so small you get the greatest tendency of the clumping of the cells. The cells become sickle in the tissues, not in the lungs, and this is where the major damage is d ...
... As long as the molecules are in the oxy state the cleft does not appear. The cleft appears in the deoxy state, when the vessels are so small you get the greatest tendency of the clumping of the cells. The cells become sickle in the tissues, not in the lungs, and this is where the major damage is d ...
Oxygen
Oxygen is a chemical element with symbol O and atomic number 8. It is a member of the chalcogen group on the periodic table and is a highly reactive nonmetallic element and oxidizing agent that readily forms compounds (notably oxides) with most elements. Photosynthesis releases oxygen, and respiration consumes oxygen. Changes in phosphate are related to changes in oxygen concentrations.Oxygen was discovered independently by Carl Wilhelm Scheele, in Uppsala, in 1773 or earlier, and Joseph Priestley in Wiltshire, in 1774, but Priestley is often given priority because his work was published first. The name oxygen was coined in 1777 by Antoine Lavoisier, whose experiments with oxygen helped to discredit the then-popular phlogiston theory of combustion and corrosion. Its name derives from the Greek roots ὀξύς oxys, ""acid"", literally ""sharp"", referring to the sour taste of acids and -γενής -genes, ""producer"", literally ""begetter"", because at the time of naming, it was mistakenly thought that all acids required oxygen in their composition.