Cell Respiration Take Home Test 1. When cells break down food
... a. provides organisms an alternative to glycolysis. c. produce most of the ATP needed for life. b. produce carbon dioxide, water, and ATP. d. All of the above 10. Water is an end product in a. lactic acid formation. c. the Krebs cycle. b. fermentation. d. the electron transport system. 11. NAD+ and ...
... a. provides organisms an alternative to glycolysis. c. produce most of the ATP needed for life. b. produce carbon dioxide, water, and ATP. d. All of the above 10. Water is an end product in a. lactic acid formation. c. the Krebs cycle. b. fermentation. d. the electron transport system. 11. NAD+ and ...
Marine Mammal Dive Response
... A mitochondrion has two membranes. The inner membrane is highly folded, which greatly increase the membrane’s surface area. This improves the ability of the mitochondrion to do which of the following? A. move the cell through water B. digest metabolic wastes in the organelle C. convert solar energy ...
... A mitochondrion has two membranes. The inner membrane is highly folded, which greatly increase the membrane’s surface area. This improves the ability of the mitochondrion to do which of the following? A. move the cell through water B. digest metabolic wastes in the organelle C. convert solar energy ...
Questions for Respiration and Photoshyntesis
... split), ATP and NADPH – used in Calvin 28. Where do they occur? Thylakoid membranes 29. What are the products of the dark reactions? G3P (used to make glucose and other organic molecules), 30. Where do they occur? Stroma 31. Describe the structure of the chloroplast and mitochondria. SEE BOARD 32. W ...
... split), ATP and NADPH – used in Calvin 28. Where do they occur? Thylakoid membranes 29. What are the products of the dark reactions? G3P (used to make glucose and other organic molecules), 30. Where do they occur? Stroma 31. Describe the structure of the chloroplast and mitochondria. SEE BOARD 32. W ...
Note sheet Chap 5, Sect 3
... Chapter 5, Section 3 The main point of photosynthesis is to produce __glucose__, which is then used _______________. Most of our energy comes in the form of _ATP_, which is produced more efficiently in the presence of __oxygen___. This is called __aerobic respiration__. Where does this occur? mitoch ...
... Chapter 5, Section 3 The main point of photosynthesis is to produce __glucose__, which is then used _______________. Most of our energy comes in the form of _ATP_, which is produced more efficiently in the presence of __oxygen___. This is called __aerobic respiration__. Where does this occur? mitoch ...
Chapter #2-Newest CPub
... It is assumed that one molecule of the compound is comprised of N types of atoms, identified by the subscript i. The number of atoms of type i in the molecule is given the symbol niatom and the mass of each type of atom is given the symbol miatom. According to the fifth statement of the atomic theor ...
... It is assumed that one molecule of the compound is comprised of N types of atoms, identified by the subscript i. The number of atoms of type i in the molecule is given the symbol niatom and the mass of each type of atom is given the symbol miatom. According to the fifth statement of the atomic theor ...
Chapter 9 - Cellular Respiration
... a) consists of a series of enzymes on the inner mitochondrial membrane b) electrons are released from NADH and from FADH2 and as they are passed along the series of enzymes, they give up energy which is used to fuel a process called chemiosmosis, which drives the process of ATP synthesis using an en ...
... a) consists of a series of enzymes on the inner mitochondrial membrane b) electrons are released from NADH and from FADH2 and as they are passed along the series of enzymes, they give up energy which is used to fuel a process called chemiosmosis, which drives the process of ATP synthesis using an en ...
Stoichiometry
... Energy is given off in combustion reactions. For organic compounds, water and carbon dioxide are usual byproducts. Ex. Metabolism of food, fuel combustion: Sucrose (C22H12O11) combusts Combustion of propane Combustion of hydrogen ...
... Energy is given off in combustion reactions. For organic compounds, water and carbon dioxide are usual byproducts. Ex. Metabolism of food, fuel combustion: Sucrose (C22H12O11) combusts Combustion of propane Combustion of hydrogen ...
Cellular respiration *vs
... • Cellular respiration (a three stage process) converts glucose and oxygen to ATP. ATP is the energy in a cell. This process converts glucose and oxygen to ATP and releases carbon dioxide and water. Here is the formula: ...
... • Cellular respiration (a three stage process) converts glucose and oxygen to ATP. ATP is the energy in a cell. This process converts glucose and oxygen to ATP and releases carbon dioxide and water. Here is the formula: ...
Unit 2
... 59) A 2.0 L tank of nitrogen gas at 1.0 atm and 25 oC is connected to a 3.0 L tank of oxygen at 2.0 atm and 25 oC. When the valve is opened, allowing the gases to mix, but not react: a) What is the new pressure of the nitrogen? b) what is the new pressure of the oxygen? c) What is the total pressur ...
... 59) A 2.0 L tank of nitrogen gas at 1.0 atm and 25 oC is connected to a 3.0 L tank of oxygen at 2.0 atm and 25 oC. When the valve is opened, allowing the gases to mix, but not react: a) What is the new pressure of the nitrogen? b) what is the new pressure of the oxygen? c) What is the total pressur ...
Unit 2
... 59) A 2.0 L tank of nitrogen gas at 1.0 atm and 25 oC is connected to a 3.0 L tank of oxygen at 2.0 atm and 25 oC. When the valve is opened, allowing the gases to mix, but not react: a) What is the new pressure of the nitrogen? b) what is the new pressure of the oxygen? c) What is the total pressur ...
... 59) A 2.0 L tank of nitrogen gas at 1.0 atm and 25 oC is connected to a 3.0 L tank of oxygen at 2.0 atm and 25 oC. When the valve is opened, allowing the gases to mix, but not react: a) What is the new pressure of the nitrogen? b) what is the new pressure of the oxygen? c) What is the total pressur ...
Unit 2
... 59) A 2.0 L tank of nitrogen gas at 1.0 atm and 25 oC is connected to a 3.0 L tank of oxygen at 2.0 atm and 25 oC. When the valve is opened, allowing the gases to mix, but not react: a) What is the new pressure of the nitrogen? b) what is the new pressure of the oxygen? c) What is the total pressur ...
... 59) A 2.0 L tank of nitrogen gas at 1.0 atm and 25 oC is connected to a 3.0 L tank of oxygen at 2.0 atm and 25 oC. When the valve is opened, allowing the gases to mix, but not react: a) What is the new pressure of the nitrogen? b) what is the new pressure of the oxygen? c) What is the total pressur ...
Unit 2
... 59) A 2.0 L tank of nitrogen gas at 1.0 atm and 25 oC is connected to a 3.0 L tank of oxygen at 2.0 atm and 25 oC. When the valve is opened, allowing the gases to mix, but not react: a) What is the new pressure of the nitrogen? b) what is the new pressure of the oxygen? c) What is the total pressur ...
... 59) A 2.0 L tank of nitrogen gas at 1.0 atm and 25 oC is connected to a 3.0 L tank of oxygen at 2.0 atm and 25 oC. When the valve is opened, allowing the gases to mix, but not react: a) What is the new pressure of the nitrogen? b) what is the new pressure of the oxygen? c) What is the total pressur ...
Chapter 6 Notes - Discount Flies
... Coeffecient = a whole number written in front of a substance which indicates the number of molecules that react. Tricks for balancing: 1. Write correct formulas for reactants and products first. Don’t ever change the formula of a substance once it is written correctly. 2. Balance O and H last. ...
... Coeffecient = a whole number written in front of a substance which indicates the number of molecules that react. Tricks for balancing: 1. Write correct formulas for reactants and products first. Don’t ever change the formula of a substance once it is written correctly. 2. Balance O and H last. ...
Muscle cramps! - WordPress.com
... can have negative implications (there is not enough oxygen at the end of the transport chain to compensate for the necessary ATP needed in the increase of muscle contractions). In order for the muscles to contract ATP is extremely necessary as it is a crucial part of the sliding filament theory, whi ...
... can have negative implications (there is not enough oxygen at the end of the transport chain to compensate for the necessary ATP needed in the increase of muscle contractions). In order for the muscles to contract ATP is extremely necessary as it is a crucial part of the sliding filament theory, whi ...
Respiration Notes (chapter 8)
... Several Types of Fermentation: -Prokaryotic (Bacteria) fermentation -Sulfer Bacteria (Coal Mines) -Iron Bacteria (TITANIC Rusticles) -Decomposing Bacteria -indicated by a black color & aromatic methane smell. -found in: ...
... Several Types of Fermentation: -Prokaryotic (Bacteria) fermentation -Sulfer Bacteria (Coal Mines) -Iron Bacteria (TITANIC Rusticles) -Decomposing Bacteria -indicated by a black color & aromatic methane smell. -found in: ...
Glycolysis and fermentation
... Lactate converts back to pyruvate, this happens in the liver Pyruvate then in presence of oxygen can be broken down into CO2 + H2O by cellular respiration ...
... Lactate converts back to pyruvate, this happens in the liver Pyruvate then in presence of oxygen can be broken down into CO2 + H2O by cellular respiration ...
Document
... Lactate converts back to pyruvate, this happens in the liver Pyruvate then in presence of oxygen can be broken down into CO2 + H2O by cellular respiration ...
... Lactate converts back to pyruvate, this happens in the liver Pyruvate then in presence of oxygen can be broken down into CO2 + H2O by cellular respiration ...
(C) A glucose reserve - Ms. Ottolini`s Biology Wiki!
... (D) Attempts to increase the photosynthetic yield in field crops should involve the lowering of CO2 levels. 7. Which of the following seems most likely from the data? (A) Light produces heat, which causes increases in the rates of photosynthesis. (B) Light causes the saturation of cytochrome oxidase ...
... (D) Attempts to increase the photosynthetic yield in field crops should involve the lowering of CO2 levels. 7. Which of the following seems most likely from the data? (A) Light produces heat, which causes increases in the rates of photosynthesis. (B) Light causes the saturation of cytochrome oxidase ...
Redox - Plusnet
... O is -2, except in OF2 Group 7 are -1, except with O or F Group 1 metals are +1 Group 2 metals are +2 H is +1, except in hydrides, e.g. NaH Al is +3 The total for an ion is its charge (e.g. -1 for CN-) More electronegative atoms get negative numbers The total for a compound is 0, even in O2, Cl2 etc ...
... O is -2, except in OF2 Group 7 are -1, except with O or F Group 1 metals are +1 Group 2 metals are +2 H is +1, except in hydrides, e.g. NaH Al is +3 The total for an ion is its charge (e.g. -1 for CN-) More electronegative atoms get negative numbers The total for a compound is 0, even in O2, Cl2 etc ...
Reading guide - Chemistry Courses: About
... 4. How do some cells use actin to move? Explain, chemically, how this is an energy utilizing process. 5. How are microtubules structurally similar to microfilaments, and how are they different? 6. Why are there drugs that target microtubules? How do colchicine and taxol work in opposite ways to prod ...
... 4. How do some cells use actin to move? Explain, chemically, how this is an energy utilizing process. 5. How are microtubules structurally similar to microfilaments, and how are they different? 6. Why are there drugs that target microtubules? How do colchicine and taxol work in opposite ways to prod ...
Reactive Oxygen Species
... A scheme of the catalytic cycle of cytochrome P450-containing monooxygenases. The binding of the substrate (RH) to ferric P450 (a) results in the formation of the substrate complex (b). The ferric P450 then accepts the first electron from CPR (cytochrome P450 reductase), thereby being reduced to the ...
... A scheme of the catalytic cycle of cytochrome P450-containing monooxygenases. The binding of the substrate (RH) to ferric P450 (a) results in the formation of the substrate complex (b). The ferric P450 then accepts the first electron from CPR (cytochrome P450 reductase), thereby being reduced to the ...
document
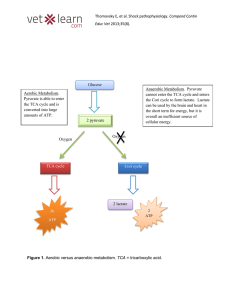
... IN which part of living organism respiration takes place? What are the main differences between aerobic and anaerobic respiration? What is the difference between aerobic and anaerobic respiration in the amount of energy released from one molecule of glucose? Victims of drowning who have stopped brea ...
... IN which part of living organism respiration takes place? What are the main differences between aerobic and anaerobic respiration? What is the difference between aerobic and anaerobic respiration in the amount of energy released from one molecule of glucose? Victims of drowning who have stopped brea ...
Anaerobic Respiration
... The initial concentration of each of these substances in the media was the same. The oxygen uptake of each culture was measured over a period of time. The results are shown in the graph below. Lactic acid ...
... The initial concentration of each of these substances in the media was the same. The oxygen uptake of each culture was measured over a period of time. The results are shown in the graph below. Lactic acid ...
Oxygen
Oxygen is a chemical element with symbol O and atomic number 8. It is a member of the chalcogen group on the periodic table and is a highly reactive nonmetallic element and oxidizing agent that readily forms compounds (notably oxides) with most elements. Photosynthesis releases oxygen, and respiration consumes oxygen. Changes in phosphate are related to changes in oxygen concentrations.Oxygen was discovered independently by Carl Wilhelm Scheele, in Uppsala, in 1773 or earlier, and Joseph Priestley in Wiltshire, in 1774, but Priestley is often given priority because his work was published first. The name oxygen was coined in 1777 by Antoine Lavoisier, whose experiments with oxygen helped to discredit the then-popular phlogiston theory of combustion and corrosion. Its name derives from the Greek roots ὀξύς oxys, ""acid"", literally ""sharp"", referring to the sour taste of acids and -γενής -genes, ""producer"", literally ""begetter"", because at the time of naming, it was mistakenly thought that all acids required oxygen in their composition.