www.xtremepapers.net
... Permission to reproduce items where third-party owned material protected by copyright is included has been sought and cleared where possible. Every reasonable effort has been made by the publisher (UCLES) to trace copyright holders, but if any items requiring clearance have unwittingly been included ...
... Permission to reproduce items where third-party owned material protected by copyright is included has been sought and cleared where possible. Every reasonable effort has been made by the publisher (UCLES) to trace copyright holders, but if any items requiring clearance have unwittingly been included ...
Environmental Chemistry
... (a) for aerobic water having a pE = +11, and (b) for anaerobic water with pE = -3 pE = 14.15 – (5/4)pH – (1/8)log([NH4+] / [NO3-]) 11 = 14.15 – (5/4) x 6 – (1/8)log([NH4+] / [NO3-]) log([NH4+] / [NO3-]) = -8(4.35) = -34.8 [NH4+] / [NO3-] = 1.6 x 10-35 pE = 14.15 – (5/4)pH – (1/8)log([NH4+] / [NO3-]) ...
... (a) for aerobic water having a pE = +11, and (b) for anaerobic water with pE = -3 pE = 14.15 – (5/4)pH – (1/8)log([NH4+] / [NO3-]) 11 = 14.15 – (5/4) x 6 – (1/8)log([NH4+] / [NO3-]) log([NH4+] / [NO3-]) = -8(4.35) = -34.8 [NH4+] / [NO3-] = 1.6 x 10-35 pE = 14.15 – (5/4)pH – (1/8)log([NH4+] / [NO3-]) ...
CHEM 1405 Practice Exam #2
... A) Solid sodium carbonate is heated to give solid sodium oxide and carbon dioxide gas. B) Sodium carbonate decomposes to sodium oxide and carbon dioxide. C) Sodium carbonate decomposes to sodium oxide and carbon dioxide gas. D) Sodium carbonate is heated to give sodium oxide and carbon dioxide. 20) ...
... A) Solid sodium carbonate is heated to give solid sodium oxide and carbon dioxide gas. B) Sodium carbonate decomposes to sodium oxide and carbon dioxide. C) Sodium carbonate decomposes to sodium oxide and carbon dioxide gas. D) Sodium carbonate is heated to give sodium oxide and carbon dioxide. 20) ...
The p-Block Elements The p-Block Elements
... boiling points are 198.4 and 239.7 K respectively. In the solid and liquid states, it is associated through hydrogen bonds as in the case of water and that accounts for its higher melting and boiling points than expected on the basis of its molecular mass. The ammonia molecule is trigonal pyramidal ...
... boiling points are 198.4 and 239.7 K respectively. In the solid and liquid states, it is associated through hydrogen bonds as in the case of water and that accounts for its higher melting and boiling points than expected on the basis of its molecular mass. The ammonia molecule is trigonal pyramidal ...
Exam Review Chapter 18-Equilibrium
... 6. What is the effect of adding more CO2 to the following equilibrium reaction? CO2 + H2O↔ H2CO3 a. More H2CO3 is produced. b. More H2O is produced. c. The equilibrium d. No Change 7. Two opposing reactions (A + B ↔C + D) occurring simultaneously at the same rate is an example of: a. reversibility. ...
... 6. What is the effect of adding more CO2 to the following equilibrium reaction? CO2 + H2O↔ H2CO3 a. More H2CO3 is produced. b. More H2O is produced. c. The equilibrium d. No Change 7. Two opposing reactions (A + B ↔C + D) occurring simultaneously at the same rate is an example of: a. reversibility. ...
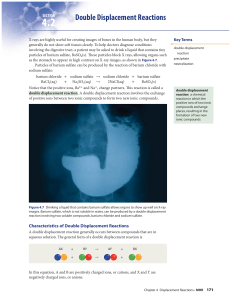
Double Displacement Reactions
... Just as there are different types of single displacement reactions, there are different types of double displacement reactions. On the following pages, you will learn about these double displacement reactions: • a reaction that forms a solid • a reaction that forms a gas • a reaction that forms wate ...
... Just as there are different types of single displacement reactions, there are different types of double displacement reactions. On the following pages, you will learn about these double displacement reactions: • a reaction that forms a solid • a reaction that forms a gas • a reaction that forms wate ...
Net ionic equation
... Why Ionic Compounds Dissolve in Water The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is ...
... Why Ionic Compounds Dissolve in Water The forces holding an ionic compound together are the strong electrical attraction that exists between cations and anions. It is therefore somewhat surprising that ionic compounds will dissolve in water. The reason some ionic compounds will dissolve in water is ...
chem A exercise package C
... Many formulas for substances cannot be explained in terms of ionic bonding. Consider the substance Cl2O. Both the chlorine and the oxygen atom need more electrons for a stable electron population. A model proposed that would allow both atoms to gain electrons is shown in the diagram on this page. Th ...
... Many formulas for substances cannot be explained in terms of ionic bonding. Consider the substance Cl2O. Both the chlorine and the oxygen atom need more electrons for a stable electron population. A model proposed that would allow both atoms to gain electrons is shown in the diagram on this page. Th ...
Chemistry Log Books - Social Circle City Schools
... Classify the following as examples of either physical properties or chemical properties. a. A piece of metal has a mass of 60.0 g._____ b. Metals react with acids to produce hydrogen gas.____ c. Tommy’s weight on the moon 40 lbs.____ d. Susan’s weight on earth is 45.0 kg.____ e. The density of water ...
... Classify the following as examples of either physical properties or chemical properties. a. A piece of metal has a mass of 60.0 g._____ b. Metals react with acids to produce hydrogen gas.____ c. Tommy’s weight on the moon 40 lbs.____ d. Susan’s weight on earth is 45.0 kg.____ e. The density of water ...
Practice Problem Set #6
... 1. Write balanced chemical equations for the reaction of hydrogen gas with oxygen, chlorine, and nitrogen. 2. Write a balanced chemical equation for the preparation of H2 (and CO) by the reaction of CH4 and water. Using a table of thermodynamic data, calculate ∆H°, ∆G°, and ∆S° for this reaction. ...
... 1. Write balanced chemical equations for the reaction of hydrogen gas with oxygen, chlorine, and nitrogen. 2. Write a balanced chemical equation for the preparation of H2 (and CO) by the reaction of CH4 and water. Using a table of thermodynamic data, calculate ∆H°, ∆G°, and ∆S° for this reaction. ...
Description: This is an advanced placement course designed to
... With the introduction in 1999 of a required laboratory-based question on the free-response section of the AP Chemistry Exam, the inclusion of appropriate experiments into each AP Chemistry course is increasingly important….. It is unlikely that every student will complete all of the 22 laboratory ex ...
... With the introduction in 1999 of a required laboratory-based question on the free-response section of the AP Chemistry Exam, the inclusion of appropriate experiments into each AP Chemistry course is increasingly important….. It is unlikely that every student will complete all of the 22 laboratory ex ...
Atoms and Molecules
... ratio of elements in a crystal of that salt. • Atoms in a crystal do not form molecules with a definitive size and number of atoms as in covalent bonds. ...
... ratio of elements in a crystal of that salt. • Atoms in a crystal do not form molecules with a definitive size and number of atoms as in covalent bonds. ...
Chemistry Name Mr. Reger Review Guide – Ch. 9
... a) What is the theoretical yield of NaClO3 if 4.0mol Cl2 is reacted with excess NaOH? b) If 94.2g NaClO3 is obtained, what is the % yield? c) A different student performs the same reaction and calculates her % yield to be 47%. What mass of NaClO3 did she obtain? 12. A student isolates 1.6g of a prod ...
... a) What is the theoretical yield of NaClO3 if 4.0mol Cl2 is reacted with excess NaOH? b) If 94.2g NaClO3 is obtained, what is the % yield? c) A different student performs the same reaction and calculates her % yield to be 47%. What mass of NaClO3 did she obtain? 12. A student isolates 1.6g of a prod ...
Review Chapters 4-6 problems Chem 105 Final Sp07
... 31. The combustion of propane involves the reaction of C3H8 with ________. 32. The percent yield of a chemical reaction is calculated by dividing the ________ yield by the theoretical yield and multiplying this ratio by 100%. 33. A French scientist named __________ introduced the law of conservation ...
... 31. The combustion of propane involves the reaction of C3H8 with ________. 32. The percent yield of a chemical reaction is calculated by dividing the ________ yield by the theoretical yield and multiplying this ratio by 100%. 33. A French scientist named __________ introduced the law of conservation ...
Balancing a Chemical Equation
... Separate the reactants from the products with an arrow. Use plus signs to separate the reactants and the products. NaHCO3(s) + HCl(aq) → NaCl(aq) + H2O(l) + CO2(g) ...
... Separate the reactants from the products with an arrow. Use plus signs to separate the reactants and the products. NaHCO3(s) + HCl(aq) → NaCl(aq) + H2O(l) + CO2(g) ...
Chemistry
... feasibility of a reaction – the Gibbs free energy (∆G). For aqueous redox reactions, the more convenient notion of electrode potential (E) is used, and the resultant cell potential (Ecell) gives a measure of thermodynamics feasibility instead. The chemical kinetics facet of a reaction can be underst ...
... feasibility of a reaction – the Gibbs free energy (∆G). For aqueous redox reactions, the more convenient notion of electrode potential (E) is used, and the resultant cell potential (Ecell) gives a measure of thermodynamics feasibility instead. The chemical kinetics facet of a reaction can be underst ...
AP Chem II Instructor: Mr. Malasky Name Period ______ Due Date
... ____ 5. The value of ΔG˚ at 25˚C for the decomposition of gaseous sulfur dioxide to solid elemental sulfur and gaseous oxygen, SO2(g) → 2 S (s,rhombic) + O2(g) is __________ kJ/mol. A) +395.2 B) +269.9 C) -269.9 D) +300.4 E) -300.4 ____ 6. The value of ΔG˚ at 25˚C for the formation of POCl3 from it ...
... ____ 5. The value of ΔG˚ at 25˚C for the decomposition of gaseous sulfur dioxide to solid elemental sulfur and gaseous oxygen, SO2(g) → 2 S (s,rhombic) + O2(g) is __________ kJ/mol. A) +395.2 B) +269.9 C) -269.9 D) +300.4 E) -300.4 ____ 6. The value of ΔG˚ at 25˚C for the formation of POCl3 from it ...
1 Mole
... Reactants: chemicals that react Products: chemicals that are formed e.x. sodium + oxygen sodium oxide Na(s) + O2(g) Na2O(s) ...
... Reactants: chemicals that react Products: chemicals that are formed e.x. sodium + oxygen sodium oxide Na(s) + O2(g) Na2O(s) ...
Ch 06
... When the reacting system reaches the activated complex stage it may reverse to reactants, or it may continue to form product molecules. In either case, repulsion forces push the molecules apart, converting potential energy to kinetic energy. Overall, there are potential energy changes as bonds are b ...
... When the reacting system reaches the activated complex stage it may reverse to reactants, or it may continue to form product molecules. In either case, repulsion forces push the molecules apart, converting potential energy to kinetic energy. Overall, there are potential energy changes as bonds are b ...
4. chemical kinetics
... Molecularity of a reaction is the number of reactant molecules taking part in rate determining step. ...
... Molecularity of a reaction is the number of reactant molecules taking part in rate determining step. ...
File
... What is the final concentration of a 1.35mol/L solution which is diluted from 125mL to 3.25L? (2%) ...
... What is the final concentration of a 1.35mol/L solution which is diluted from 125mL to 3.25L? (2%) ...
2012 Chem 13 News Exam
... The intermolecular attractions in C2H6(g) are stronger than they are in H2(g). ...
... The intermolecular attractions in C2H6(g) are stronger than they are in H2(g). ...
xmas review questions 01516 with hints
... This question will be easier following a further discussion of acids and bases in an upcoming topic. For now try to connect it to a gas producing reaction in our reaction type lab when an acid (vinegar) was added to one of these. The ion of interest is usually insoluble but not with a group 1 ion. W ...
... This question will be easier following a further discussion of acids and bases in an upcoming topic. For now try to connect it to a gas producing reaction in our reaction type lab when an acid (vinegar) was added to one of these. The ion of interest is usually insoluble but not with a group 1 ion. W ...
Fall 2008 Blank Final Exam
... Select the best multiple-choice answer by filling in the corresponding circle on the rear page of the answer sheet. If you have any questions before the exam, please ask. If you have any questions during the exam, please ask the proctor. Open and start this exam when instructed. When finished, place ...
... Select the best multiple-choice answer by filling in the corresponding circle on the rear page of the answer sheet. If you have any questions before the exam, please ask. If you have any questions during the exam, please ask the proctor. Open and start this exam when instructed. When finished, place ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.