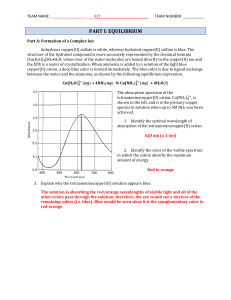
Chem 12 Prov Exam PLO Review
... • reactions are the result of collisions between reactant particles • not all collisions are successful • sufficient kinetic energy (KE) and favourable geometry are required • to increase the rate of a reaction one must increase the frequency of successful collisions • energy changes are involved in ...
... • reactions are the result of collisions between reactant particles • not all collisions are successful • sufficient kinetic energy (KE) and favourable geometry are required • to increase the rate of a reaction one must increase the frequency of successful collisions • energy changes are involved in ...
Chapter 4 Power Point Quiz
... How many grams of glucose (C6H12O6) form when 4.40 g of CO2 react? a) b) c) d) ...
... How many grams of glucose (C6H12O6) form when 4.40 g of CO2 react? a) b) c) d) ...
Answers - Scioly.org
... The pH value of pure water is 7.0, whereas natural rainwater is weakly acidic. This is caused by dissolution of atmospheric carbon dioxide. In many areas, however, rainwater is more acidic. This has many causes, some of which are natural and some of which derive from human activity. In the atmospher ...
... The pH value of pure water is 7.0, whereas natural rainwater is weakly acidic. This is caused by dissolution of atmospheric carbon dioxide. In many areas, however, rainwater is more acidic. This has many causes, some of which are natural and some of which derive from human activity. In the atmospher ...
Catalysis
... 4. Enzyme catalysed reactions are much more sensitive to catalytic poisons such as HCN, H2S, CS2 etc. The inhibitors interact with the active functional groups present on the enzyme surface and often reduce or completely destroy the catalytic activity of the enzymes 5. The activity of certain enzym ...
... 4. Enzyme catalysed reactions are much more sensitive to catalytic poisons such as HCN, H2S, CS2 etc. The inhibitors interact with the active functional groups present on the enzyme surface and often reduce or completely destroy the catalytic activity of the enzymes 5. The activity of certain enzym ...
Chapter 4 Chemical Reactions and Solution Stoichiometry 4.1
... When mixed, a double displacement reaction takes place, forming the soluble compound NaNO3 and the insoluble compound AgCl. In the reaction vessel the Ag+ and Cl ions combine, and a white solid precipitates from the solution. As the solid precipitates, the Na+ and NO3 ions remain in solution. The ...
... When mixed, a double displacement reaction takes place, forming the soluble compound NaNO3 and the insoluble compound AgCl. In the reaction vessel the Ag+ and Cl ions combine, and a white solid precipitates from the solution. As the solid precipitates, the Na+ and NO3 ions remain in solution. The ...
FREE Sample Here
... B) protons and neutrons are shared by two atoms so as to satisfy the requirements of both atoms C) outer-shell electrons of two atoms are shared so as to satisfactorily fill their respective orbitals D) outer-shell electrons of one atom are transferred to fill the inner electron shell of another ato ...
... B) protons and neutrons are shared by two atoms so as to satisfy the requirements of both atoms C) outer-shell electrons of two atoms are shared so as to satisfactorily fill their respective orbitals D) outer-shell electrons of one atom are transferred to fill the inner electron shell of another ato ...
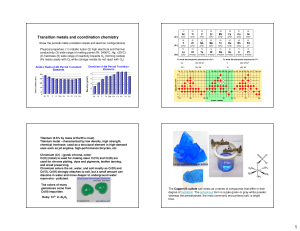
biogenic s, p, d-block elements, biological role, application in medicine
... electrons. Among p-elements there are elements that can be both cations and anions (A1, Ca, Ti, Se, Pb, PB, Sb, Bi) or only anions (В, С, Sі, N, Р, Аs, О, Те, Р, СІ, Вг, І, Аt). All cations, except A13+(1s22s22p6)have a structure of external electronic shell (n-1)d10ns2, where n is the number of per ...
... electrons. Among p-elements there are elements that can be both cations and anions (A1, Ca, Ti, Se, Pb, PB, Sb, Bi) or only anions (В, С, Sі, N, Р, Аs, О, Те, Р, СІ, Вг, І, Аt). All cations, except A13+(1s22s22p6)have a structure of external electronic shell (n-1)d10ns2, where n is the number of per ...
Enzymes - stephen fleenor
... performing a hydrolysis reaction compaction of sugar grains into a hard substance dehydration synthesis reactions the association of hydrophobic tails with hydrophilic sugar molecules ...
... performing a hydrolysis reaction compaction of sugar grains into a hard substance dehydration synthesis reactions the association of hydrophobic tails with hydrophilic sugar molecules ...
Flexbook - Ions and Ion Formation
... www.ck12.org • Atoms with high ionization energy and high electron affinity (nonmetals) tend to gain electrons and become negative ions. • Atoms with high ionization energy and low electron affinity (noble gases) tend to neither gain nor lose electrons. • Atoms that tend to lose electrons will gene ...
... www.ck12.org • Atoms with high ionization energy and high electron affinity (nonmetals) tend to gain electrons and become negative ions. • Atoms with high ionization energy and low electron affinity (noble gases) tend to neither gain nor lose electrons. • Atoms that tend to lose electrons will gene ...
Unit (1)
... 15- On dissolving in water, acids give positive ………….. ion and alkalies give negative ………….. ion. 16- Mineral acids are formed when hydrogen is joined with one of ………….. atomic groups except ………….. group. 17- The symbol of all mineral acids begin with ………….. atom, while the symbol of all bases end b ...
... 15- On dissolving in water, acids give positive ………….. ion and alkalies give negative ………….. ion. 16- Mineral acids are formed when hydrogen is joined with one of ………….. atomic groups except ………….. group. 17- The symbol of all mineral acids begin with ………….. atom, while the symbol of all bases end b ...
rate
... reach its “flash point”. The flash point is the temperature at which the vapors spontaneously combust in the presence of atmospheric oxygen. The combustion of grease produces combustion products and some nasty smelly once such as acrolein. But the take home message is that grease will ignite when th ...
... reach its “flash point”. The flash point is the temperature at which the vapors spontaneously combust in the presence of atmospheric oxygen. The combustion of grease produces combustion products and some nasty smelly once such as acrolein. But the take home message is that grease will ignite when th ...
Chemistry Entrance Material for Grade 11 to 12 Answer Key
... In all multiple choice questions, more than answer could be correct Section №: 1 Pure Substances Know where gaseous elements are located in the periodic table 01. What are the elements that are normally found as gases? H, N,O,F Cl, and Nobel gas 02. Where are these gaseous elements placed in the per ...
... In all multiple choice questions, more than answer could be correct Section №: 1 Pure Substances Know where gaseous elements are located in the periodic table 01. What are the elements that are normally found as gases? H, N,O,F Cl, and Nobel gas 02. Where are these gaseous elements placed in the per ...
Energy is the essence of chemistry It determines which reaction can
... • A negative ion is next to a positive ion. The strong attraction between these ions causes a high melting and boiling temperature. Many ionic solids are soluble in water An electrolyte solution. It conducts electricity Many are also sparingly soluble in water Calcium phosphate in bones is very spar ...
... • A negative ion is next to a positive ion. The strong attraction between these ions causes a high melting and boiling temperature. Many ionic solids are soluble in water An electrolyte solution. It conducts electricity Many are also sparingly soluble in water Calcium phosphate in bones is very spar ...
17.2.3 Interhalogen compounds(65-67)
... T-shaped structure of CzV symmetry, consistent with the presence of 10 electrons in the valency shell of the central atom (Fig. 17.7a,b). A notable feature of both structures is the slight deviation from colinearity of the apical F-X-F bonds, the angle being 175.0" for ClF3 and 172.4' for BrF3; this ...
... T-shaped structure of CzV symmetry, consistent with the presence of 10 electrons in the valency shell of the central atom (Fig. 17.7a,b). A notable feature of both structures is the slight deviation from colinearity of the apical F-X-F bonds, the angle being 175.0" for ClF3 and 172.4' for BrF3; this ...
Skill Practice 1
... c) How many neutrons does the nucleus of this ion have? 4. Tritium (an isotope of hydrogen) has 2 neutrons. How many protons does it have? What is its mass number? 5. What is the charge on a magnesium ion that has 10 electrons? 6. How many neutrons are there in a chromium atom with a mass number of ...
... c) How many neutrons does the nucleus of this ion have? 4. Tritium (an isotope of hydrogen) has 2 neutrons. How many protons does it have? What is its mass number? 5. What is the charge on a magnesium ion that has 10 electrons? 6. How many neutrons are there in a chromium atom with a mass number of ...
The Chemical Context of Life
... Figure 2.7—Electron distribution diagrams for the first 18 elements in the periodic table Hydrogen 1H ...
... Figure 2.7—Electron distribution diagrams for the first 18 elements in the periodic table Hydrogen 1H ...
111 Exam I Outline
... E. Double Exchange (Ion Exchange) Reactions 1. In a double displacement (ion exchange) reaction, the positive end and negative end of compounds "change partners" to form new products: ...
... E. Double Exchange (Ion Exchange) Reactions 1. In a double displacement (ion exchange) reaction, the positive end and negative end of compounds "change partners" to form new products: ...
summer fun - West Windsor-Plainsboro Regional School District
... CaI2 MnO2 Li2O FeI3 Cu3PO4 PCl3 NaCN Cs3N Zn (NO3)2 N2O HF (aq) ...
... CaI2 MnO2 Li2O FeI3 Cu3PO4 PCl3 NaCN Cs3N Zn (NO3)2 N2O HF (aq) ...
In Class Overview of Chapter
... kinetics and thermodynamics are pretty distinct. Thermodynamics is about the equilibrium concentrations of products and reactants and tells us nothing about how fast equilibrium is reached. Kinetics tells us how fast a reaction will occur, but doesn’t tell us the extent or direction of a reaction. E ...
... kinetics and thermodynamics are pretty distinct. Thermodynamics is about the equilibrium concentrations of products and reactants and tells us nothing about how fast equilibrium is reached. Kinetics tells us how fast a reaction will occur, but doesn’t tell us the extent or direction of a reaction. E ...
111 Exam I Outline
... E. Double Exchange (Ion Exchange) Reactions 1. In a double displacement (ion exchange) reaction, the positive end and negative end of compounds "change partners" to form new products: ...
... E. Double Exchange (Ion Exchange) Reactions 1. In a double displacement (ion exchange) reaction, the positive end and negative end of compounds "change partners" to form new products: ...
Part I - American Chemical Society
... The USNCO Subcommittee is conducting a survey in an effort to determine the impact of the Olympiad program on students. The first phase of this effort is represented by several questions added to the end of this year's exam, which should be answered on the same Scantron sheet students use for the ex ...
... The USNCO Subcommittee is conducting a survey in an effort to determine the impact of the Olympiad program on students. The first phase of this effort is represented by several questions added to the end of this year's exam, which should be answered on the same Scantron sheet students use for the ex ...
Chapter 11 * Chemical Reactions
... participate in the formation of the nonaqueous product are written. General Equation: Na2SO4 (aq) + Ba(NO3)2 (aq 2NaNO3 (aq) + BaSO4 (s) Ionic Equation: 2Na+1 (aq) + SO4-2 (aq) + Ba+2 (aq) + 2NO3-1 (aq) 2Na+1 (aq) + 2 NO3-1 (aq) + BaSO4 (s) Net Ionic Equation: Ba+2 (aq) + SO4-2 (aq) BaSO4 (s) ...
... participate in the formation of the nonaqueous product are written. General Equation: Na2SO4 (aq) + Ba(NO3)2 (aq 2NaNO3 (aq) + BaSO4 (s) Ionic Equation: 2Na+1 (aq) + SO4-2 (aq) + Ba+2 (aq) + 2NO3-1 (aq) 2Na+1 (aq) + 2 NO3-1 (aq) + BaSO4 (s) Net Ionic Equation: Ba+2 (aq) + SO4-2 (aq) BaSO4 (s) ...
chapter 9: aqueous solutions
... 1. write the separate aqueous ions (including their correct charges) on the right side 2. write the formula of the compound followed by an arrow 3. balance using coefficients 4. add state symbols (state of pure substance on the left, ((s) usually), (aq) for ions on the right) Example 1: Solid Sodium ...
... 1. write the separate aqueous ions (including their correct charges) on the right side 2. write the formula of the compound followed by an arrow 3. balance using coefficients 4. add state symbols (state of pure substance on the left, ((s) usually), (aq) for ions on the right) Example 1: Solid Sodium ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.