Chapter 2
... levels, or shells, of the atom. Chemical behavior depends on the number of valence electrons, those in the outermost shell. An atom with an incomplete valence shell is reactive. Electrons move within orbitals, three-dimensional spaces with specific shapes located within successive shells. Web/CD Act ...
... levels, or shells, of the atom. Chemical behavior depends on the number of valence electrons, those in the outermost shell. An atom with an incomplete valence shell is reactive. Electrons move within orbitals, three-dimensional spaces with specific shapes located within successive shells. Web/CD Act ...
AP Biology
... levels, or shells, of the atom. Chemical behavior depends on the number of valence electrons, those in the outermost shell. An atom with an incomplete valence shell is reactive. Electrons move within orbitals, three-dimensional spaces with specific shapes located within successive shells. Web/CD Act ...
... levels, or shells, of the atom. Chemical behavior depends on the number of valence electrons, those in the outermost shell. An atom with an incomplete valence shell is reactive. Electrons move within orbitals, three-dimensional spaces with specific shapes located within successive shells. Web/CD Act ...
1.Using the table above, decide if the element mercury (Hg) should
... 6. Below is a table showing the various methods used to extract metals from metal ores. ...
... 6. Below is a table showing the various methods used to extract metals from metal ores. ...
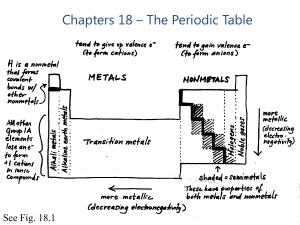
Chapters 18 – The Periodic Table
... 3. Sulfur trioxide (SO3) and sulfuric acid (H2SO4). SO3, formed from SO2 over V2O5 catalysts, is then converted to sulfuric acid. Sulfuric acid is the cheapest strong acid and is so widely used in industry that its production level is an indicator of a nation’s economic strength. Strong dehydrating ...
... 3. Sulfur trioxide (SO3) and sulfuric acid (H2SO4). SO3, formed from SO2 over V2O5 catalysts, is then converted to sulfuric acid. Sulfuric acid is the cheapest strong acid and is so widely used in industry that its production level is an indicator of a nation’s economic strength. Strong dehydrating ...
CHM1 Review for Exam 9 Topics 1. Reaction Types a. Combustion
... 7. Which equation represents a double replacement reaction? (1) 2 Na + 2 H2O 2 NaOH + H2 (2) CaCO3 CaO + CO2 (3) AgNO3 + HCl LiCl + HNO3 (4) CH4 + 2 O2 CO2 + 2 H2O 8. One hundred grams of water is saturated with NH4Cl at 50°C. According to Table G, if the temperature is lowered to 10°C. what ...
... 7. Which equation represents a double replacement reaction? (1) 2 Na + 2 H2O 2 NaOH + H2 (2) CaCO3 CaO + CO2 (3) AgNO3 + HCl LiCl + HNO3 (4) CH4 + 2 O2 CO2 + 2 H2O 8. One hundred grams of water is saturated with NH4Cl at 50°C. According to Table G, if the temperature is lowered to 10°C. what ...
Sample Questions
... 16. A hydrocarbon (a compound consisting solely of carbon and hydrogen) is found to be 85.6% carbon by mass. What is the empirical formula for this compound? 17. The empirical formula of styrene is CH; its molar mass is 104.1 g/mol. What is the molecular formula of styrene? ____ ...
... 16. A hydrocarbon (a compound consisting solely of carbon and hydrogen) is found to be 85.6% carbon by mass. What is the empirical formula for this compound? 17. The empirical formula of styrene is CH; its molar mass is 104.1 g/mol. What is the molecular formula of styrene? ____ ...
Year End Chemistry Review
... If 5.0 moles of water are decomposed, how many moles of oxygen are formed? If 5.0 liters of water are decomposed, how many liters of oxygen are formed? (Notice this is the same problem and can be done the same way.) ...
... If 5.0 moles of water are decomposed, how many moles of oxygen are formed? If 5.0 liters of water are decomposed, how many liters of oxygen are formed? (Notice this is the same problem and can be done the same way.) ...
Notes - Organization of Matter
... • These particles affect the properties of an atom • Once you understand these particles and their importance in atoms, you will be able to describe and explain all types of atoms ...
... • These particles affect the properties of an atom • Once you understand these particles and their importance in atoms, you will be able to describe and explain all types of atoms ...
CHEM102 Chemistry II Spring 11-12 Mid
... C) there is no more chemistry happening. D) the reaction rate in the reverse direction is at a minimum. E) the reaction rate in the forward direction is at a maximum. 40) For a chemical reaction to occur, all of the following must happen except A) reactant particles must collide with the correct ori ...
... C) there is no more chemistry happening. D) the reaction rate in the reverse direction is at a minimum. E) the reaction rate in the forward direction is at a maximum. 40) For a chemical reaction to occur, all of the following must happen except A) reactant particles must collide with the correct ori ...
Pre-Test: 2nd semester Final Exam Review File
... True and False. Read questions 54-68 the following statements carefully. Select A for “true” and B for “false.” 54. A chemical reaction causes a chemical change. 55. Oxidation is a physical process. 56. Combustion is a chemical reaction. 57. A chemical change makes new products. 58. Elements can be ...
... True and False. Read questions 54-68 the following statements carefully. Select A for “true” and B for “false.” 54. A chemical reaction causes a chemical change. 55. Oxidation is a physical process. 56. Combustion is a chemical reaction. 57. A chemical change makes new products. 58. Elements can be ...
Saturday Study Session 1 1st Class Reactions
... Transfer may occur to form ions Oxidation – increase in oxidation state (loss of electrons); reducing agent Reduction – decrease in oxidation state (gain of electrons); oxidizing agent ...
... Transfer may occur to form ions Oxidation – increase in oxidation state (loss of electrons); reducing agent Reduction – decrease in oxidation state (gain of electrons); oxidizing agent ...
Final Exam Practice Problems: R = 0.0821 Latm/molK NA = 6.022
... 2. A substance that can't be chemically broken down into simpler substances is considered to be A) a homogeneous mixture. B) an element. C) a heterogeneous mixture. D) a compound. E) an electron. 3. Which of the following are examples of a chemical change? A) coffee brewing B) water boiling C) leave ...
... 2. A substance that can't be chemically broken down into simpler substances is considered to be A) a homogeneous mixture. B) an element. C) a heterogeneous mixture. D) a compound. E) an electron. 3. Which of the following are examples of a chemical change? A) coffee brewing B) water boiling C) leave ...
Atomic Theory - World of Teaching
... The picture below shows three objects that can be classified in the same group. Which of the following statements is true for all three of these objects? They are metals. They rust rapidly. They weigh the same. They are the same color. ...
... The picture below shows three objects that can be classified in the same group. Which of the following statements is true for all three of these objects? They are metals. They rust rapidly. They weigh the same. They are the same color. ...
Acids - Beck-Shop
... atoms are acids. Each molecule of ethanoic acid contains four hydrogen atoms, but only the hydrogen atom on the COOH group is released as H+. Even then, only about one molecule in every hundred dissociates, so ethanoic acid is a weak acid. Most organic acids, like ethanoic acid, are weak acids. ...
... atoms are acids. Each molecule of ethanoic acid contains four hydrogen atoms, but only the hydrogen atom on the COOH group is released as H+. Even then, only about one molecule in every hundred dissociates, so ethanoic acid is a weak acid. Most organic acids, like ethanoic acid, are weak acids. ...
104 Homework Packet - Rogue Community College
... Atoms of the same element that have different weights are called ________________ of that element. ...
... Atoms of the same element that have different weights are called ________________ of that element. ...
General, Organic, and Biological Chemistry
... 46) Isotopes are atoms of the same element that have A) different atomic numbers. B) the same atomic numbers but different numbers of protons. C) the same atomic numbers but different numbers of electrons. D) the same atomic number but different numbers of neutrons. E) the same atomic mass but diff ...
... 46) Isotopes are atoms of the same element that have A) different atomic numbers. B) the same atomic numbers but different numbers of protons. C) the same atomic numbers but different numbers of electrons. D) the same atomic number but different numbers of neutrons. E) the same atomic mass but diff ...
Types of Reactions notes 02 Types of chemical reactions
... NH4NO3 N2O + H2O Fe2O3 + C Fe + CO CO2 + H2O H2CO3 CuNO3 + Na2SO4 Cu2SO4 + NaNO3 ...
... NH4NO3 N2O + H2O Fe2O3 + C Fe + CO CO2 + H2O H2CO3 CuNO3 + Na2SO4 Cu2SO4 + NaNO3 ...
Chapter 3 Stoichiometry: Calculations with Chemical Formulas and
... 2 C6H12(l) + 5 O2(g) → 2 H2C6H8O4(l) + 2 H2O(g) (a) Assume that you carry out this reaction starting with 25.0 g of cyclohexane and that cyclohexane is the limiting reactant. What is the theoretical yield of adipic acid? (b) If you obtain 33.5 g of adipic acid from your reaction, what is the percent ...
... 2 C6H12(l) + 5 O2(g) → 2 H2C6H8O4(l) + 2 H2O(g) (a) Assume that you carry out this reaction starting with 25.0 g of cyclohexane and that cyclohexane is the limiting reactant. What is the theoretical yield of adipic acid? (b) If you obtain 33.5 g of adipic acid from your reaction, what is the percent ...
Document
... (g) Potassium chloride can be made by reacting potassium with chlorine. The bonding in potassium chloride is ionic. What does this information tell you about (i) the boiling point of potassium chloride, ...
... (g) Potassium chloride can be made by reacting potassium with chlorine. The bonding in potassium chloride is ionic. What does this information tell you about (i) the boiling point of potassium chloride, ...
chemical reaction?
... Modeling Chemical Reactions • The chemical symbols in a formula tell what types of elements make up a substance • The ________________ in a chemical formula tell how many of each type of atom are in a molecule • Example: The chemical formula for rust, or iron oxide, is Fe2O3. How many iron atoms an ...
... Modeling Chemical Reactions • The chemical symbols in a formula tell what types of elements make up a substance • The ________________ in a chemical formula tell how many of each type of atom are in a molecule • Example: The chemical formula for rust, or iron oxide, is Fe2O3. How many iron atoms an ...
Atomic Structure
... found in the __________ of the atom Neutrons – have __________charge and are also found in the __________of an atom Electrons – have a __________charge and are found __________ of the nucleus Nucleus – made up of __________and __________, has an overall __________ ...
... found in the __________ of the atom Neutrons – have __________charge and are also found in the __________of an atom Electrons – have a __________charge and are found __________ of the nucleus Nucleus – made up of __________and __________, has an overall __________ ...
Chapter 7-8-9
... 22. Why do atoms share electrons in covalent bonds? a. to become ions and attract each other b. to attain a noble-gas electron configuration c. to become more polar d. to increase their atomic numbers 23. Which molecule has a single covalent bond? a. CO b. Cl c. CO d. N 24. What causes water molecul ...
... 22. Why do atoms share electrons in covalent bonds? a. to become ions and attract each other b. to attain a noble-gas electron configuration c. to become more polar d. to increase their atomic numbers 23. Which molecule has a single covalent bond? a. CO b. Cl c. CO d. N 24. What causes water molecul ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.