Part II - American Chemical Society
... Part II of this test requires that student answers be written in a response booklet with blank pages. Only this "Blue Book" is graded for a score on Part II. Testing materials, scratch paper, and the "Blue Book" should be made available to the student only during the examination period. All testing ...
... Part II of this test requires that student answers be written in a response booklet with blank pages. Only this "Blue Book" is graded for a score on Part II. Testing materials, scratch paper, and the "Blue Book" should be made available to the student only during the examination period. All testing ...
Prior knowledge catch-up student sheet for Chapter 3 Quantitative
... Chemical reactions can be represented using a formula to show reactants and products in a chemical equation, with an arrow in between. An equation must be balanced with the same number of each type of atom on each side of the arrow. To make equations balance, numbers may have to be added in front of ...
... Chemical reactions can be represented using a formula to show reactants and products in a chemical equation, with an arrow in between. An equation must be balanced with the same number of each type of atom on each side of the arrow. To make equations balance, numbers may have to be added in front of ...
quiz questions chapters 1
... B) Atoms of free elements have an oxidation number of zero. C) An atom's oxidation state signifies the number of charges for that atom in a molecule. D) Oxidation numbers allow easy identification of which atoms gained or lost electrons. ...
... B) Atoms of free elements have an oxidation number of zero. C) An atom's oxidation state signifies the number of charges for that atom in a molecule. D) Oxidation numbers allow easy identification of which atoms gained or lost electrons. ...
Dimensional Analysis (Conversions)
... Often called the “central science”, chemistry is truly the best class you will ever take in high school. My goal is to prepare you for the AP exam, for college chemistry and for life as an informed member of our republic. You will learn to move beyond the memorization of subject matter (although it ...
... Often called the “central science”, chemistry is truly the best class you will ever take in high school. My goal is to prepare you for the AP exam, for college chemistry and for life as an informed member of our republic. You will learn to move beyond the memorization of subject matter (although it ...
Matter- Types and Changes
... Chemical Formulas • CO2 contains 1 atom of carbon and two atoms of oxygen all chemically linked. • H2SO4 contains 2 hydrogen, 1 sulfur, and 4 oxygen atoms. • (NH4)2C2O4 - A subscript outside parentheses applies to everything within the parentheses; 2 N, 8 H, 2 C, 4 O ...
... Chemical Formulas • CO2 contains 1 atom of carbon and two atoms of oxygen all chemically linked. • H2SO4 contains 2 hydrogen, 1 sulfur, and 4 oxygen atoms. • (NH4)2C2O4 - A subscript outside parentheses applies to everything within the parentheses; 2 N, 8 H, 2 C, 4 O ...
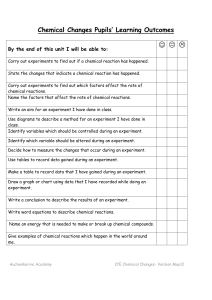
Chemical Reactions Unit Pupils` Learning Outcomes
... Identify variables which should be controlled during an experiment. Identify which variable should be altered during an experiment. Decide how to measure the changes that occur during an experiment. Use tables to record data gained during an experiment. Make a table to record data that I have gained ...
... Identify variables which should be controlled during an experiment. Identify which variable should be altered during an experiment. Decide how to measure the changes that occur during an experiment. Use tables to record data gained during an experiment. Make a table to record data that I have gained ...
Chapter 2: Matter
... nucleus of an atoms is made of _________ &___________. The _______________ of an atom are in constant motion around the nucleus. ...
... nucleus of an atoms is made of _________ &___________. The _______________ of an atom are in constant motion around the nucleus. ...
Mid-Term OR Study Guide
... polar bonds in all formulas, show where shared electrons come from with different symbols (x’s, open and solid dots, stars, different color dots, etc.), and put loops around shared electron pairs. (A) Is the bond type between a phosphorus atom and a fluorine atom ionic, polar covalent, or nonpolar c ...
... polar bonds in all formulas, show where shared electrons come from with different symbols (x’s, open and solid dots, stars, different color dots, etc.), and put loops around shared electron pairs. (A) Is the bond type between a phosphorus atom and a fluorine atom ionic, polar covalent, or nonpolar c ...
effective nuclear charge
... in a multi-electron system, electrons are simultaneously attracted to the nucleus and repelled by each other outer electrons are shielded from full strength of nucleus ...
... in a multi-electron system, electrons are simultaneously attracted to the nucleus and repelled by each other outer electrons are shielded from full strength of nucleus ...
star test review
... (a) The amount of CaSO4 (s) will decrease, and the concentration of Ca2+ (aq) will decrease. (b) The amount of CaSO4 (s) will decrease, and the concentration of Ca2+ (aq) will increase. (c) The amount of CaSO4 (s) will increase, and the concentration of Ca2+ (aq) will decrease. (d) The amount of CaS ...
... (a) The amount of CaSO4 (s) will decrease, and the concentration of Ca2+ (aq) will decrease. (b) The amount of CaSO4 (s) will decrease, and the concentration of Ca2+ (aq) will increase. (c) The amount of CaSO4 (s) will increase, and the concentration of Ca2+ (aq) will decrease. (d) The amount of CaS ...
The Periodic table
... A region of space within an electron subshell where an electron with a specific energy is most likely to be found. S subshell=1 orbital, p subshell=3 orbitals, d subshell=5 orbitals, f subshell=7 orbitals. Maximum number of electrons in a subshell is always 2. S orbital=spherical, p orbital ...
... A region of space within an electron subshell where an electron with a specific energy is most likely to be found. S subshell=1 orbital, p subshell=3 orbitals, d subshell=5 orbitals, f subshell=7 orbitals. Maximum number of electrons in a subshell is always 2. S orbital=spherical, p orbital ...
Slide 1 of 24
... On May 6, 1937, the huge airship Hindenburg erupted into a fireball. Within a short time, 210,000 cubic meters of hydrogen had burned and the airship was destroyed. The chemical reaction that occurred is “hydrogen combines with oxygen to produce water.” You will learn to represent this chemical reac ...
... On May 6, 1937, the huge airship Hindenburg erupted into a fireball. Within a short time, 210,000 cubic meters of hydrogen had burned and the airship was destroyed. The chemical reaction that occurred is “hydrogen combines with oxygen to produce water.” You will learn to represent this chemical reac ...
C:\Users\mrh70950\Documents\My Files\WordPerfect
... i. anti-addition stereochemistry yields 1-alkenes from terminal alkynes and (E)-alkenes from internal alkynes 2. double hydrogenation: addition of 2 mol of H2 to yield alkanes a. noble metal catalyst + excess H2 3. electrophilic additions (all by very similar mechanisms) a. hydrohalogenation: addit ...
... i. anti-addition stereochemistry yields 1-alkenes from terminal alkynes and (E)-alkenes from internal alkynes 2. double hydrogenation: addition of 2 mol of H2 to yield alkanes a. noble metal catalyst + excess H2 3. electrophilic additions (all by very similar mechanisms) a. hydrohalogenation: addit ...
chemistry 2.1
... © ESA Publications (NZ) Ltd – ISBN 978-0-908340-10-1 – Copying or scanning from ESA workbooks is limited to 3% under the NZ Copyright Act. ...
... © ESA Publications (NZ) Ltd – ISBN 978-0-908340-10-1 – Copying or scanning from ESA workbooks is limited to 3% under the NZ Copyright Act. ...
exo and endo experiments
... The Law of Conservation of Mass The Law of Conservation of Mass was officially established in the year 1789 by the French Chemist, Antoine Lavoisier. The Law of Conservation of Mass states that mass is neither lost nor gained in chemical reactions, it states that it simply changes form. For that rea ...
... The Law of Conservation of Mass The Law of Conservation of Mass was officially established in the year 1789 by the French Chemist, Antoine Lavoisier. The Law of Conservation of Mass states that mass is neither lost nor gained in chemical reactions, it states that it simply changes form. For that rea ...
1 - 嘉義大學
... 20. If the concentration of the product were to double, what would happen to the equilibrium constant? (A) It would double its value. (B) It would become half its current value. (C) It would quadruple its value. (D) It would not change its value. 21. What statement about equilibrium is true? (A) Whe ...
... 20. If the concentration of the product were to double, what would happen to the equilibrium constant? (A) It would double its value. (B) It would become half its current value. (C) It would quadruple its value. (D) It would not change its value. 21. What statement about equilibrium is true? (A) Whe ...
CH225h - Oregon State chemistry
... (b) If Ar, He, and Xe are mixed with equal partial pressures in a gas sample, which will effuse most rapidly through a small hole in the container ? Explain briefly. He. Effusion is directly proportional to number density (these are all equal) and inversely proportional to the square root of molecul ...
... (b) If Ar, He, and Xe are mixed with equal partial pressures in a gas sample, which will effuse most rapidly through a small hole in the container ? Explain briefly. He. Effusion is directly proportional to number density (these are all equal) and inversely proportional to the square root of molecul ...
Balancing Equations
... 1. Word Equations—Show the names of reactants and products. Example: Sodium + Chlorine Sodium chloride 2. Chemical equations—Show the formulas of reactants and products. Example: Na + Cl2 NaCl (Not Balanced Yet!) 3. Skeleton equations—Equations that are not yet balanced to represent what actuall ...
... 1. Word Equations—Show the names of reactants and products. Example: Sodium + Chlorine Sodium chloride 2. Chemical equations—Show the formulas of reactants and products. Example: Na + Cl2 NaCl (Not Balanced Yet!) 3. Skeleton equations—Equations that are not yet balanced to represent what actuall ...
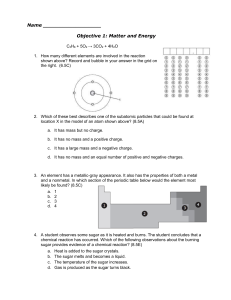
Name Objective 1: Matter and Energy C3H8 + 5O2 → 3CO2 + 4H2O
... a. C3H8 and C2H6 b. NO2 and KCl c. 2Li2S and Be4Cl2 d. 2CO and CO2 17. All of the following are indicators of a chemical change except — (8.5E) a. formation of a gas b. change in temperature c. change in the state of matter d. formation of a precipitate 18. Why is the compound CaH10P4K3O4 an inorgan ...
... a. C3H8 and C2H6 b. NO2 and KCl c. 2Li2S and Be4Cl2 d. 2CO and CO2 17. All of the following are indicators of a chemical change except — (8.5E) a. formation of a gas b. change in temperature c. change in the state of matter d. formation of a precipitate 18. Why is the compound CaH10P4K3O4 an inorgan ...
CHEMISTRY: MIDTERM EXAM REVIEW SPRING 2013 Multiple
... ____ 26. Emission of light from an atom occurs when an electron ____. a. falls into the nucleus b. moves within its atomic orbital c. jumps from a lower to a higher energy level d. drops from a higher to a lower energy level ____ 27. What must be done to be certain that a chemical change has taken ...
... ____ 26. Emission of light from an atom occurs when an electron ____. a. falls into the nucleus b. moves within its atomic orbital c. jumps from a lower to a higher energy level d. drops from a higher to a lower energy level ____ 27. What must be done to be certain that a chemical change has taken ...
Chemistry I – Fall 2004
... (C) magnetizing an iron bar (D) separating cream from milk (E) exposing photographic film to light 26. Four of these observations give evidence that air is a mixture rather than a compound. Which statement does not give such evidence? (A) Air has a variable composition. (B) Air at STP has a mass of ...
... (C) magnetizing an iron bar (D) separating cream from milk (E) exposing photographic film to light 26. Four of these observations give evidence that air is a mixture rather than a compound. Which statement does not give such evidence? (A) Air has a variable composition. (B) Air at STP has a mass of ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.